PEER REVIEWED
Horner Syndrome
Shelly Kim, OD, FAAO, and Sarah Rogers, OD
Abstract
Horner syndrome (HS) classically presents as a clinical triad: unilateral ptosis, miosis, and anhidrosis. The condition is caused by damage to the ipsilateral oculosympathetic pathway. This teaching case report presents a case of iatrogenic HS after an ipsilateral carotid endarterectomy procedure. It reviews the use of diagnostic pharmaceutical topical agents and provides a guided approach to ordering radiological imaging studies. Treatment of the condition is directed at identification and management of the underlying cause. Although most causes of HS are benign, painful HS could indicate carotid artery dissection, which carries a substantial risk of ischemic stroke and can be potentially life-threatening.
Key Words: Horner syndrome, oculosympathetic palsy, apraclonidine, optometry
Background
This case involves a 74-year-old Caucasian male diagnosed with iatrogenic Horner syndrome (HS) following recent ipsilateral carotid endarterectomy. HS is caused by disruption in the oculosympathetic pathway, which gives rise to the classic triad: ptosis (drooping of the upper eyelid), miosis (constricted pupil), and anhidrosis (absence of sweating).1 Most cases of HS are acquired due to damage to the sympathetic nerve supply, but congenital HS can occur due to trauma during birth.2 HS is usually due to a benign condition, but clinicians should always consider a more serious or life-threatening etiology such as neoplasm or carotid dissection.3 This case report highlights the importance of the optometrist’s role in detection of HS and evaluation and management of an HS patient. It focuses on the importance of accurate diagnosis, as well as radiologic imaging studies needed, to investigate the underlying cause of the condition. This case is appropriate for third- and fourth-year optometry students, optometry residents, and optometry providers.
Case Description
A 74-year-old Caucasian male presented to the eye clinic with a non-specific complaint of right eye irritation. The examiner noticed right upper eyelid ptosis. During case history and questioning, the patient reported this began about 4 days after right-side carotid endarterectomy. This surgery was performed for carotid artery stenosis 1 month prior. The patient had an uneventful left-side carotid endarterectomy approximately 1 year prior. He denied symptoms of amaurosis fugax, head and neck pain, and diplopia. He was also unaware of anhidrosis on the right side of his face.
The patient’s medical history was significant for type 2 diabetes, hypertension, hyperlipidemia, coronary artery disease status post coronary artery bypass graft, and bilateral carotid artery stenosis status post bilateral endarterectomy.
His best-corrected visual acuity was 20/50 in the right eye (OD) and 20/40 in the left eye (OS). The vision was reduced in both eyes due to moderate cataracts. Extraocular movement was smooth and unrestricted in all gazes. Confrontation visual fields were full in each eye with no pain or diplopia. Intraocular pressure (IOP) was 15 mmHg OD and 17 mmHg OS. Pupil assessment revealed anisocoria that was greater in dim illumination, consistent with miosis OD (Figure 1, Table 1). Gross external examination revealed 2-mm upper lid ptosis and reverse lower lid ptosis in the right eye.
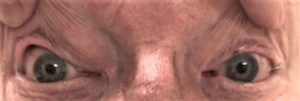 Figure 1. Miotic right pupil prior to instillation of diagnostic apraclonidine. Click to enlarge |
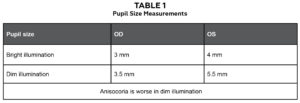 Table 1.Click to enlarge |
One drop of apraclonidine 1% was instilled into each eye at 2:29 p.m., 2:36 p.m., and 2:41 p.m. At 3:24 p.m., 55 minutes after the initial dose of apraclonidine, both pupils were re-evaluated. The right pupil dilated and the left pupil remained unchanged, indicating reversal of anisocoria. The patient was diagnosed with right-side HS with high suspicion of an iatrogenic cause. To rule out other potential causes, non-urgent MRI of the brain and brainstem and magnetic resonance angiography (MRA) of the head and neck were ordered. All studies were unremarkable. The patient’s cardiologist and vascular surgeon were alerted, and the patient was instructed to continue follow-up care with his healthcare team.
Education Guide
The Education Guide includes the necessary information for teaching and discussing the case. The learning objectives, key concepts, and discussion points should guide the teaching objectives.
