PEER REVIEWED
Systemic and Ocular Associations of Angioid Streaks
Beata I. Lewandowska, OD, MS, and Marlon J Demeritt, OD, MBA, FAAO
Abstract
Angioid streaks are bilateral, linear, crack-like dehiscence of a calcified and brittle Bruch’s membrane that develop secondary to mechanical stress. Pseudoxanthoma elasticum is the systemic disease most frequently associated with angioid streaks. Choroidal neovascularization is a common vision-threatening complication encountered in eyes with angioid streaks. It is important for optometrists to understand the systemic and ocular associations of angioid streaks because some are vision- or life-threatening. The patient described in this case report had an established personal and family history of angioid streaks without systemic association, and he developed an extrafoveal choroidal neovascular membrane in the left eye. All aspects of patient care are presented in detail. In addition, color fundus imaging and optical coherence tomography findings are thoroughly discussed and integrated into the report.
Key Words: angioid streaks, choroidal neovascular membrane
Background
Angioid streaks (AS) are bilateral, linear, crack-like dehiscence of a mineralized, brittle Bruch’s membrane (BrM) that develop secondary to mechanical stress. The age of onset is variable. White people are most commonly affected, and there is no sexual predilection. While AS are associated with multiple systemic diseases, pseudoxanthoma elasticum (PXE) is the most frequently reported. Choroidal neovascular membrane (CNVM) is a common vision-threatening complication encountered in eyes with angioid streaks.
This case report focuses on the proper approach to diagnosing and managing patients with AS. A thorough review of the clinical aspects of AS is presented to facilitate the understanding of the course of action taken to manage this patient. In addition, concepts covering the retinal anatomy and physiology are integrated into the discussion to cultivate critical thinking and reinforce clinical competence in the optometric care of eyes with AS. This case can be discussed with optometry students who have not had any patient care experience. At most U.S. optometry programs, it would be appropriate for second-year students as well as third-year students.
Student Discussion Guide
Case description
A 55-year-old African American male presented to our clinic complaining of slowly worsening blurry vision without glasses at near in the left eye more so than the right eye. The patient stated the blurriness began approximately 2 month prior. His ocular history included mild myopia with regular astigmatism and presbyopia in both eyes, pinguecula in both eyes and angioid streaks in both eyes. The angioid streaks were noted during previous annual eye examinations at our clinic and documented with dilated fundus photography 4 years prior to the current visit (Figures 1A and 1B).
 Figure 1A. Dilated retinal fundus photograph of right eye obtained 4 years prior to the current exam showing peripapillary pigment mottling, angioid streaks and crystalline bodies. Click to enlarge |
 Figure 1B. Dilated retinal fundus photograph of left eye obtained 4 years prior to the current exam showing peripapillary pigment mottling, angioid streaks and crystalline bodies. Click to enlarge |
The patient’s medical history was unremarkable. He denied suffering from allergies to any medications and reported taking ibuprofen 200 mg as needed. Family history included a visually disabled brother with a history of angioid streaks and cerebral stroke. In addition, both parents suffered from hypertension. The patient also reported a maternal history of colon cancer and a history of breast cancer in his sister. The patient denied current or past tobacco use and alcohol consumption. He reported consuming approximately four cups of tea or coffee per day and exercising (i.e., running) three to four times per week. The patient’s mood and affect were normal. He was oriented to person, place and time. Blood pressure, measured manually at 2:16 p.m. on the right arm of the patient while he was sitting, was 145/85 mmHg.
Best-corrected visual acuities were 20/20 in each eye at both distance and near. The refraction was stable from the previous exam 1 year prior. Pupils were equal, round and reactive to light without a relative afferent pupillary defect in either eye. Confrontations visual fields were full to finger counting in four quadrants in both eyes. Ocular motility was full and smooth in both eyes. Slit lamp examination of the anterior segment revealed normal adnexa, a few capped meibomian glands with debris on eyelashes, and quiet lid margins in both eyes. Pinguecula were noted nasally and temporally in both eyes. The conjunctiva was quiet in both eyes. There was a small (<1 mm) dense white subepithelial opacity in the inferior nasal peripheral cornea without surrounding active infiltration in the right eye. Trace endothelial pigment was present in both eyes. Iris was flat, intact and brown in both eyes. The anterior chamber was deep and quiet, and the angles were open in both eyes. Trace nuclear sclerotic changes were noted in the lenses of both eyes. Intraocular pressure was measured by Goldmann applanation tonometry and found to be 11 mmHg OD and 10 mmHg OS at 2:43 p.m.
