PEER REVIEWED
Differentiating Intracranial Pathology Using Visual Fields
Emily Humphreys, OD, FAAO, Amy Moy, OD, FAAO, CPCO, FNAP
One of the most crucial aspects of visual field testing in optometry is to help localize post-chiasmal lesions and rule out intracranial pathology. This case highlights a systemic anticoagulation conundrum, as a patient with both antiphospholipid syndrome and a cerebral vascular malformation presented with neurological visual field loss. Antiphospholipid syndrome is a hypercoagulable condition, putting patients at high risk for stroke and needing significant anticoagulative therapy. On the contrary, cerebral vascular malformations are at increased risk of brain bleeds, especially in the setting of anticoagulation. This case emphasizes the importance of visual field testing in localizing neurological field loss and differentiating the conditions to initiate appropriate referral.
Key Words: Visual fields, post-chiasmal lesions, cerebral vascular malformations, antiphospholipid syndrome, magnetic resonance imaging
Background
Understanding the visual pathway is essential for diagnosing post-chiasmal lesions. This case, and specifically the systemic associations, is best geared towards final-year students and optometry residents. However, the importance of visual field testing and its role in the diagnosis of neurological conditions is imperative for students of all levels to understand. The following case highlights how visual field testing helped to distinguish between complications of an existing cerebral cavernous hemangioma and a new cerebrovascular accident in a hypercoagulable patient.
In this situation, a patient with both antiphospholipid syndrome and a cerebral vascular malformation presented with neurological visual field loss. Antiphospholipid syndrome is an immune-mediated disorder that affects 1-5% of the general population and can result in increased risk of thromboembolic stroke, deep vein thrombosis, and recurrent fetal loss.1,2,3 One or more of the above events must occur for official diagnosis. In addition, the patient must be positive for one or more of the antiphospholipid antibodies (anticardiolipin, anti-beta2-glycoprotein-1, lupus anticoagulant) by laboratory testing.3 A vitamin K antagonist is often necessary to prevent these major thrombotic events, with Warfarin still being the mainstay treatment.3
Cerebral vascular malformations are abnormal vascular configurations within the central nervous system.4,5,6 This can include the retina, as well as the brain and spinal cord. There are four subtypes: arteriovenous malformations (AVMs), cavernous malformations as depicted in this case, developmental venous anomalies, and capillary telangiectasias. All four subtypes are delicate and at risk of bleeding and causing progressive neurological deficits.4,5,6 These deficits are dependent on lesion location. Of note, the risk of bleed is increased in those patients who are anticoagulated.7
The challenge occurs when both conditions occur simultaneously, as communication between neurology and hematology is required to coordinate the appropriate level of systemic anticoagulation and antiplatelet therapy. Here, visual field analysis – both confrontation and automated – was used to help direct an emergency referral. A general understanding of two conflicting systemic diagnoses was required to make an informed recommendation to multiple medical disciplines. This case highlights optometry’s role in communicating within a multidisciplinary healthcare setting. Students will learn that appropriately interpreting visual fields is essential in caring for patients with intracranial pathology. Different visual field-testing modalities will also be reviewed.
Case Description
A 63-year-old white male veteran presented with complaints of “a small blurry spot just up and to the left in both eyes” that he had noticed the day before upon awakening. The visual change persisted since onset and was most noticeable when watching TV. The patient reported “the heads of the people on the left side of the TV are blotchy and gray”. The patient was alert and oriented. His demeanor was positive and participatory, and there were no obvious changes in his cognitive status. He denied all other neurological symptoms including transient monocular vision loss, nausea/vomiting, dizziness, numbness/weakness/tingling in any extremity, or trouble speaking/swallowing. No current headache, trauma, or change in medication was noted. All giant cell arteritis symptoms were reviewed and denied including scalp tenderness, jaw pain, fever, or aching joints.
The patient’s last full dilated eye exam had been four months prior to this visit. At that time, the patient had a minimal refractive error for which he wears reading glasses only. He was best corrected to 20/20 in both eyes at distance and near. The entrance testing, anterior segment, and dilated portion of the exam were all unremarkable at that visit. He denied any change in central vision, flashes, floaters, or diplopia since his last exam. The patient had no history of ocular disease diagnosis, surgery, or trauma. No significant family history of ocular disease was noted.
