PEER REVIEWED
Phlyctenular Keratoconjunctivitis
Lindsay A. Sicks, OD, FAAO, FIACLE, FSLS, and Jennifer S. Harthan, OD, FAAO, FSLS
Abstract
This case report demonstrates the importance of proper identification and management of phlyctenular keratoconjunctivitis and pediatric blepharokeratoconjunctivitis to prevent unnecessary visual compromise.
Key Words: phlyctenular keratoconjunctivitis, pediatric blepharokeratoconjunctivitis, blepharitis, corneal neovascularization, corneal scarring, oral antibiotics
Background
Phlyctenular keratoconjunctivitis (PKC) is an acute localized nodular inflammation of the conjunctiva, limbus or peripheral cornea.1-2 It is a delayed-type (IV) hypersensitivity reaction to foreign microbial proteins.3-4 The traditional association in developing countries is with Mycobacterium tuberculosis (sensitivity to tuberculin protein). In developed countries, the most common cause is non-tuberculous hypersensitivity to Staphylococcus aureus.5-12 In North America, an association with tuberculosis has been reported among the Inuit and Native American populations of Alaska and Canada.10,13-14 PKC also has associations with chlamydia, gonorrhea, herpes simplex virus and parasitic intestinal infection.11,15-17
A phlyctenule is a focal, subepithelial inflammatory nodule of the eye, commonly found at the limbus (in the interpalpebral region) or on the bulbar conjunctiva.1,18 Repeat episodes of inflammation at the limbus can cause the phlyctenule to progress across the corneal surface (a “wandering phlyctenule”) with an elevated leading edge trailed by neovascularization.1,4,9,19 Inflammatory cells found within phlyctenules include macrophages, lymphocytes, plasma cells and polymorphonuclear leukocytes.1
Conjunctival phlyctenules cause mild to moderate symptoms such as blurred vision, tearing, foreign body sensation and itching. Corneal phlyctenules can progress to ulceration and tend to present with more moderate to severe eye pain, photophobia or blepharospasm.1,4,20-21 Complications of PKC can include corneal ulceration, neovascularization, fibrosis, scarring, reduced vision and amblyopia. Secondary infection is possible if an epithelial defect is present. Corneal melting and perforation are rare, but potential, complications of persistent inflammation.7,9,22-23
Pediatric blepharokeratoconjunctivitis (BKC) is a spectrum of chronic anterior segment inflammatory disorders in children that can lead to loss of vision.23-24 Within the past decade, the term pediatric blepharokeratoconjunctivitis has been used to describe the wide range of clinical manifestations that present with varying degrees of lid margin inflammation, meibomian gland dysfunction, conjunctival hyperemia, recurrent chalazia, conjunctival or corneal phlyctenules and keratitis, which can lead to corneal neovascularization and scarring.23-24 Some of the clinical entities encompassed under the broad term BKC include phlyctenular keratoconjunctivitis, ocular (acne) rosacea keratoconjunctivitis, atopic keratoconjunctivitis and marginal keratitis, among others.25 Still, some suggest that eyecare practitioners should label each clinical presentation individually and treat them separately, rather than combine all the inflammatory disorders into a single, perhaps poorly defined, category.25
Management of BKC involves suppressing the inflammatory response and reducing the antigens involved in the inflammatory process. It often requires a two-pronged approach with both anti-inflammatory and antibiotic therapies.26 Additionally, warm compresses are recommended for all patients.27 A majority of patients (97%) are prescribed topical antibiotic ointment (bacitracin or erythromycin).27 Other common additional therapies for BKC include topical steroids such as dexamethasone, prednisolone, loteprednol or fluorometholone, and/or one of a variety of oral antibiotics such as macrolides (erythromycin or azithromycin), tetracyclines (doxycycline, tetracycline or minocycline) or penicillins (amoxicillin/clavulanate or dicloxacillin).1,12,24,28-29 Recalcitrant, recurrent or steroid-dependent cases may benefit from more aggressive topical therapy, such as topical cyclosporine (standard or compounded preparations) or topical tacrolimus.1,24,26,29-31
Long-term maintenance of lid hygiene therapy for any associated chronic blepharitis is another critical component to continued success.24,26 Visual sequelae, such as reduced visual acuity or a large degree of anisometropia, can be successfully managed in many cases with spectacle lenses and standard contact lens designs. In cases of corneal irregularity or residual scarring, the prescription of specialty contact lens designs may optimize visual outcomes.32-37 If the corneal scarring is severe, referral for penetrating keratoplasty may also be warranted; however, corneal transplantation in children is challenging.26,38-39 Additionally, any eye prone to inflammation may have an increased risk of transplant rejection.26
This teaching case report addresses the management of a symptomatic presentation of PKC in the setting of pediatric BKC. The treatment of choice for any corneal or conjunctival phlyctenule is a topical corticosteroid to suppress ocular surface inflammation and prevent corneal scarring. Topical and oral antibiotics are also often prescribed for pediatric patients with PKC in order to manage the bioburden and inflammation related to their co-existing BKC. It is crucial to examine the potential side effects of both of these treatment options and to determine the proper dosage for any oral medications. This case will also review the potential visual sequelae of PKC and considerations for successful management with contact lenses after resolution.
This teaching case report is appropriate for third-year optometry students who have completed didactic coursework in infectious and inflammatory cornea and anterior segment disease, dry eye disease and blepharitis. Students should be proficient at slit lamp evaluation and corneal assessment. Some knowledge of didactic coursework in contact lenses and pediatric prescribing may be useful but is not required. This teaching case report would also be appropriate for fourth-year optometry students as a journal club/reading assignment in primary care, anterior segment or contact lens rotations. Optometry residents in primary eye care, ocular disease, pediatric optometry or cornea and contact lenses may also find this material useful.
Case Description
Initial visit
A 12-year-old female presented as a referral from an outside provider for evaluation and treatment of “large pannus, scarring and corneal neovascularization OS.”
The patient’s chief complaint was irritation and associated foreign body sensation OS only. She had also noticed redness and watery discharge from that eye. She described episodes of minor pain and itching OS. All symptoms had been present intermittently for the past 6 months but had worsened in the past week. She denied any history of chalazia. She denied any breathing problems or recent illness. There was no systemic disease history, and the patient denied having known tuberculosis (noting a recent negative tuberculin sensitivity test). She had no known allergies to any drugs or medication. She denied any other medical or ocular history, and there was no family history of any ocular disease. She was not taking any medications.
Uncorrected distance visual acuities were OD 20/200 and OS 20/80. With her habitual spectacles, visual acuities were OD 20/30-1 and OS 20/40. Entrance tests (color vision, cover test, extraocular muscle balance, pupils and confrontation visual fields) were within normal limits OU.
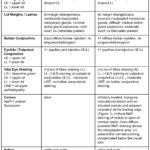 Table 1. |
 Table 2. |
Retinoscopy results were OD -2.75 -1.00 x 180, 20/25, and OS -1.00 -0.75 x 170, 20/30. Distance-balanced subjective refraction of OD -3.25 -0.75 x 180 improved vision to 20/20, and OS -0.75 -0.75 x 165 improved vision to 20/25.
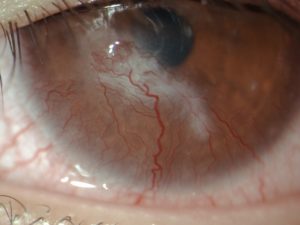
Figure 1. Initial presentation of elevated corneal phlyctenule with surrounding neovascularization and associated scarring, which demonstrates a classic triangular shape.2 Click to enlarge
Anterior segment evaluation was performed with slit lamp biomicroscopy. Table 1 lists the findings. Meibography with the Oculus Keratograph 5M (Oculus, Wetzlar, Germany) showed Grade 1 meibomian gland dropout on the lower lids OD and OS, along with Grade 1 dropout on the upper lid OD and Grade 3 dropout on the upper lid OS, according to the JENVIS Meibo Grading Scale (JENVIS Vision Research, Jena, Germany). Baseline anterior segment photographs were taken to demonstrate elements of the anterior segment condition to the patient and parent (Figure 1). The patient’s intraocular pressure (IOP) readings by Goldmann applanation tonometry were OD 14 mmHg and OS 16 mmHg. After the instillation of one drop of 1.0% tropicamide and one drop of 2.5% phenylephrine in each eye, a dilated fundus examination revealed no abnormalities.
