Choroidal Melanoma and Disclosing Bad News:
a Teaching Case Report
Danielle L. Weiler, OD, FAAO, and Tina R. Porzukowiak, OD, FAAO
Abstract
Choroidal melanoma is the most common primary intraocular tumor. These malignant tumors occur sporadically and are largely asymptomatic leading to diagnosis at routine eye examination. While melanoma is rare, eyecare providers should be aware of the clinical features congruent with the diagnosis of choroidal melanoma. Disclosing bad news to any patient may be difficult or stressful for a provider. Thus, in addition to reviewing the diagnostic criteria and treatment options for choroidal melanoma, this article discusses strategies for revealing bad news.
Key Words: choroidal melanoma, choroidal neoplasm, disclosing bad news, counseling a psychiatric patient, optometric education, communication skills
Background
Although choroidal melanoma is the most common primary intraocular tumor, the incidence is rare with approximately six out of one million individuals diagnosed annually or approximately 1,400 new cases in the United States each year.1,2 Choroidal melanoma typically arises in Caucasians with light-colored eyes and fair skin with a propensity to burn when exposed to ultraviolet light.1-3 The mean age at diagnosis is mid-50s.1 These malignant tumors occur sporadically and are largely asymptomatic leading to diagnosis at routine eye examination where it is important to differentiate from other lesions such as choroidal nevi.2,3 Choroidal melanomas have a high propensity to metastasize4,5 and are associated with high mortality rates.5 The liver is the most common site of metastasis followed by lung, bone, skin and subcutaneous tissue, and lymph node with five- and 10-year cumulative rates of 25% and 34%, respectively.4,6 Of those patients who die from choroidal melanoma, 90% die within 15 years.5,6 Prior to the Collaborative Ocular Melanoma Study (COMS), enucleation had been the standard of care due to the malignancy of these tumors, but improved survival rate following surgical removal of the eye had not been demonstrated.1 Since the publication of COMS, treatment of choroidal melanoma is dependent upon the size of the tumor with the goal of preserving the eye when possible.
As optometrists are likely to diagnose choroidal melanoma during their career, they must also be prepared to discuss the potential for malignancy. Disclosing bad news is a learned skill for healthcare providers. Imparting bad news to any patient may be difficult or stressful for a provider. When communicating bad news to patients, it is imperative to consider the patient’s mental health as presented in this case report. In addition to reviewing the diagnostic criteria and treatment options for choroidal melanoma, strategies for revealing bad news will be discussed. The target audience for this report is optometric educators with the target learner being third- or fourth-year optometry students.
Case Description
Initial presentation
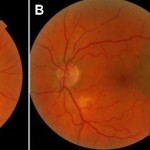
Figure 1. Fundus photographs at initial presentation, A: right eye, B: left eye.
Click to enlarge
A 65-year-old Caucasian male presented for a comprehensive eye examination in April 2013 with a chief complaint of blurred near vision in both eyes with gradual onset since his last dilated fundus exam one year prior. Ocular history was remarkable for mild cataract and dry eye syndrome in both eyes. Medical history was significant for severe bipolar disorder with psychosis, dissociative disorder, mood disorder, major depressive disorder, and history of suicidal ideations for which he was under the care of the psychiatric primary care team. Additionally, his medical history was significant for hyperglycemia, colon polyps, hypothyroidism, hypogonadism, hypercholesterolemia, hypertension, muscle cramps, osteoarthritis and sleep apnea. For these conditions, he was taking aripiprazole (Abilify), atorvastatin (Lipitor), cyclobenzaprine (Flexeril), diclofenac (Zorvolex), hydroxyzine (Vistaril), lamotrigine (Lamictal), levothyroxine (Synthroid), sertraline (Zoloft), simethicone (Gas-X) and testosterone intramuscular injections (Delatestryl). He reported an allergy to naproxen. His family history was significant for glaucoma in his maternal grandmother. He quit smoking in 1985 and reported no history of recreational drug and alcohol use. He was oriented to time, place and person with a flat affect.