The systemic history of the patient is significantly more remarkable than his ocular history. The patient was diagnosed with a cavernous malformation of the right mesial temporal lobe 5 years prior after presenting with severe vertigo that lasted for 30 minutes. At the time, he was evaluated by a neurosurgeon and was not thought to require surgery. However, a year later, he suffered a ‘stroke’ when there was a small bleed from the cavernous malformation. This event was characterized by anarthria (inability to speak), difficulty swallowing, and lasting dysphonia (difficulty speaking due to a physical disorder of mouth, vocal cords, tongue and throat). Although unable to talk, he had no difficulty knowing exactly what he wanted to say. There was no motor nor visual deficit. He recovered most of the lost function within a day, but needed to work with a speech therapist for months thereafter. Ever since that event, he finds that his memory for people and recent experience has been poor compared to pre-event. After an additional evaluation the following year, the malformation was deemed operable but the patient opted not to pursue surgery. The patient follows regularly with neurology and was last evaluated three months prior to the current presentation. Given this history, it was suggested the patient take a baseline visual field. However, he never returned for this follow-up exam. His fields by confrontation had always been documented as full in both eyes.
In addition to the cavernous malformation, the patient was diagnosed recently (three months prior) with antiphospholipid syndrome while undergoing a work-up for a possible kidney transplantation. The patient has stage five chronic kidney disease secondary to post-streptococcal glomerulonephritis at the age of 15. During the work-up, the patient was found to be positive for all three specific antiphospholipid antibodies. After an additional MRI was conducted, the patient was found to have multiple prior small infarctions (frontal, thalamic and cerebellar) representing the required thromboembolic event for an antiphospholipid syndrome diagnosis. The patient follows regularly with hematology with his last visit one month prior to the current presentation. Given the concurrent presence of a cerebral vascular malformation, the patient was managed only on low dose aspirin (81mg) for antiplatelet therapy.
Other medical diagnoses are listed below with medications:
- Hypertension (Metoprolol Tartrate)
- Hyperlipidemia (Atorvastatin)
- Gout (Allopurinol)
Exam
With the patient’s very minimal hyperopic-astigmatic prescription, he was 20/20 OD and OS. Extraocular muscles were full, extensive, smooth, and accurate OU. Pupils were equal, round, and reactive to light without afferent pupillary defect OD and OS. Cover test revealed only a 1-2pd exophoria at distance in his current correction. Color vision (Ishihara) was 11/11 OD and OS. Cranial nerves II-XII were intact and symmetric.
Finger-counting fields appeared full OD and OS. However, the patient reported during testing that while he could count fingers, the blurry spot was persistent on the left side of the examiner’s face in a more central location. This was true in both eyes. The patient was asked to “draw” this blurry spot by using an Amsler grid at 30cm. The patient documented the scotoma in the superior left quadrant of the Amsler grid in both eyes. Given these findings, kinetic red-cap field testing was initiated to further investigate the scotoma.
During this test, two caps (commonly Tropicamide or Phenylephrine bottles) are presented simultaneously on both sides of the midline. Just like finger-counting fields, the provider is sitting at eye level one-meter away and each eye is tested individually. Starting at the center, the targets are moved outward together, with one cap in the temporal and one in the nasal quadrant. The patient is asked to alert the provider if any cap “disappears or changes color or appearance” in any way in comparison to the other cap. First the superior field is tested, followed by the inferior field.
In this case, the patient was unable to see the left superior cap in either eye at the start of the test. It wasn’t until the cap was presented further into the periphery that the patient became aware of the cap again. One could virtually “trace” the visual field defect in both eyes with the red cap. Testing elevated suspicion of the neurologic nature of the scotoma, as it continued to respect the vertical midline and was almost identical OD and OS in the superior left quadrant paracentrally.
The anterior segment was unremarkable OU on slit lamp examination. There were no significant lens changes and the angles were open to dilation OU. Intraocular pressure was 12mmHg OU by Perkins applanation tonometry. The patient was dilated with both Tropicamide 1% and Phenylephrine 2.5%. The dilated portion of the exam was unremarkable and unchanged from his prior exams. The optic nerves were pink without pallor or edema and 0.45 round cupping OU. The macula was flat and clear OD and OS. The periphery was flat and intact without holes, tears, or detachments OU. Importantly, as the patient was assumed to be in a hypercoagulable state, the arteries and veins held a normal 2/3 ratio without arteriole attenuation or venous dilation OU. There were no retinal hemorrhages or emboli noted OU.