The patient was diagnosed with 1) phlyctenular keratoconjunctivitis OS, 2) pediatric blepharokeratoconjunctivitis OU, 3) chronic allergic conjunctivitis OU, and 4) paracentral corneal opacity OS. The initial treatment recommendations, including the purpose of each treatment, specific therapies prescribed and the management goals, are outlined in Table 2. The treatment of choice for PKC is a topical steroid, so the patient was prescribed prednisolone acetate 1.0% ophthalmic suspension 4 times a day OS only. Because the dose uniformity of an ophthalmic suspension depends on the homogeneity of the liquid at the time of administration, the patient was advised to shake the dropper bottle thoroughly before each use.40 To manage the BKC, the patient was advised to begin lid hygiene with warm compresses using the “bundle towel” method (heating several moistened, folded, washcloth-sized towels, wrapped together into a circular bundle) and lid scrubs OU with an individually wrapped, pre-moistened lid scrub pad. She was also prescribed bacitracin ophthalmic ointment each night at bedtime OU and oral azithromycin 250 mg/day. In addition, she was started on a short-term dose of ketotifen fumarate 0.025% ophthalmic solution twice a day OU to gauge any improvement in the signs or symptoms of ocular allergy. Preservative-free artificial tears (PFATs) were also recommended as needed up to 4 times a day OU. She was asked to return for a follow-up visit, with an IOP check, in 2 weeks. We reviewed the chronic nature of BKC with the patient and parent and reviewed the potential side effects of therapy. We advised that some of the treatments may continue for 3-6 months or more depending on the clinical course of the disease. The need for aggressive management due to the risk of sight-threatening sequelae was also discussed. A follow-up visit was set for 2 weeks later.
2-week follow-up visit
At the 2-week follow-up visit, the patient’s symptoms had improved. She had been unable to obtain the prescribed ketotifen but noted compliance with all other medications, aside from minor lapses in lid scrub use. There were no remaining subjective symptoms of eye redness or irritation OS. Refraction and best-corrected vision were stable at this visit, although the patient noted improved subjective visual acuity OS.

Figure 2. Clinical appearance after 2 weeks of treatment. Click to enlarge
On slit lamp evaluation, the lids still had mild scurf present with stable lid margin telangiectasia, moderately inspissated meibomian glands, and minimal turbid gland expression. There was still 1+ injection and 1+ papillary response on both palpebral conjunctiva. The corneal appearance had improved significantly, with regression of the corneal phlyctenule (Figure 2). Trace interpalpebral punctate staining OU, but no focal staining, was visible with sodium fluorescein. A dense scar in the inferior paracentral cornea and some inferior neovascularization with visible blood flow were present. IOP was stable at 14 mmHg OD and 14 mmHg OS.
The patient was tolerating the topical prednisolone acetate 1.0% OS and the oral azithromycin well with no side effects; therefore, they were continued at the same dosage for 2 more weeks to control the PKC. Due to the presence of persistent lid scurf and meibomian gland dysfunction OU and punctate staining OS, the warm compresses, lid scrubs, bacitracin ointment and PFATs were continued at the same dosing schedules to ensure continued management of the BKC. The patient was asked to try to obtain the ketotifen from her local pharmacy to manage the allergic response. She was also asked to look into the purchase of over-the-counter 0.02% hypochlorous acid solution spray, a topical preparation with broad-spectrum antimicrobial activity, to add to her lid hygiene regimen twice daily. This spray could be used after lid scrubs, which remove bulky debris, to further reduce the bacterial bioburden on the lids and lashes and reduce the antigens involved in the inflammatory process, thereby controlling the overall inflammatory response seen in BKC. We again reviewed the chronic nature of BKC with the patient and parent, noting the importance of long-term adherence to lid hygiene procedures. The next follow-up visit was set for 2 weeks later to allow for close observation.
1-month follow-up visit
At the 1-month follow-up appointment, the patient was symptom-free. She had been using the ketotifen twice a day OU for the past week but did not feel as though it made much of a difference. She was able to obtain the hypochlorous acid solution spray but had not started using it. She noted compliance with all other aspects of her regimen.
On slit lamp evaluation, the lids still had trace scurf present. There was still 1+ injection and 1+ papillary response on both palpebral conjunctiva. The corneal appearance was stable with persistent trace interpalpebral punctate staining OS. The patient was confirmed to have complete lid closure OU (i.e., no lagophthalmos was present). The inferior corneal neovascularization OS still showed visible blood flow but had improved slightly since the previous visit. The IOPs were stable at 12 mmHg OD and 13 mmHg OS.
We elected to continue the prednisolone acetate 1.0% at 4 times a day OS and oral azithromycin due to the persistent neovascularization and lid appearance. Warm compresses twice a day OU and bacitracin ointment each night at bedtime OU were continued, but the patient was advised she could reduce the lid scrubs to once a day in the morning OU and add in the hypochlorous acid solution spray twice a day OU. Because she had only been using ketotifen twice a day OU for 1 week, that was continued. Because punctate corneal staining that could be attributed to the use of preserved drops 6 times daily persisted, the dosing of PFATs was increased to 6 times daily OU with hopes of tapering some medications at the next follow-up in 1 month.
2-month follow-up visit
At the 2-month follow-up appointment, the patient remained symptom-free. She noted compliance with her medications and lid hygiene regimen but had only been using the PFATs 3 times a day OU and had yet to start the hypochlorous acid solution spray.
On slit lamp evaluation, the lids had minimal scurf. There was trace injection and a 1+ papillary response on both upper and lower lids OU. The corneal appearance was improved with very trace punctate staining OS and almost complete resolution of the inferior neovascularization OS. Inactive (ghost) vessels remained in the inferior cornea, but no active blood flow was visible. The IOPs were unequal at 13 mmHg OD and 18 mmHg OS.
Due to the elevated IOP OS and resolution of the neovascularization, a topical steroid taper was initiated with dosing at 3 times a day OS for a week, then twice a day OS for a week, then once a day for a week, stopping the medication before the next follow-up in 1 month. The oral azithromycin was discontinued due to improvement in the lids, lashes and corneal appearance and the now quiescent corneal neovascularization. All lid hygiene measures were continued, including warm compresses twice a day OU, lid scrubs once a day in the morning OU and bacitracin ointment each night at bedtime OU. Because the patient had not yet started using the hypochlorous acid solution spray, it was again prescribed at twice a day OU. The ketotifen was discontinued with the intent to gauge any rebound in allergic symptoms at the follow-up visit. PFATs were still recommended at least 4 times a day OU. A follow-up was set for 1 month later.
3-month follow-up visit
At the 3-month follow-up appointment, the patient remained symptom-free. She had completed the prednisolone acetate 1.0% taper and experienced no issues after stopping the oral azithromycin. She denied any persistent itching after having stopped ketotifen. She noted continued compliance with the nightly application of bacitracin ointment, hypochlorous acid solution spray twice a day OU and PFATs 4 times a day OU. She confirmed minor lapses in the use of lid scrubs once a day in the morning OU.
On slit lamp examination, the lids were essentially clear OU, with a few areas of trace scurf. There was trace injection and a trace papillary response on both upper and lower lids OU. The corneal appearance was stable, with much of the residual scarring located below the visual axis. The neovascularization was quiescent with inactive (ghost) vessels inferiorly. The IOPs were 12 mmHg OD and 13 mmHg OS.
At this visit, the antibiotic ointment was discontinued in favor of bland artificial tear (AT) ointment for longer-term maintenance therapy due to the concern for antibiotic resistance and fungal overgrowth with long-term antibiotic use.41-43 She was allowed to discontinue the lid scrubs since she was no longer regularly using them and the lashes appeared mostly clear of bulky debris. Lid hygiene with warm compresses twice a day OU and hypochlorous acid solution spray twice a day OU was continued to maintain meibomian gland function and reduce lid bacterial bioburden. The PFATs were also continued 4 times a day OU with a plan to follow-up in 3 months unless symptoms of irritation, foreign body sensation, eye pain or light sensitivity occurred sooner.