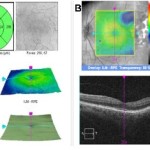 Figure 2. Spectral domain OCT macular cube (A: right eye, B: left eye): distinct foveal pit OU. |
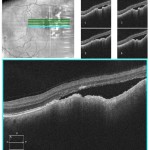 Figure 3. High-definition spectral domain OCT through the lesion OS revealing neurosensory detachment with underlying choroidal lesion with no internal reflectivity; findings consistent with choroidal melanoma. |
Best-corrected visual acuities were 20/20- right eye (OD) and 20/20 left eye (OS). Pupils were equally round and reactive to light without an afferent pupillary defect. Extraocular muscles were smooth and full in both eyes (OU). Confrontation visual fields were full to finger counting OD, OS. Slit lamp examination revealed 1+ nuclear sclerotic cataract OU and 1+ anterior cortical cataract OD. Intraocular pressures (IOPs) by Goldmann applanation tonometry were 16 mmHg OD, OS at 2:15 p.m. Dilated funduscopic examination revealed a 4DD vertical x 5DD horizontal pigmented, elevated choroidal lesion with overlying orange pigment temporal to the fovea OS (Figure 1) but was otherwise unremarkable. Optical coherence tomography (OCT) was consistent with choroidal melanoma OS (Figures 2-3).
At the end of the examination, the patient was brought into the attending office to arrange for a private setting, to minimize interruptions, and to allow for more time with the patient. He was asked if any family members were present that he would like to include in the conversation. Both parties were seated at eye level. He was asked if he knew the reason for the additional testing that had been performed. The patient was shown photographs of the new choroidal lesion and advised of clinical suspicion for malignancy and the need for additional work-up. As the patient did not show any emotion throughout the discussion about possible malignancy, he was asked if he comprehended to which he voiced understanding. Given his history of suicidal ideation, he was asked if he would like to speak to the clinical mental health care coordinator prior to leaving his appointment, but he declined this service. Finally, the need for an urgent referral to Retina was discussed with the patient, but coordination of care was not possible while the patient was in-office because the specialist was in the operating room. Therefore, the patient was informed that he would receive a phone call with an appointment. Liver function tests and chest x-ray were ordered, and the patient was advised to have these completed on the same day as the Retina appointment. Additionally, his primary care team was notified of his ocular findings to provide interdisciplinary support as needed.
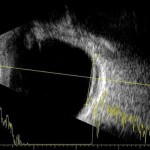
Figure 4. Ultrasonography OS: A-scan and B-scan confirming low internal reflectivity of the choroidal lesion and no associated subretinal fluid.
Click to enlarge
The following day, Retina was not able to reach the patient via phone to schedule an appointment, so the technician asked the authors for assistance with coordinating his care. Because of the patient’s flat affect and extensive psychiatric history, the authors became concerned for the patient’s welfare. Thankfully, his wife confirmed the patient’s well-being and stated she would bring him to the Retina appointment that afternoon. Ultrasonography of the choroidal lesion (Figure 4) revealed a diameter of 10.2 mm, thickness of 2.6 mm, low internal reflectivity (ultrasonic hollowness), and no associated subretinal fluid. The retinal specialist agreed with the provisional diagnosis of choroidal melanoma and referred the patient to a retinal oncologist.
Retinal oncology consultation
Two weeks later, the patient’s vision remained stable (OD 20/25, OS 20/40 pinhole 20/20). The choroidal lesion measured 7.25 mm in its greatest diameter with a height of 1.26 mm with ultrasonography. OCT demonstrated subretinal fluid, and fluorescein angiography showed hyperfluoresecence with stippled mottling of the left temporal posterior pole. Liver function tests (Table 1) and chest x-ray were normal. The impression of the retinal oncologist was a small choroidal melanoma OS. Due to the melanoma’s location adjacent to the fovea, the retinal oncologist and patient opted for observation for documented growth as treatment would likely have deleterious effects on the patient’s vision.