A formal automated visual field was performed with a Humphrey Visual Field (HVF) 24-2 SITA Standard test (Figure 1). The tests were fairly accurate in both eyes. There were mildly high false negatives in the left eye. However, our suspicions were confirmed, with a left superior paracentral scotoma in both eyes respecting the vertical meridian. While the pattern standard deviation and total deviation plots don’t represent this well, the decibel values read as 0’s in the paracentral location. Surrounding points, although mildly decreased, are >10 decibels. An upper lid defect in both eyes was suspected as there was some decrease in sensitivity superiorly that crossed the vertical midline.

Figure 1. 24-2 SITA Standard HVF. Both visual fields were readable and reliable barring high false negatives OD. Although fixation losses OD were 0/15, the fixation bar at the bottom of the test does portray a lot of movement. As the patient was fatigued, a suspected superior lid defect was present OU – representing the only area where the defect crosses the vertical midline. Although loss of sensitivity was apparent in the left-superior field in both eyes, the densest loss (represented best by the sensitivity values) was paracentral. The superior paracentral quadranopic loss appears mildly incongruous (OS>OD). Click to enlarge
Communication with Neurology and Hematology
The automated visual field, in combination with the visual field entrance tests and chief complaint gave us a strong suspicion of new-onset intracranial pathology in the right occipital lobe. The hospital’s neurology team was paged at the conclusion of the exam to present the emergent findings of the case (Figure 2). The paracentral nature of the homonymous neurological deficit isolates the lesion to the occipital lobe pole on the right side of the brain, in the inferior striate cortex (below the calcarine fissure).8,9 Given that the cavernous malformation is located in the medial aspect of the right temporal lobe, it would be unlikely to cause a paracentral defect in the event of a bleed. A full homonymous quadranopsia or potentially hemianopia would be expected in that case, depending on the extent of the bleed.8,9 The patient is at high risk of a stroke in the setting of antiphospholipid syndrome. It was agreed that an MRI was needed to confirm this assumption. The neurology resident recommended communication with hematology to coordinate care. An MRI of the brain was performed the same day. The patient was scheduled to return for a follow-up visual field with the eye clinic in three months. Although visual field recovery in an ischemic stroke is unlikely, the majority of recovery is witnessed in the first 10 days, and is negligible after 10-12 weeks.10

Figure 2. This sample conversation represents key points to communicate with neurology in the care of this patient. Click to enlarge
MRI Report (Figure 3)
The MRI officially confirmed a stable cavernous malformation and a new occipital lobe infarct, consistent with visual field findings. The patient was admitted to the hospital’s inpatient facility with a care team consisting of neurology, neurosurgery, and hematology.
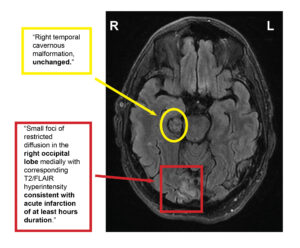
Figure 3. Horizontal MRI section representing both the new occipital lobe infarct and the unchanged cavernous malformation. Click to enlarge
Inpatient Admission
The patient was admitted for 1 week. During this time, the following decisions were made regarding his systemic management:
Hematology: Initially, the aspirin dose was increased to 325mg. Upon discharge, the patient commenced Warfarin. The patient was advised never to interrupt anticoagulation given high risk of thrombotic events even in the setting of the cavernous malformation. Continued work-up for renal transplantation was recommended.
Neurology and Neurosurgery: Given the stability of the cavernous malformation for five years, surgical resection of the malformation was not recommended. Given antiphospholipid syndrome, the risk of thromboembolic events vastly outweighs the risk of rebleeding from the malformation. Therefore, high-dose anticoagulation was recommended. Annual monitoring was recommended with MRI imaging.
Optometry Three-Month Follow-Up
The patient reported no change in his “blurry spot” after three months. A visual field confirmed this (Figure 4). A 30-2 HVF SITA Standard test was inadvertently performed instead of a 24-2 threshold test during this appointment, making direct comparison challenging. However, the paracentral 0 decibel values remain. Lids were not taped, and again, a superior lid defect was suspected. One could also postulate that the repeatable peripheral superior left defects could be due to the historical bleed of the cavernous malformation. Given the ischemic nature of the event, the findings were as expected and will likely remain stable without resolution.10 Annual follow-ups were scheduled going forward.