Key concepts
- Sympathetic innervation of the eye
- Hallmark signs and symptoms of HS
- Critical-thinking in diagnosis, using diagnostic tests in-office to aid in making the diagnosis
- The importance of the eyecare provider in a team of healthcare professionals
Learning objectives
At the conclusion of this case discussion, participants should be able to:
- Describe the classic triad seen in HS
- Try to illicit localizing signs with a thorough case history
- Understand the different underlying causes of HS
- Differentiate HS from other pupil and ptosis conditions
- Assess the urgency of the condition and outline a management plan for the patient
Discussion questions
1. Knowledge, concepts, facts, and information required for critical review of the case:
- Describe the classic presentation of a patient with HS
- Understand the different topical pharmaceutical agents used in the diagnosisand localization of HS: cocaine, hydroxyamphetamine, and apraclonidine
2. Differential diagnosis
- What other conditions are differential diagnoses?
- How can HS be differentiated from other pupil and ptosis anomalies?
- What in-office pharmaceutical test was performed in this case to diagnose the condition?
- Should other differential diagnoses have been considered?
3. Patient management and the role of the optometrist
- What are appropriate management options?
- What is an appropriate follow-up schedule?
- What is the prognosis for a patient with HS?
- What is the urgency of additional testing?
- Summarize interprofessional team strategies for improving care coordination and communication to improve patient outcomes
4. Critical-thinking
- Could the diagnosis be made without topical pharmaceutical agents?
- How would the treatment and management plan change if the patient develops late-onset pain?
- How would management have changed in a patient with limited insurance coverage?
- Considering the high likelihood of iatrogenic HS in this case, did the patient require subsequent radiologic studies?
Discussion
HS is a rare condition that presents with unilateral ptosis, miosis, and facial anhidrosis due to disruption of sympathetic innervation to the eye.4 It was first described in animals by Francois Pourfour du Petit in 1727. It was investigated further by the French physiologist Claude Bernard, who observed the condition in a soldier who sustained a gunshot injury to his neck in 1854.3,4 The condition was formally described and correctly attributed to oculosympathetic paresis by Swiss ophthalmologist Johann Friedrich Horner.3
Anatomy
To understand the features of this syndrome, it is critical to understand the sympathetic innervation to the eye. The nerve supply is mediated by three neurons that originate in the posterolateral hypothalamus and end as the long ciliary nerves to supply the iris dilators and superior tarsal muscles (Müller’s muscles).4
The central, first-order, neurons are located in the posterolateral hypothalamus and descend through the midbrain and pons uncrossed, terminating at the C8-T1 level of the spinal cord in the intermediolateral cell columns (ciliospinal center of Budge and Waller).1,4 The pre-ganglionic, second-order, neurons exit from the ciliospinal center of Budge and Waller at the level of C8-T1 and pass across the pulmonary apex.1 They ascend through the stellate ganglion to synapse in the superior cervical ganglion at the level of C3-C4.4 The post-ganglionic, third-order, neurons branch off into the sudomotor and vasomotor fibers, which follow the external carotid artery and innervate the sweat glands and blood vessels of the face.4,5 The remaining fibers travel in the wall of the internal carotid artery and continue to the cavernous sinus where they join the abducens nerve (CN VI) before joining the ophthalmic division (V1) of the trigeminal nerve (CN V) as the long ciliary nerves.4
Etiology
HS is an uncommon condition and occurs with a frequency of approximately 1 in 6,000 people. It can occur at any age and in any ethnicity.6 It is primarily an acquired condition secondary to systemic or local diseases or iatrogenic causes.7 However, approximately 5% of cases are congenital.5
First-order neurons are mostly affected by intracranial conditions that can include:4,8
- stroke, most commonly lateral medullary infarction (Wallenburg syndrome)
- Arnold-Chiari malformation
- basal meningitis
- intracranial tumors (basal skull or pituitary)
- demyelinating lesions
- spinal cord tumors
- neck/cervical cord trauma
- syringomyelia
Second-order neurons travel across the thoracic region and are affected by:4,8
- malignancies involving the apex of the lung (Pancoast tumor, mesothelioma)
- cervical rib injuries
- mediastinal lymphadenopathies
- lumbar epidural anesthesia
- iatrogenic (thyroidectomy, radial neck dissection, tonsillectomy, coronary artery bypass grafting, chiropractic manipulation, or carotid angiography)5,9
Third-order neurons are near the internal carotid artery and cavernous sinus and are affected by:
- carotid cavernous fistula
- carotid artery dissection (commonly due to trauma) or aneurysm
- other internal carotid artery lesions (thrombosis, endarterectomy, stenting, radial neck surgery, trauma)
- cluster headaches or migraines
- Raeder paratrigeminal syndrome
- herpes zoster infection10
- giant cell arteritis
- idiopathic causes
Clinical features
Although HS classically presents with a triad of symptoms ― ptosis, miosis, and anhidrosis ― the clinical features may vary depending on the location of the lesion and degree of innervation.1
Ptosis
The ipsilateral upper lid appears drooped due to paresis of the sympathetically innervated Müller’s muscle.1 The Müller’s muscle is responsible for approximately 2 mm of elevation of the upper eyelid. The ptosis may be subtle, temporary, and variable. One study found that 12% of HS patients presented without an upper eyelid ptosis.1 The lower eyelid may appear mildly elevated, a “reverse ptosis,” due to weakening of the retraction of the lower eyelid as well.1 The resulting narrowed palpebral fissure may induce an appearance of enophthalmos.