 Figure 5. Fundus photography OS nine months following initial presentation. |
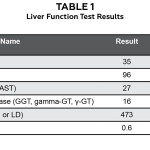 Table 1 |
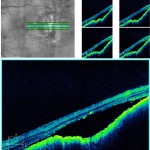 Figure 6. High-definition spectral domain OCT through choroidal melanoma OS revealing neurosensory detachment with subretinal fluid. |
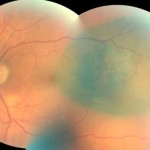 Figure 7. Fundus photography OS two months after brachytherapy (artifact at inferior-temporal vascular arcade). |
Subsequent follow-up visit
The patient returned to the authors’ care nine months following his initial presentation stating that his left eye had become painful approximately one week prior. He described the pain as a constant dull ache of the anterior segment. He reported that he had two additional visits with the retinal oncologist with no changes in the choroidal melanoma. He reported no changes to his medical history, medications or allergies. Visual acuities were 20/25 OD and 20/30-2 pinhole 20/25 OS. Pupils remained equally round and reactive to light without an afferent pupillary defect. External and slit lamp examination were stable OU. IOP by Goldmann applanation tonometry was 13 mmHg OD, OS at 1:48 p.m. Dilated fundus examination revealed interval growth of the choroidal melanoma (Figure 5) and OCT showed subretinal fluid (Figure 6). The patient was informed the cause of his eye pain was from dry eye syndrome for which he was instructed to use warm compresses with lid massage and prescribed carboxymethylcellulose 0.5% ophthalmic solution four times daily in both eyes. Additionally, he was counseled regarding the growth of the melanoma and the need to return to the retinal oncologist for treatment despite possible vision loss.
At his retinal oncology visit the following week, plaque brachytherapy was recommended. Additionally, computed tomography (CT) of the chest and abdomen to rule out metastasis was ordered, and radiation oncology was consulted. CT of the chest and abdomen was stable without evidence of metastatic disease. Radiation oncology reported the tumor had a basal diameter of 8.4 mm by 5.5 mm, height of 1.74 mm, and macula-to-tumor margin of 0 mm. They recommended delivering a dose of 85 Gy to the tumor apex with a 14-mm I-125 plaque and indicated they would coordinate a surgery date with the retinal oncologist. The patient underwent fine-needle biopsy and plaque brachytherapy placed at the tumor apex for one week. The fine-needle biopsy was sent for DecisionDx-UM classification and found to be Class 1A which is associated with a low risk of clinical metastasis within five years.
The patient returned to the authors’ care two months after plaque brachytherapy with a complaint of double vision following the surgery that had since resolved. He reported no changes to his medical history, medications or allergies. Best-corrected visual acuities were 20/25 OD and 20/20-2 OS. Pupils remained equally round and reactive to light without an afferent pupillary defect. Extraocular movements were full OU and confrontation visual fields were full to finger counting OD, OS. External and slit lamp examination were stable OU. IOP by Goldmann applanation tonometry was 13 mmHg OD, OS at 8:14 a.m. Dilated fundus examination revealed a decrease in the overlying orange pigment and stable size of the melanoma (Figure 7). The patient was reassured that his visual acuity had not been affected by plaque brachytherapy, but the cataract in his right eye was causing a slight decrease in vision. He was instructed to continue care with the retinal oncologist as directed and to follow-up with the authors in one year or as needed if he experienced a change in vision.
Education Guide
The education guide contains discussion points to help facilitate the thoughtful discussion of the case.
Key concepts
- Recognition of clinical signs of choroidal melanoma
- Understanding timely referral to retinal oncology and systemic work-up for choroidal melanoma
- The importance of individualized approach to breaking bad news
- The simulation of communicating bad news improves future clinical outcomes
Learning objectives
At the conclusion of this learning activity, participants should be able to:
- Describe the diagnostic criteria of choroidal melanoma
- Review the management of choroidal melanoma
- Understand the key components to effectively impart bad news to patients
- Become familiar with the skills required to deliver bad news to patients
Discussion questions
- Knowledge, concepts, facts and information required for critical review of the case
- Does the patient fit the typical demographics for the ocular diagnosis?
- Does the patient’s general health play a role in the ocular diagnosis?
- What are the clinical characteristics of choroidal melanoma that help differentiate it from choroidal nevus?