6-month follow-up visit
At the 6-month follow-up appointment, the patient remained symptom-free and denied any flare-ups or recurrences. She noted compliance with nighttime AT ointment OU, hypochlorous acid solution spray twice a day OU and PFATs approximately twice a day OU. She had been using warm compresses at least once a day, but not always twice daily as directed. It was suggested she switch from the “bundle towel” warm compress to a microwaveable warm compress mask to improve adherence to therapy.
On slit lamp examination, the lid margins and lashes were essentially clear OU, with a few areas of trace scurf. There was trace injection and a trace papillary response on both upper and lower lids OU. The corneal appearance was stable with some reduction in density of the corneal opacity since the last visit (Figure 3). The neovascularization was still quiescent with inactive (ghost) vessels inferiorly. Some mild corneal thinning was noted in the area of scarring OS when visualizing with an optic section.
Distance-balanced subjective refraction at this visit showed results of OD -4.00 -0.75 x 160, 20/20, and OS -1.25 -2.75 x 160, 20/25. A topography image was obtained to examine the difference in astigmatism between the two eyes. The topography images can be seen in Figure 4 (OD) and Figure 5 (OS), with irregular corneal astigmatism evident OS. The IOPs were 14 mmHg OD and 13 mmHg OS.
The patient and parent were advised to continue regular lid hygiene with warm compresses twice a day OU, hypochlorous acid solution spray twice a day OU and PFATs OU as needed due to the chronic nature of the BKC. They were advised to call the office if the patient experienced any symptoms of recurrence such as irritation, foreign body sensation, eye pain or light sensitivity.
We discussed the option of contact lens fitting at this visit. The patient would be a candidate for a soft toric, gas permeable or specialty contact lens to achieve her best vision and reduce the anisometropia. She would require close monitoring due to the presence of the residual corneal neovascularization and scarring. She declined a contact lens fitting at this visit but would consider it in the future.
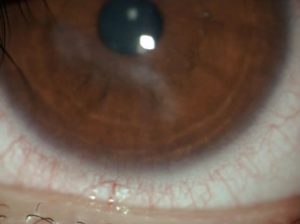 Figure 3. Clinical appearance after 6 months of follow-up. Click to enlarge |
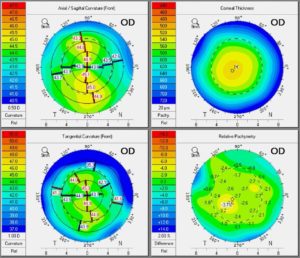 Figure 4. Topography maps OD showing normal cornea with regular astigmatism. |
 Figure 5. Topography maps OS showing corneal irregularity from residual scarring. |
Education Guidelines
Key concepts
- Identification of hallmark signs and symptoms of PKC and pediatric BKC
- Differentiating PKC from infectious or other etiologies
- Proper prescription of oral antibiotics in pediatric patients including correct dosage calculation
- Using patient education to assist in adherence to the prescribed treatment plan
- Recognizing the potential for vision loss/amblyopia that exists with PKC and BKC
- Proactively recommending contact lenses upon resolution and when indicated
Learning objectives
- Identify and list the signs and symptoms of PKC
- Describe the natural history/course of PKC
- Describe populations at a higher risk for PKC, including those with co-existing pediatric BKC
- Differentiate PKC from other conditions that may present similarly
- Determine a management plan for a pediatric patient presenting with PKC and associated BKC
Discussion questions
1. Knowledge, concepts, facts and information required for critical review of the case
- Describe the signs and symptoms of PKC
- Describe the signs and symptoms of pediatric BKC
- Describe the etiology of PKC
- Describe several conditions that fall under the spectrum of disease that is pediatric BKC
- Discuss common ocular and systemic diseases to screen for in someone presenting with PKC
- Describe the potential side effects of treatment with topical steroids
- Describe the potential side effects of treatment with oral antibiotics
- Describe the potential side effects of treatment with topical antibiotics
- Describe common sequelae following the resolution of PKC
- Describe various options for vision correction following the resolution of PKC
2. Differential diagnosis
- What clinical observations and tests are used to diagnose PKC?
- Which clinical entities should be included in the differential diagnosis? How might they be ruled out?
3. Patient management
- What is the treatment of choice for PKC?
- What are common management options for pediatric BKC?
- Which antibiotics (oral or topical) are useful in these conditions?
- What patient education should be given to the guardian(s) of a patient with this condition?
- What are some concerns with the dosing of oral medications in pediatric patients?
- What tests can help determine potential contact lens options for patients with corneal neovascularization?
4. Critical-thinking concepts
- What if symptoms worsen or do not improve?
- What if IOP starts to rise during treatment?
- When fitting a patient with corneal neovascularization in contact lenses, what are the concerns and potential issues?
Teaching instructions and assessment methodology
This case could be incorporated into clinical or didactic discussions to achieve learning objectives. Students could also independently provide written answers to the learning objectives or discussion questions.
Clinical skills could be assessed through small group evaluation of anterior segment photography and corneal topography images, including a discussion of normal vs. abnormal findings and with a focus on describing the results as they are recorded in the electronic health record (e.g., in the written description of slit lamp findings or the interpretation/report for diagnostic testing). There could also be a discussion of the utility of photography to facilitate understanding of normal vs. abnormal findings and change over time, as well as their role in patient education. A series of presentation slides could show the case’s progression and resolution, with a discussion of which therapies to continue or discontinue based on findings.
The student knowledge base could be further assessed through student-directed presentations to compare/contrast and rule out potential differentials in the diagnosis. A role-play could occur with the preceptor playing the patient’s role and students performing a case history. The student(s) would need to ask appropriate history questions, request particular ocular examination results (provided by the preceptor) and discuss the differential diagnosis. The student(s) would then discuss the primary diagnosis, suggest an appropriate management plan, discuss the prognosis, provide patient education, and determine a follow-up schedule.
Discussion
BKC has an average age of onset of around 3 or 4, but patients may not present for treatment until they are school age or older.23,27,44 The condition is more common and severe among Asian and Middle Eastern children.45-46 These children are more likely to develop severe corneal complications, such as phlyctenular disease, scarring, visual loss and amblyopia.45,47 A particularly aggressive phenotype has also been identified in Caucasian children and adolescents that persists into early adult years and, in some cases, requires systemic immunosuppression.46 In a study of patients with chronic BKC, the majority (63%) of whom were Caucasian, corneal involvement was noted in 81% of subjects.48 In another study of BKC patients, the condition was most often bilateral (97% of cases) but potentially asymmetric (21% of cases) and commonly presented with eyelid inflammation (100% of cases).27,48
Symptoms of BKC include redness, tearing, foreign body sensation, itching, photophobia and mild discharge.23-24 Signs of BKC include conjunctival hyperemia, recurrent chalazia, meibomian gland inflammation and inspissation, lid margin telangiectasia and thickening, eyelash crusting, scales or collarettes, corneal punctate epithelial keratitis and marginal corneal infiltrates.19,23-24,44 By definition, those with BKC will have lid disease, so a thorough evaluation of the eyelashes, lid margin, conjunctiva and cornea are required for diagnosis.24 Reports indicate between 14% and 50% of pediatric patients with BKC present with classic phlyctenules while up to 62% have inferior corneal vascularization and infiltrates.11,23-24 Punctate keratitis is common, occurring in a reported 55-87% of patients with active disease.23,27
In PKC, pediatric populations are more affected than adults, and females tend to be more affected than males.5,12,26,28,49 Sex predilection has been linked to the expression of various steroid hormone receptors on the ocular surface.5,50 PKC is unilateral in 59% of patients and bilateral in 41% of patients.11 There is also a risk of recurrence. In one study of recurrent cases, the mean duration from the last episode of PKC to recurrence was 7.2 ± 3.6 months, and the mean duration from steroid withdrawal to recurrence was 2.4 ± 1.3 months.31 Recurrence rates may be higher in patients with systemic disease-associated PKC (such as those with tuberculosis or intestinal parasite).11,17 Meibomitis was present 100% of the time in a study of recurrent, steroid-dependent PKC patients, which highlights BKC as a spectrum of disorders that includes PKC.31
There are also potential geographic implications to PKC. The disease shows a warm-weather predilection and is more of a risk to those in crowded living conditions or at high endemic risk for tuberculosis.11,17,51 In North America, populations in Canada and Alaska have been followed for the tuberculosis-associated form of phlyctenular disease.13-14,38
Symptoms of PKC include varying degrees of blurry vision, tearing, irritation, foreign body sensation and itching. If a corneal phlyctenule is present, the symptoms may progress to pain, photophobia and blepharospasm.1,4,20 A secondary bacterial infection could also occur, causing mucopurulent discharge.1 Symptomatic morbidity in PKC can cause frequent absences from school, inability to keep the eyes open, and psychological disturbances.7
Signs of PKC can include single or multiple nodular lesions located near the limbus or on the cornea, with associated conjunctival hyperemia, corneal neovascularization or corneal scarring. In later stages, PKC may trigger or morph into Salzmann’s nodular degeneration.52 Vision change or loss is possible due to refractive shift, induced astigmatism, corneal scarring, corneal neovascularization or the development of amblyopia.24,53