Dilated fundus examination revealed clear vitreous in both eyes. The optic disks were round and pink with distinct borders. The cup-to-disk ratio was observed to be 0.30/0.30 in both eyes. Peripapillary pigment mottling was noted to surround both optic nerves with progressively thinning, radially oriented AS in both eyes. Pigmentary changes were noted in both maculae. Approximately 1 disk diameter inferior to the fovea of the left eye, a semilunar elevation with internal hemorrhaging was noted. Crystalline bodies were present inferior to the optic nerves and along the inferior arcades in both eyes. The retinal periphery was intact 360 degrees OU with inferior-temporal spots of retinal pigment epithelium (RPE) hyperplasia in both eyes.
To document the retinal exam findings, dilated fundus photography (Figures 2A and 2B), optical coherence tomography (OCT) and OCT angiography (OCTA) were performed. The new crescent-shaped lesion, most likely a CNVM, a common complication in eyes with AS, was seen in the left eye (Figure 2B). The suspicion of CNVM was further confirmed by OCT (Figure 3) and OCTA (Figures 4A and 4B). While it was difficult to appreciate the changes associated with the AS, Figure 3 depicted a hyper-reflective lesion present in the subretinal space of the left eye with adjacent serous detachment and overlying retinal edema most consistent with neovascularization. An OCTA (Zeiss Cirrus) retina slab from the left eye showed neovascularization in the outer retina (Figure 4A), and a lacy vascular network in the avascular complex, most consistent with type 2 or classic CNVM, was seen on an OCTA outer retina slab from the left eye (Figure 4B).
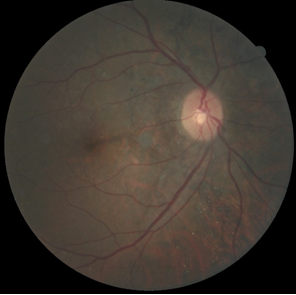 Figure 2A. Dilated retinal fundus photograph of the right eye showing peripapillary pigment mottling and angioid streaks. |
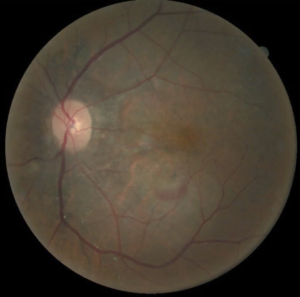 Figure 2B. Dilated retinal fundus photograph of the left eye showing peripapillary pigment mottling, angioid streaks and subretinal hemorrhage. Click to enlarge |
 Figure 3. Optical coherence tomography of the left eye showing a hyper-reflective lesion above the retinal pigment epithelium with adjacent serous separation and Bruch’s membrane undulations. Click to enlarge |
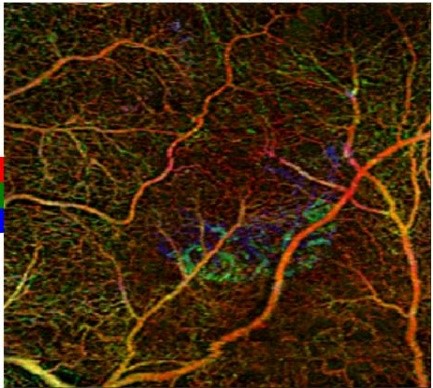 Figure 4A. Retina slab from optical coherence tomography angiography (Zeiss Cirrus) scans in the left eye depicting neovascularization in the outer retina. Click to enlarge |
 Figure 4B. Outer retina slab from optical coherence tomography angiography (Zeiss Cirrus) scans in the left eye depicting a lacy vascular network in the avascular complex consistent with type 2 or classic CNVM. Click to enlarge |
A report was sent to the patient’s primary care physician with a request to rule out the most common systemic associations of angioid streaks: PXE, sickle cell disease, Ehlers-Danlos syndromes (EDS) and Paget’s disease (PD). The patient was promptly referred for further evaluation and management to the retinal specialist who had treated his brother’s eyes in the past. Three months after the referral, the patient reported receiving injections in the left eye, most likely one of the available anti-vascular endothelial growth factor (VEGF) medications. He reported stable vision in both eyes. He also stated that no evidence of the systemic diseases commonly associated with AS was found.