- Differential diagnosis
- What is the differential diagnosis (after case history and after the ocular health exam)?
- What tests should be performed to narrow the differential diagnosis?
- Patient management
- What treatment would be most appropriate for the patient?
- How quickly should the patient undergo treatment?
- How should the patient be managed after treatment?
- Communication with the patient regarding diagnosis, prognosis, treatment options and potential sequela
- What are the important elements of breaking bad news?
- How should the patient be educated regarding the diagnosis and prognosis?
- Given the patient’s known psychiatric disease, how does this change the approach to the delivery of bad news?
- What is the risk of the fellow eye developing the same condition?
- What aspects of treatment may be challenging for the patient?
- What potential treatment side effects should the patient be familiar with?
- Critical thinking
- What were the complicating factors in this care?
- How would you have managed this case differently?
Learning assessment
- Knowledge base of the condition can be tested via photos, OCT, ultrasonography
- Knowledge base of the differential diagnosis can be tested through development of a table comparing characteristics of different posterior segment pigmented lesions
- Critical thinking skills can be assessed by case reports that are either hypothetical examples or from a review of the literature
- The skills required to relay bad news can be assessed via small group discussion, role-playing with a trained simulated patient, or during teaching moments in clinic
Discussion
The purpose of this case report is to help clinical novices review the clinical findings congruent with choroidal melanoma and develop strategies to break bad news. A faculty member or teaching assistant could lead a discussion of the case presentation in either a large classroom setting or small group. In the course of the discussion, students should be given the initial case presentation in a step-wise fashion (i.e., history, fundus photo, ocular coherence tomography, ultrasonography). This will enable the learner to critically think through the clinical presentation and differential diagnosis.
Following the discussion to arrive at the ocular diagnosis, the key components of breaking bad news can be discussed. After this didactic component, students can be given the opportunity to role-play breaking bad news and be given constructive feedback from the exercise. This case provides an additional layer of complexity given the patient’s psychiatric history, but it allows for students to practice incorporating an individualized approach to breaking bad news. Some educators may view this additional complexity as a limitation depending upon the level of the trainee, so the psychiatric history could be pared down at the discretion of the instructor.
Choroidal melanoma
Choroidal melanomas are malignant tumors arising from abnormal proliferation of pigmented cells in the choroid. In comparison, choroidal nevi are benign melanocytic lesions of the choroid. Making the correct diagnosis of choroidal nevus vs. choroidal melanoma is crucial. In this case, the patient presented for an annual comprehensive eye exam with a 4 DD vertical x 5 DD horizontal pigmented, elevated choroidal lesion with overlying orange pigment temporal to the fovea OS that had not been present on the dilated fundus exam one year prior. The primary differential diagnosis considered in this case was choroidal nevus. Additional differential diagnoses for pigmented lesions of the posterior segment include congenital hypertrophy of the retinal pigment epithelium (RPE), adenoma or adenocarcinoma of the RPE, reactive hyperplasia of the RPE, congenital simple hamartoma of the RPE, combined hamartoma of the retina and RPE, and choroidal detachment.7
 Table 2 |
 Table 3 |
Several mnemonics for characterizing lesions, including ABCDE as used in dermatology, have been described in the literature. With the ABCDE mnemonic, a provider looks at the following clinical characteristics of the lesion to determine suspicion for malignancy: asymmetry, borders, color, diameter and elevation. More specific to eye care, the mnemonic “To Find Small Ocular Melanoma Using Helpful Hints Daily” (TFSOM-UHHD) described by Shields et al.8 is outlined in Table 2. These TFSOM-UHHD risk factors may be used in predicting the malignant transformation of choroidal lesions thereby directing a management plan. The presence of three or more risk factors is associated with a greater than 50% change of tumor growth in five years, whereas the absence of all risk factors results in a 3% chance.8,9 The most reliable way to diagnose choroidal melanoma is clinical examination; however, ultrasonography, fluorescein angiography, indocyanine green angiography, enhanced depth OCT, autofluorescence, and fine-needle biopsy can also be helpful.10