Figure 4. 30-2 SITA Standard HVF. The fields were readable but with less reliability than the initial visual field with high fixation losses OD>OS and high false negatives OU. Again, the superior lid defect was suspected OU. The paracentral nature of the left superior quadranopsia is more easily visualized in the 30-2 as opposed to the 24-2 – best represented in the gray scale and sensitivity values. It becomes difficult to compare and monitor for any resolution, as two different test modalities were used. However, stability was expected and recorded. Click to enlarge
Education Guidelines
This case is appropriate for third- and fourth-year optometry interns under the guidance of a facilitator. This can include but is not limited to clinical grand rounds, workshops, seminars and the traditional classroom. Students can be given the case details and discussion questions, either as individuals or in groups, and tasked with answering the questions with peer reviewed literature, either during the sessions or with answers brought to the session. During the session the facilitator should review the key concepts, discussion questions and assist students with fitting Fresnel prism to practice glasses and performing the Park’s and double Maddox rod tests on each other. Assessment of learning can be performed through grading of the discussion questions and participation in answering open ended questions, fitting of the Fresnel prism and performing Park’s and double Maddox rod tests.
Learning objectives
- To enhance competence in interpretation of Humphrey visual fields and identifying correlation with neurological defects
- Mapping neurological deficits, focusing specifically on the occipital lobe
- Introduce additional visual field entrance testing: kinetic red cap fields
Key concepts
At the conclusion of this case report, readers should be able to:
- Visual pathway, focusing on post-chiasmal lesions and resulting visual field defects
- Visual field testing modalities, including automated and non-automated options
- Basics of systemic anticoagulation and common conditions that may require it to prevent neurological complication
- Interdisciplinary management and communication within a multi-disciplinary setting
Learning Assessment
- Present students with simplified cases or pictures of neurological visual field loss and have them localize where in the brain they may expect a lesion or where on the visual field a specific lesion would present? (Figure 5)
- Example: “Draw a visual field representing damage to the optic radiations in the parietal lobe on the right side of the brain.”
- Example: “Where would you expect a lesion in the brain based on the following visual field?”
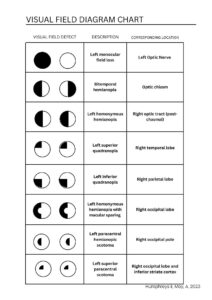
Figure 5. Table representing common neurological visual field defects. This can be used as a study or assessment tool. Click to enlarge
- Have students interpret different HVF printouts with neurological defects: analyzing reliability, location, and differentials.
- Have students pick a specific visual field test – either automated or entrance – and describe advantages and disadvantages. Focus on the difference of static versus kinetic, as well as automated versus non-automated. Priority should be given to the tests discussed in this paper (Figure 6).
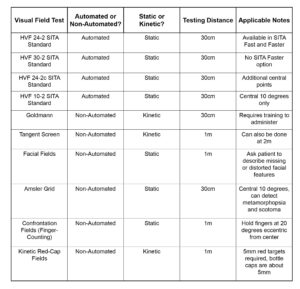
Figure 6. Table of common visual field tests, and those highlighted in this paper. Specific test parameters are listed and can aid in test initiation.12 Click to enlarge
Discussion questions
- How might you approach taking a case history in a patient with a localized blurry spot in their vision?
- What questions are important to ask in your case history to rule out stroke or other neurological conditions if suspected?
- What would your differential diagnoses be for this patient?
- What additional testing could you do for this patient during their eye exam to help work through your differentials?
- What are some important instructions to tell your patient before and during automated visual field testing to help improve reliability and patient performance?
- Discuss different conditions that use different visual field tests:
- HVF: 10-2? 24-2? 30-2?
- When would you use kinetic (e.g. Goldmann, Octopus) versus static fields (HVF). List some static versus kinetic visual field entrance tests.
- When communicating to neurology about this patient’s condition, what are some important aspects of the eye exam to point out?
- What other specialties may this patient follow-up with for his ongoing systemic management?
- How would you present the findings and need for referral to the patient?