Educator’s Guide
Key concepts
- Understanding the epidemiology and etiology of AS
- Understanding the pathogenesis of AS
- Understanding clinical manifestations of AS
- Understanding differential diagnoses for AS
- Understanding systemic diseases associated with AS
- Understanding the most common ocular complication associated with the presence of AS
- Understanding treatment options for the most common ocular complication associated with the presence of AS
- The importance of patient education about this condition
Learning objectives
At the conclusion of this case discussion, optometry students should be able to:
- Recognize and describe the ocular signs associated with AS
- Describe various common systemic conditions associated with AS
- Have a basic understanding of the pathogenesis of CNVM formation in the presence of AS
- Describe the available diagnostic tools and their application in the diagnosis of AS and CNVM
- Possess general knowledge about patient education and the management options for the common ocular complications associated with the presence of AS
Discussion questions
1. Knowledge, concepts, facts, information required for critical review of the case
a. Describe the etiology of AS
b. Describe the epidemiology of AS
c. Describe the pathogenesis of AS
d. Describe the diagnostic tools available to clinicians that aid in the diagnosis, evaluation and monitoring of AS
2. Differential diagnosis
a. What are the characteristics of the differential diagnoses of angioid streaks?
b. Describe the common systemic conditions associated with AS
c. Describe the pathophysiology of CNVM in the presence of AS
d. Describe the diagnostic tools available to clinicians that can aid in the diagnosis, evaluation and monitoring of CNVM
3. Patient management and the role of the optometrist
a. What treatment options are available to this patient?
b. What is appropriate timing for specialist consultation?
c. How would you counsel a patient with AS?
d. How would you counsel a patient with AS and choroidal neovacularization?
4. Higher-order critical-thinking concepts
a. Describe the peripapillary and macular findings observed on the retinal fundus photographs of this patient. What is different about the photographs taken at the current visit compared with previous photographs?
b. Analyze and interpret the OCT and OCTA findings. How do these findings relate to the clinical findings and the final diagnosis?
c. Identify and compare the risks and benefits of the different treatments available to patients with CNVM in the presence of angioid streaks. If you were the patient, which treatment would you choose and why?
d. What is the prognosis for the left eye? Predict the consequences of delayed treatment of the CNVM. Describe how these consequences may affect the patient’s quality of life.
Teaching instructions and assessment methodology
The purpose of this teaching case report is to lead a discussion in a classroom or clinical setting on the topic of AS, including their systemic and ocular associations. Second- or third-year optometry students should be presented with the case in a stepwise fashion so they can analyze the clinical data, extract the critical aspects from the case history and exam findings, and arrive at differential diagnoses. They can then be presented with the results of OCT and OCTA to finalize their diagnosis and create and present a management plan for the patient. Formative assessment can take place during the activity.
Discussion
Angioid streaks are bilateral, linear, crack-like dehiscence of a calcified and brittle BrM. They typically emerge from the optic disk as dark reddish-brown bands of variable width. CNVM is a common vision-threatening complication encountered in patients with AS.
Epidemiology and etiology of angioid streaks
First reported by Doyne in 18891 in a patient with retinal hemorrhages secondary to trauma, AS were named by Knapp in 18922 and determined to be a dysfunction of BrM by Kopler in 1917.3-4 If the overlying retinal layers remain unaffected, AS have no adverse effects on visual function.3,4,5 However, in some cases AS may lead to impaired visual function through degeneration within the retinal pigment epithelium (RPE) and accompanying photoreceptor defects, which result in metamorphopsia and scotoma.3-5
It is important to remember that complications associated with AS often occur in patients of working age and therefore can significantly impact quality of life. AS rarely occur in patients younger than 10 years.3,5 AS are usually found in patients age 20-50.3,5
Pathogenesis
Angioid streaks are a result of cracks in an abnormally thickened and calcified BrM. They may result from a combination of diffuse elastic degeneration of BrM, iron deposition in elastic fibers from hemolysis with secondary mineralization, and impairment of nutrition supply secondary to sickling, stasis and small vessel occlusion.3 There is a loss of migration of the pigment granules in the RPE. When full-thickness breaks occur, there is disruption of the choriocapillaris, atrophy of the RPE and loss of the overlying photoreceptor cells.