The COMS defined choroidal melanomas by size and delineated the optimal treatment for each (Table 3). More recently, the American Joint Committee on Cancer proposed staging choroidal melanomas based upon intraocular examination, serum tests (complete blood count and liver function tests) and imaging.11 The use of baseline imaging is controversial because the yield of finding metastasis is low; however, patients who demonstrate metastasis at presentation are often spared aggressive treatment of the primary lesion.11 When opting to order baseline imaging, one of the following protocols should be used: 1) CT of the chest and abdomen with liver protocol, 2) whole body positron-emission tomography (PET)-CT, or 3) liver magnetic resonance imaging (MRI) and chest CT.11
The management of melanoma depends upon the tumor size, tumor location, associated features, status of the fellow eye, patient’s systemic status and patient’s desire.10 Management choices for choroidal melanoma include transpupillary thermotherapy, plaque radiotherapy, charge particle irradiation, local resection, enucleation or orbital exenteration.10 The management of small choroidal melanomas remains controversial. Observation can delay treatment and increase the risk of metastasis11 by a factor up to eight if TFSOM-UHHD risk factors are present.12 With this said, the possibility of vision loss from treatment must be considered, such as in this case. However, recent reports do not recommend observation except in unique situations such as in an elderly, medically unstable patient, a patient with advanced non-ocular malignancy, or a patient with vision loss in one eye and small- to medium-sized melanoma in the better seeing eye.11,13 Since the publication of the COMS trial results, brachytherapy has become the treatment of choice for choroidal melanomas with a diameter less than 18 mm and apical height less than 12 mm.13 Brachytherapy is a type of radiotherapy where a high concentration of radiation is delivered locally to the tumor.3 The choice of isotope is based upon the tumor size, tumor depth and location of the melanoma.3,11 However, brachytherapy is not without complication, including cataract formation, neovascular glaucoma, radiation retionopathy, maculopathy and optic neuropathy.14 Additionally, Sener et al. prospectively evaluated for ocular motility disturbances following plaque brachytherapy and postulated that patients could experience transient diplopia following the procedure as is seen with glaucoma implants.15 This could account for the postoperative diplopia this patient experienced.
Disclosing bad news and counseling a psychiatric patient
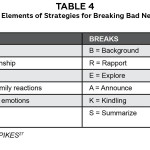
Table 4
Click to enlarge
Sharing bad news with patients is necessary in any medical field. Many providers feel under-trained and uncomfortable communicating bad news,16 and the evidence base for best practices in imparting bad news is limited.17 However, a growing body of evidence shows physicians’ communication skills play a crucial role in how well patients cope with bad news.18,19 Thus, delivery of bad news requires not only knowledge of the diagnosis but also fundamental communication competency and professionalism.20 Bad news is any information that could negatively impact a patient’s expectations about their present or future.16,21 There are individual differences in how patients perceive bad news based upon their life experience, personality, spiritual beliefs, philosophical standpoint, perceived social supports, emotional hardiness and mental capacity.16,22,23 These important considerations must be weighed against the importance of building trust in the provider-patient relationship with disclosure of bad news.24 With this said, revealing bad news does not need to be a single event but can be a gradual building of knowledge.17 Additionally, a multidisciplinary approach to offering support to the patient may be beneficial.
Several guidelines such as ABCDE,25 BREAKS26 and SPIKES27 have been developed to prepare healthcare providers to impart bad news. The key elements of each guideline are summarized in Table 4. These guidelines divide the interaction into three basic steps:28 preparation, the information itself, and an empathic response. SPIKES is a six-step strategy developed by oncologists27 and is the most popular framework.19,29 Although each step is not required with every episode of disclosing bad news, the steps are meant to be followed sequentially.27 Some limitations of the SPIKES protocol include that it may not be useful for all healthcare professions such as nursing, the communication strategy is too formulaic, and it is unclear if the strategy follows patient preferences for receiving bad news.19,30 Ultimately, this recommendation is meant to serve as a general guide and should not be viewed as overly prescriptive.18 Medical students who had an experience breaking bad news to a trained standardized patient followed by feedback performed better on a clinical proficiency than students without this experience.31 In optometry, most outpatient visits are well-suited for incorporating this method when breaking bad news; however, it may be difficult in the midst of a hectic clinic schedule to ensure there is adequate time in a private setting with no interruptions. Key elements from SPIKES with patient communication examples are outlined in Table 5.