Pupils
The ipsilateral pupil appears miotic due to the disruption of the sympathetic nervous supply. This leaves the parasympathetic supply inhibited and results in pupil constriction. The pupillary reaction to light and accommodation is normal, as these systems are not dependent on the sympathetic nervous supply.1
Paresis of the iris dilator muscle impairs pupillary dilation, leading to “dilation lag.” This can be seen clinically by illuminating from below with a hand-held light source and abruptly shutting off the lights. The normal pupil dilates immediately. The Horner pupil dilates several seconds later due to denervation of the parasympathetic tone.
Anhidrosis
Ipsilateral anhidrosis is dependent on the level of degeneration. First-order neuron lesions affect the ipsilateral body due to their central origin. Second-order neuron lesions cause ipsilateral facial anhidrosis. Post-ganglionic, third-order, neuron lesions occur after the vasomotor and sudomotor fibers have branched off and show little involvement of the face.
Facial anhidrosis can be evaluated clinically with the sweat friction test.11 A prism bar and the patient’s forehead are cleaned with alcohol. After allowing for the alcohol to dry, the prism bar is placed flat against one side of the forehead. It is drawn down with mild pressure. The same is done to the other side of the forehead. The side with anhidrosis will provide less resistance compared to the normal side.
Immediately following sympathetic denervation, the temperature of the skin can rise due to the affected vasomotor control. This leads to vasodilation of blood vessels, which can be seen clinically as facial flush, conjunctival injection, and epiphora.
Iris heterochromia
IpsilateraI iris heterochromia can occur in congenital HS.2 The sympathetic activity deficiency can cause interference with pigmentation of the melanocytes in the iris stroma, which can be seen clinically as iris hypopigmentation on the affected side.2
History and examination
A detailed history and physical examination are crucial in localizing the lesion in HS. One study concluded that the cause of HS will be known at the initial neuro-ophthalmologic consultation in two-thirds of patients.12
The following points should be considered when discussing the case history:
- history of exposure to a miotic or mydriatic agent to rule out pharmacologic mimics of HS (common agents that affect the pupil are pilocarpine, brimonidine, belladonna, scopolamine patches)
- balance, hearing, or sensory abnormalities can indicate a first-order neuron lesion
- past trauma or surgeries involving the face, head, neck, shoulder, or back may indicate a second-order neuron lesion5
- headache, diplopia, or pain can indicate a third-order neuron lesion (painful HS must be treated urgently as it is associated with carotid artery dissection, which carries an increased risk of ischemic stroke and mortality)
- presence and location of anhidrosis
- consider previous infections, including herpes zoster, tuberculosis, and syphilis1,4
- the value of examining old photographs to investigate the duration should not be underestimated3
During the physical examination, consider:
-
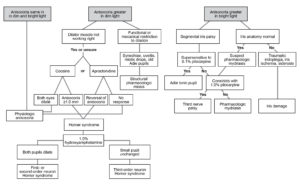
Figure 2. Anisocoria flowchart. Click to enlarge
unilateral facial flush, which may indicate congenital HS
- reactivity of pupils to light and accommodation (light-near dissociation may indicate an etiology separate from HS, such as dorsal midbrain syndrome, Argyll-Robertson syndrome, Adie’s tonic pupil)13
- measurement of ptosis and consider other causes of ptosis: cranial nerve 3 palsy, levator dehiscence, myasthenia gravis
- vision, including color vision and visual fields
- must differentiate other causes of anisocoria such as physiologic anisocoria, trauma, Adie’s tonic pupil (Figure 2)
Pharmacological testing in-office
Any combination of clinical signs of HS should be confirmed with diagnostic pharmaceutical agents in-office. These agents can aid in localizing the lesion responsible for causing the HS.