The etiology of BKC (the entity which encompasses PKC) is multifactorial but includes chronic inflammation.24,54 The inflammatory response is exacerbated by increased bacterial bioburden on the eyelids and lashes. The predominant pathogens in pediatric BKC are Staphylococcus aureus, Staphylococcus epidermidis and Propionibacterium acnes.54 The goals of treatment for this spectrum of conditions include reducing bacterial bioburden on the eyelids, suppressing bacterial lipase activity on the eyelids, reducing antigens involved in the inflammatory process, and reducing the overall inflammatory response.54-56
Differential diagnosis
PKC is typically diagnosed based on history, symptoms and slit lamp examination. A baseline IOP assessment at the time of diagnosis is critical, as measurement should occur before starting any corticosteroid therapy. Many studies report that IOP rises 3-6 weeks after starting topical corticosteroids, though some elevation of pressure can be found in most patients as early as the first or second week. To identify any steroid response (using the same IOP measurement method as baseline), the IOP should be measured again 2 weeks later and then monthly for 2-3 months and again at 6 months.57 Clinicians should monitor refractory cases or patients who require even more prolonged therapy at least every 6 months. An IOP increase of >6 mmHg is generally considered clinically significant, though each case can vary.58 Because PKC more often affects a pediatric population, it should also be noted that the possibility of a steroid response is greater in children younger than 6.58-60 In refractory cases where complete withdrawal of the steroid is not possible, yet a steroid response occurs, clinicians may consider other strategies such as altering the dosing, concentration or formulation of steroid, adding a topical beta-blocker, or switching to a steroid-sparing agent.57
Several differential diagnoses are considered if a vascularized corneal lesion is noted. The appearance, onset, staining pattern and any associated findings will help clinicians arrive at the correct diagnosis. The differential diagnosis for phlyctenular eye disease includes: corneal infiltrate, ocular (acne) rosacea keratoconjunctivitis, nodular episcleritis, Salzmann’s nodular degeneration, trachoma pannus, luetic or viral interstitial keratitis, chlamydial conjunctivitis, inflamed pinguecula (pingueculitis), inflamed pterygium, conjunctival intraepithelial neoplasia, limbal vernal keratoconjunctivitis, allergic conjunctivitis, Demodex blepharitis, microbial keratitis, marginal ulcer, peripheral ulcerative keratitis, herpes simplex keratitis and ocular cicatricial pemphigoid.4,7,18,24,48,61-63 It should be noted that a clinical entity termed “meibomitis-related keratoconjunctivitis (MRKC)” has been suggested to present identically to the corneal presentation of phlyctenular keratitis and with treatment focusing on eradicating ocular surface inflammation.5,64-65 For the purposes of this case report, we will consider MRKC part of the varied spectrum of pediatric BKC as others have suggested.64
Management
The treatment of choice for any corneal or conjunctival phlyctenule is a topical corticosteroid. One older case series detailed topical steroid treatment ranging from 1-4 times daily for 1-4 years in a series of six patients with PKC.8 A more recent review of BKC noted that lower potency steroids, such as loteprednol and fluorometholone, are often sufficient and that dosing is quickly tapered from more frequent to less frequent, and then a slow taper occurs thereafter, to as little as once or twice a week to control the inflammation.24
Steroid treatment not only suppresses the ocular surface inflammation but also prevents corneal scarring, which could lead to visual compromise. It does carry a significant side effect profile, including the risk of elevated IOP, cataract formation, corneal melt and secondary bacterial or fungal infection.26,29,66-68 Some patients may also become “steroid-dependent,” meaning that when the anti-inflammatory therapy is withdrawn, the condition recurs, usually within a few months.29,31 Children are more vulnerable to an increase in IOP with topical steroid use than adults, so special attention to IOP is warranted in this population to prevent ocular hypertension.59 A secondary goal is to avoid corticosteroid-induced glaucoma, which can occur if the ocular hypertensive response is of sufficient magnitude for a prolonged duration and results in damage to the optic nerve.57 Because of the risks associated with topical steroids, practitioners tend to try to dose sparingly and at the lowest concentration possible.68 Some prefer “soft” steroids, such as loteprednol etabonate or fluorometholone, especially in cases of potential long-term therapy.66 A lack of insurance formulary coverage and resulting high out-of-pocket cost left this patient seeking an alternative to our initial consideration of a branded, ester-based “soft” topical loteprednol 0.5% suspension. Instead, generic prednisolone acetate 1.0% ophthalmic suspension was prescribed.
A multifaceted approach to treatment is recommended in pediatric BKC, with warm compresses seen as a mainstay of therapy (both for a flare-up and for long-term management) and recommended in 100% of cases.27 Additional considerations are removing any bulky debris (eyelid scurf) from anterior blepharitis with a specialized eyelid wipe or wash once or twice daily. This cleaning makes the lid more receptive to the addition of hypochlorous acid solution spray, a topical preparation with broad-spectrum antimicrobial activity, including to both Staphylococcus aureus and Staphylococcus epidermidis.69-70
Both topical and oral antibiotic therapies have been used to treat patients with significant levels of bacterial bioburden and inflammation related to BKC. Several oral antibiotics have been suggested for the treatment of BKC in children. Oral tetracyclines, such as doxycycline, are often avoided in pediatric patients under 8-9 years old due to the risk of tooth discoloration.26 The drugs should also be avoided in pregnant women or anyone allergic to tetracyclines. Some practitioners do still consider the use of tetracyclines for patients older than 10-12 years old, as the medication has been shown to be effective in the management of PKC and BKC in children in some studies.7,71 In an older case series of six children age 8-14 years with PKC who did not respond to topical corticosteroid and topical antibiotic therapy, Zaidman and Brown prescribed oral tetracycline 250 mg 2-3 times a day until 3 weeks after the patient was completely asymptomatic.8 They then decreased the dose by 250 mg per day, at 3- 4-week intervals, until the patient was using just 250 mg once a day. Overall, the approach of prescribing oral antibiotics, including tetracyclines, for BKC was not supported by a 2016 Cochrane Review.72
For younger children in whom tetracyclines are contraindicated, oral macrolide antibiotics may be used as an alternative and have been shown to have anti-inflammatory activity.73 It is unclear if the macrolide mechanism of action in BKC is a direct effect on lipid synthesis or an influence on the microflora.24 Both erythromycin and azithromycin concentrate readily within polymorphonuclear leukocytes and macrophages, inflammatory cells found in phlyctenules.1,74 Erythromycin is the most commonly prescribed macrolide antibiotic for pediatric BKC as it is generally well-tolerated and safe, with little incidence of allergic reaction.75 One potential side effect to monitor is dose-dependent gastrointestinal upset (e.g., diarrhea, abdominal pain, nausea or vomiting).74,76 Erythromycin in oral suspension or chewable tablet form can be prescribed for pediatric patients.44 Recommendations in the literature range from full strength (250 mg/kg/day) to one-quarter of full strength (50 mg/kg/day) dosing for BKC.24 The literature also shows total daily doses ranging from 60 to 500 mg/day, at once or divided over the day depending on patient weight and severity of disease.44 Most children are tapered off oral antibiotic therapy within 6 months of treatment, though treatment can last up to 12 months, depending on the disease and other ocular comorbidities.24,44,48,56
In cases where oral erythromycin cannot be used (such as allergy), case reports show the efficacy of oral azithromycin, a second-generation macrolide that is available in oral suspension form.26-27 Azithromycin has also been shown to achieve high tissue concentrations and is a more stable, more potent, better absorbed and more well-tolerated macrolide than erythromycin with fewer side effects such as gastrointestinal upset.74,76-77 A recent study also suggests that oral azithromycin may have enhanced efficacy in meibomian gland dysfunction due to the addition of lipid-promoting activity, which stimulates meibomian gland epithelial cell function. This is occurring in addition to the antibacterial and anti-inflammatory actions of azithromycin, functions that are shared by oral doxycycline as well as other oral tetracyclines prescribed for meibomian gland dysfunction.78 Clinicians should be aware that prescription of azithromycin may have long-term effects on the ocular microbiome and more research is needed to fully understand the implications for pediatric patients.79
The dosage of oral azithromycin for pediatric BKC, as described in the literature, is either 15 mg/kg/day or 5 mg/kg/day.8,80 Zaidman advocates once-daily dosing at 15 mg/kg/daily, stating it is better tolerated, more available and more convenient than erythromycin.24,81 Choi and Djalilian also published a small case series using oral azithromycin at a lesser dosage of 5 mg/kg/day, in conjunction with topical anti-inflammatory agents, with promising results.80 When using oral agents to treat BKC, it is generally recommended to treat for 3-6 months and then taper according to the clinical course.24 In this case, the patient’s weight was initially estimated (and later confirmed) by the parent as 100-115 lb (45-52 kg), so the prescribed dosage of azithromycin at a concentration of 250 mg/5 mL, based on an estimated 50 kg weight, was 250 mg/day (or 5 mL of suspension) by mouth. Several apps, such as Epocrates or Pedi QuikCalc, are available for helping to calculate the dosage for oral medication given a patient’s weight. Practitioners can also consult with the pediatrician or local pharmacist.