Table 5
Click to enlarge
In this case, the patient had multiple psychiatric diagnoses making it unclear how he would perceive bad news, and the authors became concerned for his welfare when the retina clinic was unable to contact him. In general, psychiatric patients may be uncooperative or hostile, may be at increased risk for suicide, or may become distressed more easily than the general population leading to some information being withheld.23 Despite this, most psychiatric patients and providers alike believe patients have a right to information about their treatment and diagnosis.23,32
In the psychiatric literature, there are two similar yet distinct personality types that could apply to this patient based on his flat affect with short answers throughout his exam: aloof or avoidant. The uninvolved-aloof personality type has the tendency toward isolation and may give an impression of aloofness and solitariness.21 While the aloof person seems indifferent, he or she uses isolation for protection from painful experiences and emotion.21 When communicating with aloof patients, providers should keep in mind that these patients are sensitive and fragile despite their apathetic demeanor, but uninvolved-aloof patients may accept family involvement.21 In contrast, the avoidant personality type exhibits social inhibition and avoidance of social interaction.33 The avoidant person is hypersensitive to criticism and has a fear of negative evaluation.33 Thus, the avoidant person has profound anxiety about exposure of perceived weakness and being considered inferior.33 Avoidant patients would appreciate gradual disclosure of information and are prone to fantasy with fear of illness becoming greater in their imagination.33 The avoidant patient may have difficulty disclosing these oversized fantasies, which could lead to suicidal ideation.33
Conclusion
Choroidal melanoma, while rare, is the most common primary intraocular tumor. It is essential for primary eyecare providers to be aware not only of the clinical features congruent with these malignant tumors but also to be prepared to break bad news to these patients. Using the SPIKES six-step strategy for imparting bad news will assist in building trust in the doctor-patient relationship.
References
- Margo CE. The Collaborative Ocular Melanoma Study: an overview. Cancer Control. 2004;11(5):304-9.
- Cheung A, Scott I, Murray T, Shields C. Distinguishing a choroidal nevus from a choroidal melanoma; ophthalmology pearls, EyeNet Magazine 2012. [04/14/2016]. Available from: https://www.aao.org/eyenet/article/distinguishing-choroidal-nevus-from-choroidal-mela?february-2012
- Hirata C, Wolfe G, Grimes A. Choroidal melanoma: A case report and updated review of new diagnostic tests and treatments. J Am Optom Assoc. 2012;83(6)(June 2012):219-24.
- Diener-West M, Reynolds SM, Agugliaro DJ, Caldwell R, Cumming K, Earle JD, et al. Development of metastatic disease after enrollment in the COMS trials for treatment of choroidal melanoma: Collaborative Ocular Melanoma Study Group Report No. 26. Arch Ophthalmol. 2005;123(12):1639-43.
- Kujala E, Makitie T, Kivela T. Very long-term prognosis of patients with malignant uveal melanoma. Invest Ophthalmol Vis Sci. 2003;44(11):4651-9.
- Kaliki S, Shields CL. Uveal melanoma: relatively rare but deadly cancer. Eye (Lond). 2016.
- Ly A, Nivison-Smith L, Hennessy M, Kalloniatis M. Pigmented lesions of the retinal pigment epithelium. Optom Vis Sci. 2015;92(8):844-57.
- Shields CL, Furuta M, Berman EL, Zahler JD, Hoberman DM, Dinh DH, et al. Choroidal nevus transformation into melanoma: analysis of 2514 consecutive cases. Arch Ophthalmol. 2009;127(8):981-7.
- Shields CL, Cater J, Shields JA, Singh AD, Santos MC, Carvalho C. Combination of clinical factors predictive of growth of small choroidal melanocytic tumors. Arch Ophthalmol. 2000;118(3):360-4.
- Shields CL, Manalac J, Das C, Ferguson K, Shields JA. Choroidal melanoma: clinical features, classification, and top 10 pseudomelanomas. Curr Opin Ophthalmol. 2014;25(3):177-85.