AS predispose the tissue to localized rupture that may occur spontaneously or secondary to blunt trauma. AS can cause subretinal hemorrhages from choroidal fibrovascular ingrowth or CNVM.3-5 In the former case, the hemorrhage can resolve spontaneously. In the latter case, metamorphopsia and/or scotoma may be experienced secondary to the presence of hemorrhages and edema, leading to significant loss of vision from development of a fibrotic scar.3-5
Clinical manifestations
On fundus biomicroscopy, AS appear as reddish-brownish, bilateral, jagged, progressively narrowing lines deep in the retinal layers. These lines appear to radiate from an annular area of peripapillary pigment mottling surrounding the optic nerve (Figures 1A, 1B, 2A, 2B). Visual acuity is not affected even when the AS cross into the foveal area unless there is degeneration within the RPE.
Differential diagnosis
Choroidal rupture

Figure 5. Fundus photograph representing how the site of a previous choroidal rupture caused by ocular trauma would appear. Click to enlarge
Choroidal rupture is a break in the choroid, the BrM and the RPE with an intact overlying neurosensory retina. It is often seen in eyes that suffered trauma. It often presents as a white crescent-shaped streak concentric to the optic disk (Figure 5).
During a closed globe injury, the eyeball first undergoes a mechanical compression followed by a rapid hyperextension of the tissues. The sclera possesses sufficient tensile strength to resist the compression, and the elastic retina stretches. However, the BrM does not have sufficient tensile strength or elasticity and it breaks. The injured choriocapillaris and choroidal vessels bleed into the sub-RPE and/or subretinal space. Initially, the deep hemorrhage may hide the choroidal rupture. As the blood clears over time, the subretinal streak appears.
CNVM that may cause delayed vision loss develops in 5% to 10% of eyes with a choroidal rupture.6 Over time, the CNVM involutes but tends to recur.
Fluorescein angiography (FA) typically shows hypofluorescence of the choroidal rupture in early frames and staining in late frames due to fluorescein leakage from adjacent choriocapillaris. Fundus autofluorescence (FAF) typically shows hypoautofluorescence of the rupture site where RPE is missing, with hyperautofluorescence at the edge of the choroidal rupture. OCT shows a loss of the continuity of RPE at the site of rupture with thinning of underlying inner choroid.
Lacquer cracks
The multiple yellowish-white irregular, horizontally oriented linear or stellate lesions known as lacquer cracks may be observed in eyes with high myopia (defined as a spherical equivalent exceeding -6 diopters and/or axial length longer than 26.5 mm) or pathological/degenerative myopia (defined as high axial myopia with characteristic pathological changes at the posterior pole). These eyes often have tilted disks with scleral crescents, posterior pole staphylomas, pigmentary changes and chorioretinal atrophy. Lacquer cracks are believed to be healed mechanical breaks of the RPE, BrM and choriocapillaris complex. The incidence of pathological myopia ranges from 5% to 11%, and CNV may develop in 57% of eyes with lacquer cracks.7-8
FAs of lacquer cracks show linear hyperfluorescence without leakage. On FAF, they appear hypoautofluorescent. On OCT, lacquer cracks appear as interruptions of the RPE and increased hyper-transmission into the deeper tissue beyond the RPE.
Diagnostic tools for AS
FA, indocyanine green angiography (ICGA), FAF, OCT and infrared and red-free retinography are useful in diagnosing, evaluating and monitoring AS.
Angiography
In 1961, Novotny and Alvis described a method for the study of retinal blood flow dynamics, which used intravascular fluorescein and retinal fundus photography equipped with excitation and barrier filters that captured the luminescence of the fluorescein as it passed through the retinal vasculature.9 Hypofluorescence can occur secondary to a blockage or a vascular filling defect. Hyperfluorescence can occur secondary to leakage, fluorescein staining, pooling, autofluorescence or a transmission defect. Choroidal circulation can be better visualized using ICGA.
Historically, the diagnosis of AS was confirmed with FA, with the streaks appearing as hyperfluorescent.