Discussion
This case emphasizes the importance of visual field testing in localizing neurological field loss and differentiating the conditions to initiate appropriate referral. Post-chiasmal lesions that fall along the primary visual pathway will result in homonymous visual field defects (see Figure 5).8,9 The defects will respect the vertical midline and will be in the contralateral visual field in respect to the side of the lesion. This is due to the crossing of nasal retinal fibers in the optic chiasm. Therefore, the right optic tract contains information pertaining to the left visual field and vice versa. Following a synapse in their respective lateral geniculate nuclei in the thalamus, fibers in the visual pathway spread out to form the optic radiations, as they course through the parietal and temporal lobes.9 Inferior visual field information is carried through the parietal lobe, and superior visual field information is carried through the temporal lobe. It’s this dispersal of fibers that can result in quadranopsias if lesions are isolated to either lobe specifically.9 The more posterior the lesion falls along the visual pathway, the more congruous the visual field loss tends to be.9
The retinotopic organization of the occipital lobe is highly specific and can be extremely helpful in localizing defects.8,9 The macula is disproportionately represented in a concept known as magnification: 37% of the mesial surface of the occipital lobe represents the central 15 degrees of visual field.8 Therefore, post-chiasmal paracentral homonymous visual field loss is pathognomonic for an occipital lobe lesion, specifically involving the medial pole.8 Both maculopathies and optic neuropathies, if present bilaterally, could mimic paracentral homonymous defects and need to be ruled out.11 Often these can be visualized on dilated exam, however, may require additional testing. Central serous chorioretinopathy, branch retinal artery occlusions, or acute macular neuroretinopathy, are just a few conditions that, if bilateral, could present with visual fields that may be difficult to distinguish from a post-chiasmal lesion.11 However, these defects are less likely to be symmetric between the eyes and are more likely to cross the vertical meridian.11
There are many types of visual field examinations, both automated and non-automated. The choices made during the exam may be based on in-office availability. In this case, multiple visual field tests were used to work towards the diagnosis – both static and kinetic, as well as automated and non-automated. All testing is typically performed monocularly. Entrance testing is a non-automated perimetry option. These tests can also be static or kinetic. Confrontation fields (finger-counting), Amsler grid, facial fields, and kinetic red-cap fields were all used with this patient. During finger-counting, fields the targets are presented in the periphery (at about a 20-degree eccentricity from center12), therefore, the patient was able to count fingers in all quadrants easily OD and OS. Amsler grid and facial fields more easily highlighted the paracentral nature of the scotoma. Red-cap fields could trace the scotoma and was the only kinetic field option utilized in this case. In an analysis of different visual field entrance tests, Kerr et al. (2010) found that kinetic 5mm red-target fields have a higher combined overall sensitivity and specificity in comparison to finger counting fields.12
Static versus kinetic perimetry is an important consideration in test choice. There are automated and non-automated versions of both. Automated static perimetry, most commonly the HVF, has been demonstrated to be comparable to manual kinetic perimetry in the detection of both glaucoma and neurological disease.9 While automated static perimetry has largely replaced manual kinetic perimetry in an office setting, there are arguments that manual kinetic perimetry options, such as Goldmann and tangent screen, may be better at localizing neurological disease.9 An example of this is the detection of macular sparing. This phenomenon happens with lesions in the occipital lobe. However, a threshold 24-2 or 30-2 HVF does not have the capability to accurately detect this given the degrees of separation of the central points tested.9 Both Goldmann and the tangent screen have techniques that can specifically test this.9
In this case, the HVF was utilized for the automated perimetry option, and specifically the 24-2 SITA Standard test. Typically, performing the same test at follow-up visits is recommended for continuity and comparison.13 This was not the case in our patient, where a 30-2 SITA Standard was run at the three-month follow-up. The literature supports either 24-2 or 30-2 static central HVF testing for neurological and glaucomatous conditions, citing only developing idiopathic intracranial hypertension or pituitary lesions as cases where a 30-2 may be required to pick up more peripheral loss.14,15,16 The benefit of 24-2 testing is reduced test time, as it shortens the test in comparison to the 30-2 by about 28%.15 Although not run in this case, test time can be shortened even further by using SITA Fast of SITA Faster testing.13,17 Not specifically tested in relation to neurological disorders, both SITA Standard and SITA Fast testing have been deemed comparable in terms of glaucoma management.13