- Weis E, Salopek TG, McKinnon JG, et al. Management of uveal melanoma: a consensus-based provincial clinical practice guideline. Curr Oncol. 2016;23(1):e57-64.
- Shields CL, Shields JA, Kiratli H, De Potter P, Cater JR. Risk factors for growth and metastasis of small choroidal melanocytic lesions. Ophthalmology. 1995;102(9):1351-61.
- Shields JA, Shields CL. Management of posterior uveal melanoma: past, present, and future: the 2014 Charles L. Schepens lecture. Ophthalmology. 2015;122(2):414-28.
- Wen JC, Oliver SC, McCannel TA. Ocular complications following I-125 brachytherapy for choroidal melanoma. Eye (Lond). 2009;23(6):1254-68.
- Sener EC, Kiratli H, Gedik S, Sanac AS. Ocular motility disturbances after episcleral plaque brachytherapy for uveal melanoma. J AAPOS. 2004;8(1):38-45.
- Fallowfield L, Jenkins V. Communicating sad, bad, and difficult news in medicine. Lancet. 2004;363(9405):312-9.
- Tuffrey-Wijne I. A new model for breaking bad news to people with intellectual disabilities. Palliat Med. 2013;27(1):5-12.
- VandeKieft GK. Breaking bad news. Am Fam Physician. 2001;64(12):1975-8.
- Seifart C, Hofmann M, Bar T, Riera Knorrenschild J, Seifart U, Rief W. Breaking bad news-what patients want and what they get: evaluating the SPIKES protocol in Germany. Ann Oncol. 2014;25(3):707-11.
- Guneri P, Epstein J, Botto RW. Breaking bad medical news in a dental care setting. J Am Dent Assoc. 2013;144(4):381-6.
- Kallergis G. Informing cancer patient based on his type of personality: the uninvolved – aloof patient. J BUON. 2014;19(1):278-82.
- Kobayashi T, Kato S, Takeuchi M. Considering patients’ mental capacity when giving them bad news may help their well-being: a case of suicide attempt after being informed of lung cancer diagnosis. Case Rep Psychiatry. 2014;2014:645769.
- Cleary M, Hunt GE, Walter G. Delivering difficult news. Views of mental health staff in inpatient settings. J Psychosoc Nurs Ment Health Serv. 2010;48(6):32-9.
- Horikawa N, Yamazaki T, Sagawa M, Nagata T. The disclosure of information to cancer patients and its relationship to their mental state in a consultation-liaison psychiatry setting in Japan. Gen Hosp Psychiatry. 1999;21(5):368-73.
- Rabow MW, McPhee SJ. Beyond breaking bad news: how to help patients who suffer. West J Med. 1999;171(4):260-3.
- Narayanan V, Bista B, Koshy C. ‘BREAKS’ Protocol for Breaking Bad News. Indian J Palliat Care. 2010;16(2):61-5.
- Baile WF, Buckman R, Lenzi R, Glober G, Beale EA, Kudelka AP. SPIKES-A six-step protocol for delivering bad news: application to the patient with cancer. Oncologist. 2000;5(4):302-11.
- Alelwani SM, Ahmed YA. Medical training for communication of bad news: A literature review. J Educ Health Promot. 2014;3:51.
- Dunning R, Laidlaw A. The application of the Practitioners in Applied Practice Model during breaking bad news communication training for medical students: a case study. Scott Med J. 2015;60(4):170-5.
- Dean A, Willis S. The use of protocol in breaking bad news: evidence and ethos. Int J Palliat Nurs. 2016;22(6):265-71.
- Colletti L, Gruppen L, Barclay M, Stern D. Teaching students to break bad news. Am J Surg. 2001;182(1):20-3.
- Cleary M, Hunt GE, Escott P, Walter G. Receiving difficult news. Views of patients in an inpatient setting. J Psychosoc Nurs Ment Health Serv. 2010;48(6):40-8.
- Kallergis G. Informing cancer patient based on his type of personality: the avoidant patient. J BUON. 2013;18(2):527-31.