Fundus autofluorescence
FAF can also aid in visualization of AS. This imaging modality relies on the light emitted from lipofuscin in the RPE cells. As such, it represents metabolic activity of the RPE. The typical pattern in the attenuated, atrophied or absent RPE in AS is that of a hypoautofluorescent fissure. Partial repopulation of the AS with RPE cells can be visualized as punctate areas of normal autofluorescence within the hypoautofluorescent streak.
Optical coherence tomography
OCT is a valuable tool for non-invasive imaging of the retinal layers at baseline as well as for comparison on subsequent visits. Calcium deposits can be observed as localized areas of hyper-reflectivity on OCT. Spectral-domain OCT can help in visualization of the defects of the BrM in eyes with AS.
Infrared imaging
AS may pass unnoticed on color fundus photographs. Infrared imaging enhances the visualization of AS. They appear as brick-red colored and well-demarcated dark fissures against a lighter background.
Systemic associations

Table 1. Click to enlarge
AS can be idiopathic, but they can also be associated with several systemic conditions recalled through a popular mnemonic PEPSI: PXE, EDS, PD, sickle cell disease, with I standing for idiopathic. While there are reports of multiple other associations with systemic conditions (Table 1), those details are beyond the scope of this introductory article to AS.
Pseudoxanthoma elasticum (P)
PXE, also known as Grönblad-Strandberg syndrome, is an autosomal recessive mutation in the ABCC6 (ATP binding cassette subtype C number 6) gene on the short arm of chromosome 16 (16p13.1) resulting in fragmentation and calcification of elastic fibers found in the skin, retina and cardiovascular tissue.11 Approximately 80% of patients with PXE have AS.11 PXE is more common in females than in males.11 To confirm the diagnosis of PXE, a skin biopsy is necessary. If PXE is present, the skin biopsy will reveal mid-dermal clumps of calcified and fragmented elastic fibers. Systemic complications of PXE include yellowish skin papules (peu d’orange) commonly found on the neck, stomach, armpits and groin, calcification of the arteries, which leads to occlusive arterial disease that may lead to cerebrovascular disease, and mucosal bleeding in the stomach and the intestines.11 The disease can be life-threatening, causing sudden death due to a hemorrhage.3,11
The reported incidence of AS in patients with PXE varies between 59% and 87%.3
Ehler-Danlos syndromes (E)
EDS are a heterogenous group of 13 heritable connective tissue disorders characterized by genetic mutations that result in the abnormal synthesis of collagen.12 Common systemic manifestations include increased skin hyperextensability, joint hypermobility and tissue fragility.12 The vascular subtype (vED), which is associated with an autosomal dominant mutation in COL3A1, responsible for the synthesis of type III collagen, can manifest with vascular aneurysms and vascular rupture leading to hemorrhages.12
It is worth noting that according to the current literature, AS in EDS are extremely rare.13 In 1966, Green et al. reported a case of a mother and daughter with EDS who demonstrated AS in their retinas.14 This report has been referenced in the didactics of eye care as an association between EDS and AS.
Paget’s disease (P)
PD, also known as osteitis deformans, is a treatable skeletal disorder in which bones grow abnormally in size and shape secondary to focal areas of excessive osteoclastic bone resorption accompanied by a secondary increase in osteoblastic bone formation.15 It typically affects the femur, pelvis, lower lumbar area and cranium. While the cause is unknown, a strong genetic mutation (at the SQSTM1 loci of chromosomes 5 and 6) as well as viral (paramyxoviruses) influences have been shown. PD is more common in males and in patients older than 55 years. The diagnosis of PD involves radiographic imaging for bone deformities and laboratory testing. An elevated plasma total alkaline phosphatase (ALP) level is the most clinically useful marker of disease activity.15 ALP is an enzyme normally produced in several organs with the highest concentrations found in the bones and liver. It is not uncommon to find normal levels of ALP in patients with PD. Therefore, one should also measure γ-glutamyltranspeptidase levels to differentiate from liver disease that can also result in elevated ALP activity. The incidence of AS in patients with PD ranges from 8% to 15%.3
Sickle cell disease (S)
Sickle cell disease is inherited from parents with an autosomal recessive pattern. It is more common in individuals with sub-Saharan ancestry. Human adults have red blood cells (RBC) with hemoglobin A, which is composed of two alpha-globin proteins with two beta-globin proteins. In sickle hemoglobin (HbS), a single point mutation substitutes valine for glutamic acid at the sixth position in the beta-globin chain. RBCs containing HbS have a lifespan of 10 to 20 days, unlike healthy RBCs that live between 90 and 120 days. The “sickling” or collapse of the red blood cells leads to vascular occlusions, which result in ischemia of surrounding tissue and finally necrosis of the blood vessel wall. Diagnosis of sickle cell disease is made by obtaining a complete blood count, blood smears to identify sickling RBCs, and electrophoresis.