Central visual field testing can also aid in paracentral defect diagnosis. Both facial fields and an Amsler grid are non-automated tests used in this case. Unfortunately, as mentioned above, both the 24-2 and 30-2 automated HVF tests have difficulty mapping this area, as the degree of separation between central points is too large (6 degrees).9 This can be noted in our patient, as the defect is more obvious on the gray scale, but only represented by a single decibel value of 0 adjacent to the center target. This can make diagnosis challenging, and may even require an additional test to be run. The central 10-2 HVF program is better able to detect defects within the central 10 degrees, as points are only separated by 2 degrees.9,18,19 However, running two different tests is often unrealistic in practice. The HVF 24-2C Faster is a newer algorithm that tests an additional 10 central points, in conjunction with the typical 24 degrees of a SITA standard 24-2.18 In a comparison study to the 24-2 SITA Standard, the 24-2C Faster detected five and a half more central points on the total deviation plot and two more points on the pattern deviation plot than the 24-2, and was later confirmed accurate by the 10-2.19 The 10-2 remains the best mapper and predictor of visual field loss in the central 10 degrees. However, a test like the 24-2C reduces test time and acts as a good “first pass” for central loss while testing a larger visual field area.18,19
Establishing that the patient had recent onset neurogenic vision loss was only part of the diagnostic puzzle. The patient’s conflicting diagnoses and management resulted in a medical conundrum that needed confirmation by MRI and emergent referral to the appropriate specialty. In this case, a basic understanding of both antiphospholipid syndrome and cerebral cavernous malformations is a necessity for any eye care provider to help start and facilitate communication between the appropriate medical professionals.
It would be very easy in this case to overlook other causes and attribute the field loss to the cavernous malformation, as any bleeding into the right mesial temporal lobe could result in a left superior quadranopsia.16 Cavernous malformations, also known as cavernous angiomas or hemangiomas, are classified as a cerebral vascular malformation. Other cerebral vascular malformations include venous anomalies, arterio-venous malformations (AVMs), and capillary telangiectasias. Cavernous malformations are the least common of vascular malformations, occurring in 0.4% of the general population.5 While more commonly sporadic, cavernous malformations can be familial, with genetic mutations identified in both Hispanic and white non-Hispanic families specifically.5,6 Just like in the retina, all cavernous malformations have the classic “mulberry” appearance consisting of dilated thin-walled capillaries with a simple endothelial lining and a thin fibrous adventitia.5,6 This puts them at risk for leaking blood into surrounding tissues.5,6 Any spontaneous hemorrhage of a cerebral cavernous malformation may result in seizures or progressive neurological deficits, such as visual field loss if along the primary visual pathway.5,6
Vascular malformations are fragile, as the lesions are dynamic and often undergo intralesional thrombosis and hemorrhage.7 Evidence suggests that previous hemorrhage increases the risk of subsequent hemorrhage.20 In addition, multiple case reports describe instances where antithrombotic treatment resulted in an intracranial hemorrhage from an existing cavernous malformation. However, no larger study supports this hypothesis.7 Given this, the consensus remains that patients should try and avoid any antithrombotic or anticoagulant therapy, especially if the patient has had a symptomatic hemorrhage in the past.7 Neurosurgical excision is the treatment of choice, with good surgical resection outcomes in about 97% of patients.20,21 However, resection depends largely on cerebral malformation location, as brainstem lesions are much more difficult to resect than those in the cerebrum.21 A large-scale literature review and meta-analysis performed by Horne et al. (2016) found that the cerebral malformations most likely to result in hemorrhage were those that initially presented with hemorrhage or focal neurological deficit, or if the malformation was located in the brainstem.20 Therefore, resection is typically offered only to those who have been previously symptomatic.21 The patient in this case chose to avoid excision, given stability over a 5 year period.