Considerations in the topical management of BKC include management of the inflammatory response and increased bacterial bioburden on the eyelids and lashes. The predominant pathogens in pediatric BKC are Staphylococcus aureus, Staphylococcus epidermidi and Propionibacterium acnes; therefore, a combination of lid scrubs, hypochlorous acid solution and topical antibiotic therapy can be employed to reduce the bacterial bioburden, and therefore lipase activity, near the lids and lashes. When choosing a topical antibiotic, one could start with broad gram-positive coverage with a particular aim at Staphylococcus and Streptococcus organisms, using bacitracin or erythromycin ointment at night to the lids and lashes for BKC.24,77 Another approach in anterior blepharitis suggests that first-line therapy is broad-spectrum coverage with polymyxin-B/trimethoprim ointment (or similar) and then topical azithromycin 1.0% if first-line treatment fails.82 Topical azithromycin has been shown to be effective in the adult population as an off-label medication to manage blepharitis, meibomian gland dysfunction and rosacea, and also in the pediatric population for blepharitis.26-67,82-91 In one study, topical azithromycin was used to treat steroid-dependent PKC in pediatric patients.29 The drug has been cleared for the treatment of bacterial conjunctivitis in pediatric patients age 1 and older; thus, use in pediatric BKC would be considered off-label.92 Fixed-combination products containing topical steroids and antibiotics may be an additional convenient and effective option for addressing lid margin disease.93
Prolonged use of antibiotic-containing topical preparations may result in antibiotic resistance and potential for overgrowth of non-susceptible organisms, particularly fungi.41-43 Methods of preventing resistance in chronic cases include adhering to appropriate dosing schedules, cycling or rotating the antibiotic being used, employing oral therapy, and discontinuing topical therapy when the clinical picture improves.94 Additionally, some ophthalmic antibiotic ointment package inserts recommend the removal of “scales and crusts” before the application of ointment; thus, the use of a lid scrub as a preliminary step in the eyelid cleaning regimen is advisable.41
Recalcitrant, recurrent or steroid-dependent cases of PKC and BKC may benefit from more aggressive topical therapy, such as topical cyclosporine (standard or compounded preparations) or topical tacrolimus.1,24,26,29-31 In cases where the patient cannot tolerate medical therapy, a sub-tenon steroid injection may be possible.95 There are also reports in the literature of extreme, recurrent cases requiring oral steroids or systemic immunosuppression therapy for resolution. One case used azathioprine and methotrexate to manage BKC, and another used infliximab and methotrexate to manage PKC.46,96 In recalcitrant cases, it is also prudent to ensure no systemic tuberculosis is present. In children, confirmation with blood testing (via Quantiferon Gold) is recommended over tuberculin skin testing.97 One should also thoroughly examine the ocular surface to rule out other etiologies or comorbidities with similar presentation to BKC/PKC and treat any underlying disease such as dry eye, allergic conjunctivitis and Demodex blepharitis.26,98
Compliance can be a challenge with pediatric patients.99 Anecdotal success with warm compress therapy has been noted when the activity is coupled with bathing, listening to music or watching television.24,27 Lid scrubs could be modified to be performed in conjunction with showering to improve ease of use and compliance.48
For the patient in this teaching case report, several signs of BKC were present, any one of which could confirm the diagnosis: lid scurf, meibomian gland inspissation, turbid meibomian gland expression, lid wiper epitheliopathy and line of Marx staining. The patient was instructed on the performance of warm compresses twice daily for at least 10 minutes using the “bundle towel” method to improve meibomian gland secretions.100 The “bundle towel” method involves heating several moistened, folded, washcloth-sized towels wrapped together into a circular bundle. The bundle’s concentric geometry helps the towels maintain heat inside while the outer towels are used consecutively. Warm compresses using the “bundle towel” method have been shown to increase inner eyelid surface temperatures above the required therapeutic level (>104 degrees Fahrenheit) if performed correctly.100 The patient was advised not to rub the eyes or massage the lids after performing warm compresses to prevent corneal warpage from occurring.101
The patient was instructed to begin eyelid hygiene twice daily using an individually wrapped, pre-moistened lid scrub pad to provide mechanical removal of scurf. To further manage the patient’s lid margin inflammation and anterior blepharitis, as well as to reduce the bacterial bioburden on the lid, bacitracin ophthalmic ointment was prescribed for use at night on the eyelids and eyelashes OU.24,67 Additionally, oral antibiotic azithromycin was prescribed at 250 mg/day by mouth, as her case was considered severe with concomitant lid disease and untreated symptoms for the past 6 months. She was also advised to begin over-the-counter hypochlorous acid 0.02% solution spray twice daily on the eyelids OU to reduce the bacterial bioburden, as we suspected compliance with the spray might be easier for a pediatric patient. The spray could be easily integrated into the consistent long-term eyelid hygiene regimen that is often necessary to manage BKC and prevent visual sequelae.
In this case, an oral antibiotic was also prescribed at the initial visit due to the presence of the sight-threatening phlyctenule and concomitant anterior and posterior meibomian gland dysfunction. The efficacy of oral azithromycin for meibomian gland dysfunction and blepharitis in adults has been established.67,102-106 In a case series of three pediatric patients with BKC treated with oral azithromycin (dosed 5 mg/kg, once daily) and topical anti-inflammatory agents, Choi and Djalilian note two patients had associated phlyctenules that resolved during treatment.80 In the case series, oral azithromycin was continued for approximately 2 months for each patient, but the authors discuss continuing various oral antibiotics for anywhere from 1-8 months for BKC. In an adult population, dosing is more commonly done as pulsed therapy (e.g., 500 mg per day over 3 consecutive days each week for 1 month; or 1 g per week for 3 consecutive weeks) or as a 5-day course with 500 mg on the first day and 250 mg each day for 4 consecutive days.103,105
In this case, the patient’s self-estimated weight was 100-115 lb If we assume the lower weight of the given range, it converts to 45 kg. At 5 mg/kg, that is a total daily dose of 225 mg. Assuming the higher weight of 115 lb, which converts to 52 kg, the total daily dose would be 260 mg. Given this range, we prescribed 250 mg/day based on the prescribing recommendations of Choi and Djalilian (5 mg/kg/day). The patient’s exact weight of 110 lb was provided at a follow-up appointment, and the dosage was confirmed as correct. Consultation with a pharmacist can also ensure that any dosage calculations are appropriate.