AS are most common in patients with HbS or sickle cell anemia (SCA).16 Pain and/or swelling of hands and feet are often an early manifestation of SCA. Organ damage from SCA can affect any organ system, including the bones, spleen, liver, brain, lungs, kidneys and joints.
The incidence of AS in patients with sickle cell hemoglobinopathies varies between 0.9% and 6%.3
Idiopathic (I)
AS are idiopathic in less than 50% of cases and associated with concurrent disease in more than half of patients presenting with AS.3
Ocular complications: choroidal neovascularization
CNVM is a growth of new blood vessels de novo (vasculogenesis) or from pre-existing blood vessels (angiogenesis). The new blood vessels often originate from the choroid and pass through a break in BrM into the sub-RPE or subretinal space. These vessels form networks or nets. CNVM appears to be a repair process initiated by tissue loss and/or damage that results in choroidal scar formation. Any pathologic process that involves damage to the RPE, the BrM and/or choriocapillaris can be complicated by CNVM. Upregulation of VEGF, which is a known factor in angiogenesis, is seen in CNVM. Its cause remains unclear. VEGF upregulation is known to occur secondary to hypoxia, high glucose and protein kinase C activation, advanced glycation end products, reactive oxygen species, activated oncogenes, and a variety of cytokines. Once secreted, VEGF binds to its tyrosine kinase receptors in endothelial cells activating several signal transduction pathways. This in turn induces vascular permeability, endothelial cell proliferation and cell migration, which results in the formation of new vessels.
CNVM is the major cause of vision loss and it affects 70-86% of patients with AS. CNVM also develops in the contralateral eye of 71% of these patients.3-4,17 Overall, the most common causes of CNVM are age-related macular degeneration, presumed ocular histoplasmosis syndrome, myopic macular degeneration, trauma and AS. CNVM can be asymptomatic when extrafoveal or it may present with symptoms of metamorphopsia or scotoma when present within the temporal arcades. OCT, FAF and angiography are used to confirm the diagnosis and evaluate treatment efficacy.
Diagnostic tools for CNVM
Amsler grid
In 1947, Marc Amsler developed a grid of horizontal and vertical lines to monitor the central visual field.18 Amsler grid is most commonly used to detect visual disturbances in the macular area of the eye.
Angiography
FA and ICGA were historically used to confirm the diagnosis of CNVM, determine the need for retreatment, and determine treatment efficacy. Classic or subretinal CNVM, which is classified as a type 2 lesion, is better visualized by FA than ICGA. Because of their irregular and ill-defined features, type 1 lesions, such as occult or sub-RPE CNVM, are difficult to detect on FA. Therefore, the fundamental advantage of ICGA is it allows for early detection and localization of type 1 CNVM.
OCT and OCTA
OCT has become an essential tool in diagnosis as well as determining the need for treatment, monitoring course, and determining the need for retreatment of eyes with CNVM. With advances in OCT technology, it is now possible not only to visualize subretinal fluid, pigment epithelial detachments and overlying retinal edema but also to identify CNVM. Recently, the introduction of OCTA has allowed physicians to visualize not only the retinal structures but also retinal vascular flow without the injection of dye.
Treatment for angioid streaks
Eyes with AS that are asymptomatic are observed. Systemic associations should be ruled out by properly obtaining a detailed history and with the assistance of a medical doctor when needed. Home Amsler grid testing should be recommended to monitor for potential ocular complications, and patients should be advised to report any changes immediately to their eye doctor.