General knowledge about antiphospholipid syndrome was necessary in this case to come to the correct diagnosis, as a new occipital lesion was suspected. Antiphospholipid antibody syndrome (APS) is a systemic autoimmune disorder characterized by recurrent thromboembolic events in the presence of antiphospholipid antibodies.1,2,3 APS can be either a primary condition, or secondary in the presence of an additional autoimmune disease, most commonly systemic lupus erythematosus (SLE). The pathogenesis of APS is not fully understood, but essentially results from the action of autoantibodies on the body’s coagulation pathways. There are many autoantibodies involved, but three that are most common and typically tested for to confirm diagnosis: anticardiolipin (present in 70% of cases), anti-beta2-glycoprotein-1 (present in 10% of cases), and lupus anticoagulant (present in 20% of cases).2,3 For an official diagnosis, patients must test positive for at least one of the above autoantibodies, in addition to having at least one occurrence of venous or arterial thrombosis.3
The most common thrombotic events experienced by patients with APS are deep vein thrombosis, pregnancy morbidity, and stroke.2 Stroke is considered an arterial thrombotic event and occurs in around 19.8% of patients with APS unless properly and highly anticoagulated.1,2 Amaurosis fugax, representing cerebral ischemia, is the most common ocular manifestation in APS, reported in around five percent of patients.2 Although more rare, non-arteritic anterior ischemic optic neuropathy and retrobulbar optic neuritis have also been described. The cornerstone treatment is long-term anticoagulation with oral Warfarin. Additional adjunct therapies with thromboprophylaxis properties (aspirin/plavix) or direct anticoagulation properties (rivaroxaban/apixaban/edoxaban) are up for debate and their role is not entirely understood.2 Eye care providers should be aware of neuro-ophthalmic conditions that may result from APS. A good understanding of anticoagulation and antiplatelet therapies is also important for many conditions encountered regularly by optometrists.
Conclusion – Start here with the rest of this paper
In large multidisciplinary healthcare settings, optometrists can play a pivotal role as part of a patient’s medical team. Expertise in analysis of visual field testing, as well as general systemic knowledge of hematological and neurological disorders, helped channel the patient into appropriate inpatient care. An optometrist’s role in communicating with other healthcare providers, especially in urgent or emergent situations, can help save a patient’s life. This case represented a complex example of how visual fields were used to help decipher the consequences of conflicting conditions, APS and a cerebral cavernous malformation, as well as the importance of interdisciplinary communication that can start at the optometrist.
Summary
SOPs (congenital or acquired) are frequently encountered in optometric practice and primary care optometrists should be comfortable in their ability to diagnose, treat and co-manage cases of SOP. Ocular trauma is a common cause of SOP implicated in 40% of acquired cases of SOP. Occasionally direct trauma to the superior oblique tendon can also result in unilateral SOP. Since a high number of traumatic SOP cases fail to show abnormal findings on imaging,3,8 determination of exact etiology can be complicated. However, most cases of traumatic SOP resolve spontaneously over a period of weeks to 6 months. If diplopia persists longer than 6 months, surgery may be required to realign the eyes.
References
-
- Dhir V, Pinto B. Anitophospholipid syndrome: A review. J Mahatma Gandhi Inst Med Sci. 2014; 19(1):19-27. doi: 10.4103/0971-9903.126231
- Rato ML, Bandeira M, Romão VC, Aguiar de Sousa D. Neurological Manifestations of the Antiphospholipid Syndrome – an Update. Curr Neurol Neurosci Rep. 2021; 21(8):41. doi: 10.1007/s11910-021-01124-z
- Miyakis S, Lockshin MD, Atsumi T, Branch DW, Brey RL, Cervera R, Derksen, RHMW, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS) J Thromb Haemost. 2006 Feb;4(2):295–306. doi: 10.1111/j.1538-7836.2006.01753.x