In cases where patients are using other medications, have comorbidities, have a history of sensitivity to the drug class in question, or are unsure of their weight or unable to obtain it, consultation with the primary care physician or pediatrician is highly recommended. Parents should inform their child’s physician of any oral antibiotic therapy prescribed by the optometrist for the management of ocular conditions. Optometrists can also consult with the physician and co-manage any changes in ocular therapy should the patient need concurrent oral antibiotic treatment for any other indication.
Topical cyclosporine of varying concentrations has also been suggested as a steroid-sparing alternative, adjunctive or sole treatment for phlyctenular disease4,24,26,29,80,83,96,107-108 The safety and efficacy of commercially available topical cyclosporine products to increase tear production in patients with keratoconjunctivitis sicca have not been established in patients younger than 16 for the 0.05% ophthalmic emulsion and younger than 18 for the 0.09% ophthalmic nanomicellar suspension; thus, use in pediatric patients would be considered off-label.109-110
For the patient described in this teaching case report, treatment for bilateral allergic conjunctivitis was also recommended due to the papillary reaction noted on each eye’s palpebral conjunctiva. Initially, it was difficult to determine the exact cause of the papillary response noted. Because inflammation and allergy symptoms often co-exist, it is no surprise that pediatric BKC is sometimes misdiagnosed as allergic conjunctivitis if it presents with chronic conjunctivitis, though a typical allergic response would not also have the concomitant lid disease.24,108 In some chronic cases of staphylococcal blepharitis, the presenting conjunctivitis has been attributed to a toxin reaction.111
Patient education for these conditions includes informing the parent or guardian that long-term maintenance of lid hygiene procedures and topical anti-inflammatory therapy (such as cyclosporine) may be required.24 The oral antibiotic therapy may continue for up to 12 months or more, depending on the patient’s comorbidities and clinical response.24,27,56,80,106 While PKC is not infectious, it may recur. It may also have visual sequelae that require correction or cannot be fully corrected.
Anterior segment photographs can be useful for educating caretakers on the course of their child’s disease, especially if communication between the optometrist and the caretaker (for example, if there is a language barrier) is limited. Images can demonstrate the severity of initial presentation, record progression and improvement over time, and document clinical resolution.
Contact lenses
The scarring and neovascularization often associated with conjunctival and corneal phlyctenules carry a risk for visual compromise and amblyopia in the pediatric population.24 The corneal appearance may cause a practitioner to shy away from fitting contact lenses, citing hypoxia concerns and worsening neovascularization. However, the etiology of neovascularization in phlyctenular disease is not hypoxic but rather inflammatory.112 Patients with a history of PKC may have medically necessary reasons to wear lenses, such as reduced vision with glasses, high ametropia (sphere or cylinder) or anisometropia. In addition to clear vision, contact lenses confer self-esteem benefits for children.113
New refraction and topography performed after PKC resolution will help determine whether amblyopia or irregular astigmatism limits a patient’s best-corrected vision. This patient’s anisometropia may have been present before the corneal condition or exacerbated by it. In either case, with reduced best-corrected acuity OS, a contact lens may assist with achieving an improved visual result after the resolution of the phlyctenule. The application of a diagnostic gas permeable contact lens will help determine the eye’s best-corrected vision if any corneal irregularity exists. Contact lens management should include close monitoring and use of materials highly permeable to oxygen to ensure corneal neovascularization does not progress, despite its known inflammatory etiology, and that new neovascularization does not develop.114 If the benefits are deemed to outweigh the risks, patients with a history of PKC and associated neovascularization can successfully wear soft, gas permeable or specialty lenses, each with appropriate monitoring.
Conclusion
Optometrists can effectively manage the signs and symptoms of PKC and pediatric BKC. This teaching case report is intended to educate eyecare providers and optometry students on the clinical course and management of phlyctenular eye disease, highlighting the importance of thorough clinical examination, immediate identification, appropriate treatment and follow-up care. Prompt recognition of the acute nature of phlyctenular disease can stem symptoms and progression, while recognizing the chronic nature of pediatric BKC can prevent recurrence. Appropriate management of these two associated conditions can help minimize debilitating symptoms and prevent profound vision loss in children. Because tetracyclines are contraindicated in pediatric patients, oral macrolides such as erythromycin and azithromycin are often used. However, a recent comprehensive Cochrane Review found insufficient evidence for the prescription of any oral antibiotics in pediatric BKC. Thus, management with aggressive lid hygiene efforts, topical steroids and topical antibiotics should be a priority. Practitioners should also consider the advantages of contact lens wear despite the presence of residual corneal neovascularization. When appropriately prescribed and managed after the resolution of phlyctenular disease, contact lenses can provide optimal visual function and overcome limitations posed by corneal scarring, irregularity, anisometropia or astigmatism.
References
- Mondino BJ. Inflammatory diseases of the peripheral cornea. Ophthalmology. 1988;95:463-472.
- Cosar CB, Sridhar MS. Clinical signs in cornea and ocular surface. Indian J Ophthalmol. 2018;66:202-206.
- Roy FH. Ocular Differential Diagnosis. JP Medical Ltd; 2012.
- Neiberg MN, Sowka J. Phlyctenular keratoconjunctivitis in a patient with Staphylococcal blepharitis and ocular rosacea. Optometry. 2008;79:133-137.
- Suzuki T, Mitsuishi Y, Sano Y, et al. Phlyctenular keratitis associated with meibomitis in young patients. Am J Ophthalmol. 2005;140:77-82.
- Duane TD, Tasman W, Jaeger EA. Duane’s Ophthalmology. Lippincott Williams & Wilkins; 2013.
- Culbertson WW, Huang AJ, Mandelbaum SH, et al. Effective treatment of phlyctenular keratoconjunctivitis with oral tetracycline. Ophthalmology. 1993;100:1358-1366.
- Zaidman GW, Brown SI. Orally administered tetracycline for phlyctenular keratoconjunctivitis. Am J Ophthalmol. 1981;92:178-182.
- Ostler HB. Corneal perforation in nontuberculous (Staphylococcal) phlyctenular keratoconjunctivitis. Am J Ophthalmol. 1975;79:446-448.
- Thygeson P. The etiology and treatment of phlyctenular keratoconjunctivitis. Am J Ophthalmol. 1951;34:1217-1236.
- Rohatgi J, Dhaliwal U. Phlyctenular eye disease: a reappraisal. Jpn J Ophthalmol. 2000;44:146-150.
- Beauchamp GR, Gillette TE, Friendly DS. Phlyctenular keratoconjunctivitis. J Pediatr Ophthalmol Strabismus. 1981;18:22-28.
- Fritz MH, Thygeson P, Durham DG. Phlyctenular keratoconjunctivitis among Alaskan natives. Am J Ophthalmol. 1951;34:177-184.
- Reed H, Hildes JA. Corneal scarring in Canadian Eskimos. Can Med Assoc J. 1959;81:364-366.
- Al-Hussaini MK, Khalifa R, Al-Ansary AT, et al. Phlyctenular eye disease in association with Hymenolepis nana in Egypt. Br J Ophthalmol. 1979;63:627-631.
- Hussein AA, Nasr ME. The role of parasitic infection in the aetiology of phlyctenular eye disease. J Egypt Soc Parasitol. 1991;21:865-868.
- Al-Amry MA, Al-Amri A, Khan AO. Resolution of childhood recurrent corneal phlyctenulosis following eradication of an intestinal parasite. J AAPOS. 2008;12:89-90.
- Chern KC, Zegans ME. Ophthalmology Review Manual. Lippincott Williams & Wilkins; 2000.
- Weisenthal R. Chapter 3: Clinical Approach to Ocular Surface Disease. In: Rapuano CJ, J Timothy Stout Md, McCannel CA, editors. 2020-2021 Basic and Clinical Science Course, Section 08: External Disease and Cornea eBook. American Academy of Ophthalmology; 2020.
- Mondal I, Anand N, Mishra P, et al. Phlyctenular keratoconjunctivitis and tuberculosis. Journal of Medical Science and Clinical Research. 2017;5:23692-23693.
- Singal A, Aggarwal P, Pandhi D, et al. Cutaneous tuberculosis and phlyctenular keratoconjunctivitis: a forgotten association. Indian J Dermatol Venereol Leprol. 2006;72:290-292.
- Ahn YS, Lee JA, Cho YK. Corneal perforation in phlyctenular keratitis. Journal of the Korean Ophthalmological Society. 2014;55:298-303.