Treatment options for choroidal neovascularization
The goal of CNVM treatment is to prevent choroidal scarring and accompanying scotoma. However, currently, there is no cure for this chronically active disease. Initially, laser photocoagulation and photodynamic therapy were used with relatively poor results and frequent recurrence.19 In 2015, Alagöz et al. showed that 65% of eyes with CNVM due to angioid streaks demonstrated an improvement or stabilization in visual acuity with a mean number of five intravitreal injections of bevacizumab over a mean period of 23 months.17 Over the past decade, intravitreal anti-VEGF medications, such as bevacizumab, ranibizumab and aflibercept, have shown promising results of reducing disease activity and stabilizing or improving visual acuity.20-21 Early diagnosis and intervention are crucial to improving outcomes; therefore, patients with AS who develop CNVM should be urgently referred to a retinal specialist for intravitreal injections of an anti-VEGF agent.
Patient education
Although 50% of AS are not associated with systemic conditions, systemic workup to rule out possible common associations should be considered in all patients with AS. It is important to inform patients about the presence of AS and to advise them to use protective eyewear to guard against accidental blunt trauma. Consideration should also be given to any activities that may put patients at risk for trauma to the eyes or head such as boxing, martial arts, basketball or soccer. It is also wise to advise patients with AS to seek care as soon as possible in the event of trauma or any vision change. An Amsler grid along with written instructions for use should be given to all patients diagnosed with AS so they may monitor their central vision. Current treatments for CNVM show more favorable results in patients with less advanced disease, suggesting the initiation of treatment should be as early as possible.
Optometrists play an important role in educating patients about the status of their vision and the health of their eyes. There are no standardized methods for patient education. Patients may require different strategies for counseling; therefore, a patient-centered approach is encouraged. Discussion about the findings and education about the condition as well as the possible ocular complications and systemic associations is warranted. For patients presenting with AS without evidence of retinal complications, periodic Amsler grid testing at home is recommended for both eyes. It is important to educate patients on the proper use of an Amsler grid as well as advise them what they should do if they notice vision changes in either eye. In addition, it is important to stress the need for regular re-evaluations of the ocular fundus.
Critical-thinking concepts
This case illustrates key concepts and goals in the care of a patient with AS. Visual acuity remains very good in eyes with AS; therefore, a change, no matter how small, can indicate the presence of CNVM, the most common ocular complication in eyes with AS. For this reason, Amsler grid testing should be performed early in the clinical examination to identify any macular abnormalities. In the case presented here, the CNVM observed with funduscopy and confirmed with OCT and OCTA was located a significant distance from the fovea and without subfoveal extension. Therefore, it was an unlikely cause for the patient’s complaint of blurry vision at near. Presbyopia or uncorrected refractive error are the most likely cause of monocular blurry vision at near in this patient’s age group in the absence of pathology affecting the central cornea (e.g., dry eye, edema or scarring) or the central lens (e.g., sclerosis or opacification).
Assessment of learning objectives
Instructors can evaluate students on the learning objectives of this case report in the following ways. The case can be shared with the students ahead of the learning session or presented on slides projected onto a large classroom screen at the beginning of the learning experience. Students can be asked to describe, compare and contrast the findings seen on the fundus photographs as well as the OCT and OCTA scans. Students should be able to accurately describe AS. Further discussion should focus on the differential diagnosis and common systemic associations of AS as well as the most common ocular complications. It is important to discuss the role of an optometrist in managing this condition and in patient education and counseling. Formative assessment can take place during group discussions of the case. Knowledge assessment can also be summative through oral or written quizzes.
Conclusion
Angioid streaks are cracks in Bruch’s membrane. In 50% of patients, they are associated with systemic diseases that require proper management. The most frequent systemic association is PXE. By themselves, AS do not cause decreased visual acuity even when crossing into the foveal area. CNVM is the most common retinal complication found in eyes with AS and should be suspected if symptoms of blurry or distorted vision occur. Patients need an urgent referral to a retinal specialist for treatment of CNVM in hopes of preserving vision. Finally, patients with a systemic association need a referral to the appropriate specialist for evaluation and treatment of the associated systemic pathology.
References
- Doyne RW. Choroidal and retinal changes: the results of blows on the eyes. Trans Ophthalmol Soc UK. 1889;9:128-40.