- Al-Shahi R, Bhattacharya JJ, Currie DG, Papanastassiou V, Ritchie V, Roberts, RC, Sellar RJ, et al. Prospective, population-based detection of intracranial vascular malformations in adults: the Scottish Intracranial Vascular Malformation Study (SIVMS). Stroke. 2003 May;34(5):1163-9. Epub 2003 Apr 17. doi: 10.1161/01.STR.0000069018.90456.C9
- Rigamonti D, Hadley MN, Drayer BP, Johnson PC, Hoenig-Rigamonti K, Knight JT, Spetzler, RF. Cerebral cavernous malformations. Incidence and familial occurrence. N Engl J Med. 1988 Aug 11;319(6):343-7. doi: 10.1056/NEMJ198808113190605
- Aiba T, Tanaka R, Koike T, Kameyama S, Takeda N, Komata T. Natural history of intracranial cavernous malformations. J Neurosurg. 1995 Jul;83(1):56-59. doi: 10.3171/jns.1995.83.1.0056
- Flemming KD, Link MJ, Christianson JH, Brown RD. Use of antithrombotic agents in patients with intracerebral cavernous malformations. J Neurosurg. 2013 Jan;118(1):43-46. doi: 10.3171/2012.8.JNS112050
- Wong AM, Sharpe JA. Representation of the visual field in the human occipital cortex: a magnetic resonance imaging and perimetric correlation. Arch Ophthalmol. 1999 Feb;117(2):208-217. doi: 10.1001/archopht.117.2.208
- Wong AM, Sharpe JA. A comparison of tangent screen, goldmann, and humphrey perimetry in the detection and localization of occipital lesions. Ophthalmology. 2000 Mar;107(3):527-44. doi: 10.1016/s0161-6420(99)00092-5
- Gray CS, French JM, Bates D, Cartlidge NEF, Venables GS, James OF. Recovery of visual fields in acute stroke: homonymous hemianopia associated with adverse prognosis. Age Ageing. 1989 Nov;18(6):419–421. doi: 10.1093/ageing/18.6.419
- Padungkiatsagul T, Leung LS, Moss HE. Retinal Diseases that Can Masquerade as Neurological Causes of Vision Loss. Curr Neurol Neurosci Rep. 2020 Sep 15;20(11):51. doi: 10.1007/s11910-020-01071-1
- Kerr NM, Chew SSL, Eady EK, Gamble GD, Danesh-Meyer HV. Diagnostic accuracy of confrontation visual field tests. Neurology. 2010 Apr 13;74(15):1184-1190. doi: 10.1212/WNL.0b013181d90017
- Heijl A, Patella VM, Chong LX, Iwase A, Leung CK, Tuulonen A, Lee GC, et al. A New SITA Perimetric Threshold Testing Algorithm: Construction and a Multicenter Clinical Study. Am J Ophthalmol. 2019 Feb;198:154-165. doi: 10.1016/j.ajo.2018.10.010
- Hepworth LR, Rowe FJ. Programme choice for perimetry in neurological condition (PoPiN): a systematic review of perimetry options and patterns of visual field loss. BMC Ophthalmol. 2018;18(241):1-9. doi: 10.1186/s12886-018-0912-1
- Rowe FJ, Chenye CP, Garcia-Fiñana M, Noonan CP, Howard C, Smith J, Adeoye J. Detection of visual field loss in pituitary disease: peripheral kinetic versus central static. Neuroophthalmol. 2015 Jun;39(3):116-24. doi: 10.3109/01658107.2014.990985
- Khoury JM, Donahue SP, Lavin PJ, Tsai JC. Comparison of 24-2 and 30-2 perimetry in glaucomatous and nonglaucomatous optic neuropathies. J Neuroophthalmol. 1999 Jun;19(2):100-8.
- Saunders LJ, Russel RA, Crabb DP. Measurement precision in a series of visual fields acquired by the standard and fast versions of the Swedish interactive thresholding algorithm: analysis of large-scale data from clinics. JAMA Ophthalmol. 2015 Jan;133(1):74-80. doi: 10.1001/jamaopthalmol.2014.4237
- Behera G, Nath A, Ramasamy A, Kaliaperumal S. Comparing Static Perimetry Protocols of Central Field Testing among Patients with Glaucoma. Optom Vis Sci. 2023 Jun 1;100(6):406-411. Epub 2023 May 1. doi: 10.1097/OPX.0000000000002020
- Phu J, Kalloniatis M. Comparison of 10-2 and 24-2C Test Grids for Identifying Central Visual Field Defects in Glaucoma and Suspect Patients. Ophthalmology. 2021 Oct;128(10):1405-1416. Epub 2021 Mar 17. doi: 10.1016/j.ophtha.2021.03.014
- Horne MA, Flemming KD, Su IC, Stapf C, Jeon JP, Li D, Maxwell, SS, et al. Clinical course of untreated cerebral cavernous malformations: a meta-analysis of individual patient data. Lancet Neuro. 2016 Feb;15(2):166-173. doi: 10.1016/S1474-4422(15)00303-8
- Chang EF, Gabriel RA, Potts MB, Berger MS, Lawton MT. Supratentorial cavernous malformations in eloquent and deep locations: surgical approaches and outcomes. J Neurosurg. 2011 Mar;114(3):814-827. doi: 10.3171/2010.5.JNS091159