- Farpour B, McClellan KA. Diagnosis and management of chronic blepharokeratoconjunctivitis in children. J Pediatr Ophthalmol Strabismus. 2001;38:207-212.
- Hammersmith KM. Blepharokeratoconjunctivitis in children. Curr Opin Ophthalmol. 2015;26:301-305.
- Shoaib KK. Severity of pediatric blepharokeratoconjunctivitis in Asian eyes. Am. J. Ophthalmol. ajo.com; 2012. p. 210; author reply 210-211.
- Traish A. Pediatric Ocular Surface Disease. In: Djalilian AR, editor. Ocular Surface Disease: A Case-Based Guide. Cham: Springer International Publishing; 2018. p. 233-254.
- Hammersmith KM, Cohen EJ, Blake TD, et al. Blepharokeratoconjunctivitis in children. Arch Ophthalmol. 2005;123:1667-1670.
- Teo L, Mehta JS, Htoon HM, et al. Severity of pediatric blepharokeratoconjunctivitis in Asian eyes. Am J Ophthalmol. 2012;153:564-570.e1.
- Doan S, Gabison E, Gatinel D, et al. Topical cyclosporine A in severe steroid-dependent childhood phlyctenular keratoconjunctivitis. Am J Ophthalmol. 2006;141:62-66.
- Kymionis GD, Kankariya VP, Kontadakis GA. Tacrolimus ointment 0.03% for treatment of refractory childhood phlyctenular keratoconjunctivitis. Cornea. 2012;31:950-952.
- Yoon CH, Kim MK, Oh JY. Topical tacrolimus 0.03% for maintenance therapy in steroid-dependent, recurrent phlyctenular keratoconjunctivitis. Cornea. 2018;37:168-171.
- Winn B, Ackerley RG, Brown CA, et al. The superiority of contact lenses in the correction of all anisometropia. Cont Lens Anterior Eye. 1986;9:95-100.
- Winn B, Ackerley RG, Brown CA, et al. Reduced aniseikonia in axial anisometropia with contact lens correction. Ophthalmic Physiol Opt. 1988;8:341-344.
- Evans BJW. Orthoptic indications for contact lens wear. Cont Lens Anterior Eye. 2006;29:175-181.
- Steiner AA. Corneal contact lenses – special value in severe anisometropia in children. Calif Med. 1960;92:348-349.
- Mets M, Price RL. Contact lenses in the management of myopic anisometropic amblyopia. Am J Ophthalmol. 1981;91:484-489.
- Roberts CJ, Adams GGW. Contact lenses in the management of high anisometropic amblyopia. Eye. 2002;16:577-579.
- Smith RE, Dippe DW, Miller SD. Phlyctenular keratoconjunctivitis: results of penetrating keratoplasty in Alaskan natives. Ophthalmic Surg. 1975;6:62-66.
- Huang C, O’Hara M, Mannis MJ. Primary pediatric keratoplasty: indications and outcomes. Cornea. 2009;28:1003-1008.
- Diestelhorst M, Kwon KA, Süverkrup R. Dose uniformity of ophthalmic suspensions. J Cataract Refract Surg. 1998;24:672-677.
- Bacitracin Ointment [Internet]. Bethesda, MD: U.S. National Library of Medicine, DAILYMED; [cited 2020 Nov 5]. Available from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=6ed2f2bd-9d2f-46af-a44c-95a02ca034de.
- Bertino JS Jr. Impact of antibiotic resistance in the management of ocular infections: the role of current and future antibiotics. Clin Ophthalmol. 2009;3:507.
- Bremond-Gignac D, Chiambaretta F, Milazzo S. A European perspective on topical ophthalmic antibiotics: current and evolving options. Ophthalmol Eye Dis. 2011;3:29-43.
- Meisler DM, Raizman MB, Traboulsi EI. Oral erythromycin treatment for childhood blepharokeratitis. J AAPOS. 2000;4:379-380.
- Viswalingam M, Rauz S, Morlet N, et al. Blepharokeratoconjunctivitis in children: diagnosis and treatment. Br J Ophthalmol. 2005;89:400-403.
- Hamada S, Khan I, Denniston AK, et al. Childhood blepharokeratoconjunctivitis: characterising a severe phenotype in white adolescents. Br J Ophthalmol. 2012;96:949-955.
- Mehta JS, Sagoo MS, Tuft SJ. Subconjunctival crystals in paediatric blepharokeratoconjunctivitis. Acta Ophthalmol Scand. 2006;84:557-558.
- Jones SM, Weinstein JM, Cumberland P, et al. Visual outcome and corneal changes in children with chronic blepharokeratoconjunctivitis. Ophthalmology. 2007;114:2271-2280.
- Jo DH, Kim MK, Wee WR, et al. Analysis of clinical characteristics in phlyctenular keratoconjunctivitis at a tertiary center. Journal of the Korean Ophthalmological Society. 2011;52:7-13.
- Suzuki T, Kinoshita Y, Tachibana M, et al. Expression of sex steroid hormone receptors in human cornea. Curr Eye Res. 2001;22:28-33.
- Rhee ST. Seasonal development of eye diseases. Journal of the Korean Ophthalmological Society. 1973;14:355-362.
- Tabarra KF. Phlyctenulosis. In: Roy FH, Fraunfelder FW, Fraunfelder FT, editors. Roy and Fraunfelder’s Current Ocular Therapy. Elsevier Health Sciences; 2008. p. 388-389.
- Hunter K, Affeldt J, Schneider K. Hyperopic refractive shift secondary to phlyctenular keratoconjunctivitis. Invest Ophthalmol Vis Sci. 2004;45:2863-2863.
- Medsinge A, Nischal KK. Managing blepharokeratoconjunctivitis in children: a review. Expert Rev Ophthalmol. 2013;8:485-499.
- Daniel MC, O’Gallagher M, Hingorani M, et al. Challenges in the management of pediatric blepharokeratoconjunctivitis/ocular rosacea. Expert Rev Ophthalmol. 2016;11:299-309.
- Cetinkaya A, Akova YA. Pediatric ocular acne rosacea: long-term treatment with systemic antibiotics. Am J Ophthalmol. 2006;142:816-821.
- Kersey JP, Broadway DC. Corticosteroid-induced glaucoma: a review of the literature. Eye. 2006;20:407-416.
- Razeghinejad MR, Katz LJ. Steroid-induced iatrogenic glaucoma. Ophthalmic Res. 2012;47:66-80.
- Kwok AK, Lam DS, Ng JS, et al. Ocular-hypertensive response to topical steroids in children. Ophthalmology. 1997;104:2112-2116.
- Juberias JR, Calonge M, Montero J, et al. Phlyctenular keratoconjunctivitis a potentially blinding disorder. Ocul Immunol Inflamm. 1996;4:119-123.
- Liu S, Pavan-Langston D, Colby KA. Pediatric herpes simplex of the anterior segment: characteristics, treatment, and outcomes. Ophthalmology. 2012;119:2003-2008.
- Blaustein BH, Gurwood AS. Recurrent phlyctenular keratoconjunctivitis: a forme fruste manifestation of rosacea. Optometry. 2001;72:179-184.
- El-Asrar AMA, Tabbara KF. Tetracycline treatment of phlyctenulosis. Ophthalmology. 1994;101:1161.
- Suzuki T, Kinoshita S. Meibomitis-related keratoconjunctivitis in childhood and adolescence. Am. J. Ophthalmol. ajo.com; 2007. p. 160-161; author reply 161.
- Suzuki T. Meibomitis-related keratoconjunctivitis: implications and clinical significance of meibomian gland inflammation. Cornea. 2012;31 Suppl 1:S41-S44.
- Sheppard JD, Comstock TL, Cavet ME. Impact of the topical ophthalmic corticosteroid loteprednol etabonate on intraocular pressure. Adv Ther. 2016;33:532-552.
- Geerling G, Tauber J, Baudouin C, et al. The international workshop on meibomian gland dysfunction: report of the subcommittee on management and treatment of meibomian gland dysfunction. Invest Ophthalmol Vis Sci. 2011;52:2050-2064.
- Nazir SA, Murphy S, Siatkowski RM, et al. Ocular rosacea in childhood. Am J Ophthalmol. 2004;137:138-144.