- Knapp H. On the formation of dark angioid streaks as an unusual metamorphosis of retinal hemorrhage. Arch Ophthalmol. 1892;21:289-92.
- Chatziralli I, Saitakis G, Dimitriou E, Chatzirallis A, Stoungioti S, Theodossiadis G, Theodossiadis P. Angioid streaks: a comprehensive review from pathophysiology to treatment. Retina. 2019 Jan;39(1):1-11.
- Georgalas I, Papaconstantinou D, Koutsandrea C, Kalantzis G, Karagiannis D, Georgopoulos G, Ladas I. Angioid streaks, clinical course, complications, and current therapeutic management. Ther Clin Risk Manag. 2009 Feb;5(1):81-9.
- Gliem M, Finger RP, Fimmers R, Brinkmann CK, Holz FG, Charbel Issa P. Treatment of choroidal neovascularization due to angioid streaks: a comprehensive review. Retina. 2013 Jul-Aug;33(7):1300-14.
- Ament CS, Zacks DN, Lane AM, et al. Predictors of visual outcome and choroidal neovascular membrane formation after traumatic choroidal rupture. Arch Ophthalmol. 2006 Jul;124(7):957-66.
- Ikuno Y, Sayanagi K, Soga K, Sawa M, Gomi F, Tsujikawa M, Tano Y. Lacquer crack formation and choroidal neovascularization in pathologic myopia. Retina. 2008 Oct;28(8):1124-31.
- Ohno-Matsui K, Morishima N, Ito M, Tokoro T. Indocyanine green angiographic findings of lacquer cracks in pathologic myopia. Jpn J Ophthalmol. 1998 Jul-Aug;42(4):293-9.
- Novotny HR, Alvis DL. A method of photographing fluorescence in circulating blood in the human retina. Circulation. 1961 Jul;24:82-6.
- Tripathy K, Quint JM. Angioid streaks. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan. 2021 Aug 21. PMID: 30844178.
- Chassaing N, Martin L, Calvas P, Le Bert M, Hovnanian A. Pseudoxanthoma elasticum: a clinical, pathophysiological and genetic update including 11 novel ABCC6 mutations. J Med Genet. 2005 Dec;42(12):881-92.
- Maraj B, Harding-Theobald E, Karaki F. Vascular Ehlers-Danlos syndrome presenting as a pulsatile neck mass: a case report and review of literature. J Gen Intern Med. 2018 Jul;33(7):1192-1195.
- Singman EL, Jefferson JD. Angioid streaks are not a common feature of Ehlers Danlos syndrome. JAMA Ophthalmol. 2019;137(3):239.
- Green WR, Friedman-Kien A, Banfield WG. Angioid streaks in Ehlers-Danlos syndrome. Arch Ophthalmol. 1966 Aug;76(2):197-204.
- Walsh JP. Paget’s disease of bone. Medical journal of Australia. 2004 Sep 6;181(5):262.
- lagouz M, Jyothi S, Gupta B, Sivaprasad S. Sickle cell disease and the eye: old and new concepts. Surv Ophthalmol. 2010 Jul-Aug;55(4):359-77.
- Alagöz C, Alagöz N, Özkaya A, et al. Intravitreal bevacizumab in the treatment of choroidal neovascular membrane due to angioid streaks. Retina. 2015 Oct;35(10):2001-10.
- Augustin AJ, Offermann I, Lutz J, Schmidt-Erfurth U, Tornambe P. Comparison of the original Amsler grid with the modified Amsler grid: result for patients with age-related macular degeneration. Retina. 2005 Jun;25(4):443-5.
- Pece A, Avanza P, Galli L, Brancato R. Laser photocoagulation of choroidal neovascularization in angioid streaks. Retina. 1997;17(1):12-6.
- Finger RP, Charbel Issa P, Schmitz-Valckenberg S, Holz FG, Scholl HN. Long-term effectiveness of intravitreal bevacizumab for choroidal neovascularization secondary to angioid streaks in pseudoxanthoma elasticum. Retina. 2011 Jul-Aug;31(7):1268-78.
- Vaz-Pereira S, Collaço L, De Salvo G, van Zeller P. Intravitreal aflibercept for choroidal neovascularisation in angioid streaks. Eye (Lond). 2015 Sep;29(9):1236-8.