- Wang L, Bassiri M, Najafi R, et al. Hypochlorous acid as a potential wound care agent: part I. Stabilized hypochlorous acid: a component of the inorganic armamentarium of innate immunity. J Burns Wounds. 2007;6:e5.
- Hauser W. Protect patients’ eyes by encouraging a three-step ocular wellness regimen. Optometry Times. 2020 Oct;12:14.
- Koo G-H, Lee J-S. Treatment of phlyctenular keratoconjunctivitis with oral tetracycline in recurrent phlyctenulosis. Journal of The Korean Ophthalmological Society. 2003;44:2675-2679.
- O’Gallagher M, Banteka M, Bunce C, et al. Systemic treatment for blepharokeratoconjunctivitis in children. Cochrane Database Syst Rev. 2016;CD011750.
- Ianaro A, Ialenti A, Maffia P, et al. Anti-inflammatory activity of macrolide antibiotics. J Pharmacol Exp Ther. 2000;292:156-163.
- Whitman MS, Tunkel AR. Azithromycin and clarithromycin: overview and comparison with erythromycin. Infect Control Hosp Epidemiol. 1992;13:357-368.
- Shah F. Erythromycin. Pediatr Rev. 1998;19:140-141.
- Klein JO. History of macrolide use in pediatrics. Pediatr Infect Dis J. 1997;16:427-431.
- Duncan K, Jeng BH. Medical management of blepharitis. Curr Opin Ophthalmol. 2015;26:289-294.
- Liu Y, Kam WR, Ding J, et al. Can tetracycline antibiotics duplicate the ability of azithromycin to stimulate human meibomian gland epithelial cell differentiation? Cornea. 2015;34:342-346.
- Doan T, Hinterwirth A, Worden L, et al. Post-antibiotic ocular surface microbiome in children: a cluster-randomized trial. Ophthalmology. 2020;127:1127-1130.
- Choi DS, Djalilian A. Oral azithromycin combined with topical anti-inflammatory agents in the treatment of blepharokeratoconjunctivitis in children. J AAPOS. 2013;17:112-113.
- Zaidman GW. The pediatric corneal infiltrate. Curr Opin Ophthalmol. 2011;22:261-266.
- Veldman P, Colby K. Current evidence for topical azithromycin 1% ophthalmic solution in the treatment of blepharitis and blepharitis-associated ocular dryness. Int Ophthalmol Clin. 2011;51:43-52.
- Doan S, Elbim C, Gabison E, et al. Efficacy of topical cyclosporine A in phlyctenular keratoconjunctivitis associated with severe corneal inflammation. Invest Ophthalmol Vis Sci. 2003;44:681-681.
- Opitz DL, Tyler KF. Efficacy of azithromycin 1% ophthalmic solution for treatment of ocular surface disease from posterior blepharitis. Clin Exp Optom. 2011;94:200-206.
- Luchs J. Azithromycin in DuraSite for the treatment of blepharitis. Clin Ophthalmol. 2010;4:681-688.
- Luchs J. Efficacy of topical azithromycin ophthalmic solution 1% in the treatment of posterior blepharitis. Adv Ther. 2008;25:858-870.
- Mantelli F, Di Zazzo A, Sacchetti M, et al. Topical azithromycin as a novel treatment for ocular rosacea. Ocul Immunol Inflamm. 2013;21:371-377.
- Opitz DL, Harthan JS, Fromstein SR, et al. Diagnosis and management of meibomian gland dysfunction: optometrists’ perspective. Clin Optom. 2015;7:59-69.
- Opitz DL, Harthan JS. Review of azithromycin ophthalmic 1% solution (AzaSite®) for the treatment of ocular infections. Ophthalmol Eye Dis. 2012;4:1-14.
- Zandian M, Rahimian N, Soheilifar S. Comparison of therapeutic effects of topical azithromycin solution and systemic doxycycline on posterior blepharitis. Int J Ophthalmol. 2016;9:1016-1019.
- Doan S, Gabison E, Chiambaretta F, et al. Efficacy of azithromycin 1.5% eye drops in childhood ocular rosacea with phlyctenular blepharokeratoconjunctivitis. J Ophthalmic Inflamm Infect. 2013;3:38.
- AzaSite Package Insert, 2012 [Internet]. Whitehouse Station, NJ: Inspire Pharmaceuticals, Inc.; c2011. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/050810s014lbl.pdf.
- Pflugfelder SC, Karpecki PM, Perez VL. Treatment of blepharitis: recent clinical trials. Ocul Surf. 2014;12:273-284.
- Kowalski RP, Dhaliwal DK. Ocular bacterial infections: current and future treatment options. Expert Rev Anti Infect Ther. 2005;3:131-139.
- Bondalapati S, Cabrera MT. Sub-tenon triamcinolone acetonide injections for topical medication intolerance in chronic blepharokeratoconjunctivitis. Cornea. 2014;33:999-1001.
- Valério Sequeira Valadares J, Bastos-Carvalho A, Pedroso Franco JM, et al. Phlyctenular keratoconjunctivitis – an atypically severe case treated with systemic biologic immunosuppressive therapy. GMS Ophthalmol Cases. 2014;4:Doc02.
- Kimberlin DW, Brady MT, Jackson M, Long SS. Red Book: 2018-2021 report of the committee on infectious diseases. American Academy of Pediatrics; 2018.
- Liang L, Safran S, Gao Y, et al. Ocular demodicosis as a potential cause of pediatric blepharoconjunctivitis. Cornea. 2010;29:1386-1391.
- Winnick S, Lucas DO, Hartman AL, et al. How do you improve compliance? Pediatrics. 2005;115:e718-e724.
- Murakami DK, Blackie CA, Korb DR. All warm compresses are not equally efficacious. Optom Vis Sci. 2015;92:e327-e333.
- McMonnies CW, Korb DR, Blackie CA. The role of heat in rubbing and massage-related corneal deformation. Cont Lens Anterior Eye. 2012;35:148-154.
- Kashkouli MB, Fazel AJ, Kiavash V, et al. Oral azithromycin versus doxycycline in meibomian gland dysfunction: a randomised double-masked open-label clinical trial. Br J Ophthalmol. 2015;99:199-204.
- Greene JB, Jeng BH, Fintelmann RE, et al. Oral azithromycin for the treatment of meibomitis. JAMA Ophthalmol. 2014;132:121-122.
- Yildiz E, Yenerel NM, Turan-Yardimci A, et al. Comparison of the clinical efficacy of topical and systemic azithromycin treatment for posterior blepharitis. J Ocul Pharmacol Ther. 2018;34:365-372.
- Igami TZ, Holzchuh R, Osaki TH, et al. Oral azithromycin for treatment of posterior blepharitis. Cornea. 2011;30:1145-1149.
- Al-Hity A, Lockington D. Oral azithromycin as the systemic treatment of choice in the treatment of meibomian gland disease. Clin Experiment Ophthalmol. 2016;44:199-201.
- Ismail A-S, Taharin R, Embong Z. Topical cyclosporin as an alternative treatment for vision threatening blepharokeratoconjunctivitis: a case report. Int Med Case Rep J. 2012;5:33-37.
- Rousta ST. Pediatric blepharokeratoconjunctivitis: is there a “right” treatment? Curr Opin Ophthalmol. 2017;28:449-453.
- Restasis Package Insert, 2012 [Internet]. Irvine, CA: Allergan, Inc.; c2012. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/050790s020lbl.pdf.
- Cequa Package Insert, 2019 [Internet]. Cranbury, NJ: Sun Pharma Global FZE; c2019. Available from: https://cequapro.com/pdf/CequaPI.pdf.
- Tetz MR, Klein U, Völcker HE. Staphylococcus-associated blepharokeratoconjunctivitis. Clinical findings, pathogenesis and therapy. Ophthalmologe. 1997;94:186-190.
- Roshandel D, Eslani M, Baradaran-Rafii A, et al. Current and emerging therapies for corneal neovascularization. Ocul Surf. 2018;16:398-414.
- Walline JJ, Jones LA, Sinnott L, et al. Randomized trial of the effect of contact lens wear on self-perception in children. Optom Vis Sci. 2009;86:222-232.
- Barnett M, Johns LK. Contemporary Scleral Lenses: Theory and Application. Bentham Science Publishers; 2017.