Topical cocaine: Cocaine acts as an indirect sympathomimetic agent, inhibiting the re-uptake of norepinephrine from the synaptic junction of the post-ganglionic fibers and the iris dilator muscle.14,15 Cocaine solution (ranging from 2-10%) is instilled into both eyes.4 Both pupils are evaluated after at least 30 minutes. If the sympathetic innervation is disrupted, norepinephrine cannot be released from the nerve terminal, and topical cocaine produces less pupillary dilation than in the normal eye.13,14 Anisocoria of 0.8 mm or more is diagnostic for HS.4
Disadvantages of using cocaine include inability to localize the lesion, high cost to compound, detection in urine, limited availability, and strict policy in most medical facilities on regulating controlled substances.12
Topical apraclonidine: This is the test of choice due to practicality and high sensitivity.3 It is commercially available as Iopidine 0.5-1% and is indicated to lower IOP in glaucoma patients. It can be used off-label for HS due to its weak alpha 1-adrenergic and strong alpha 2-adrenergic activity.1,16 The upregulation of alpha 1-receptors in HS results in denervation supersensitivity such that the affected pupil has greater mydriasis compared to the normal eye.17,18 The normal pupil is unaffected or minimally constricts due to the weak alpha-2 adrenergic effect.1
When apraclonidine is used prior to glaucoma laser surgery, the protocol is to instill one to two drops prior to the procedure.16 The effect of lowering IOP can be seen within the hour.16 However, the use of apraclonidine in confirming HS is an off-label use of the medication. A definitive protocol has not been established. The amount of time required for the maximum mydriatic effect on an affected pupil has not been well-studied. In addition, incomplete lesions of the sympathetic supply can produce mydriasis of the affected pupil without resulting in reversal of anisocoria, making it difficult to establish a definitive diagnosis.12 To illicit maximum response of the affected eye while minimally affecting the unaffected eye, practitioners may instill more than one drop of apraclonidine. The most common protocol in clinical practice is:
- instill 1 drop of apraclonidine 0.5-1% solution into both eyes (or 2 drops 5-10 minutes apart)15,19
- evaluate both pupils after 30-60 minutes1,10, 15,20
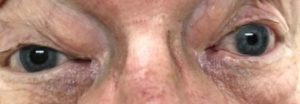
Figure 3. Reversal of anisocoria after apraclonidine testing. Click to enlarge
If the result is reversal of anisocoria where the affected pupil dilates while the normal pupil remains unchanged or constricts, this is confirmatory for HS (Figure 3). In some studies, topical apraclonidine has been shown to be superior to topical cocaine in detecting HS.13,15
Disadvantages to using topical apraclonidine include the inability to localize the lesion and limited effectiveness in acute HS as the denervation sensitivity takes 5-8 days to develop.1
Topical hydroxyamphetamine: This test is helpful in differentiating between pre- and post-ganglionic lesions. Hydroxyamphetamine causes the release of endogenous norepinephrine from the post-ganglionic axons into the neuromusclular junction of the iris dilator muscle. Two drops of 1% hydroxyamphetamine solution are instilled into each eye and the pupils are evaluated after 1 hour. Dilation of both pupils indicates a lesion of the first neuron or second neuron. If the smaller pupil fails to dilate, it indicates a lesion of the third-order neuron or post-ganglionic neuron.
The disadvantage to using topical hydroxyamphetamine is the need to wait 24-48 hours after cocaine testing.
Imaging
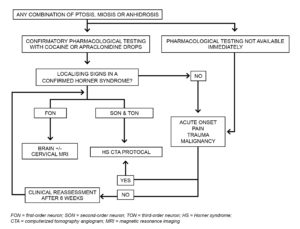
Figure 4. Diagram of recommended imaging techniques based on clinical findings associated with Horner syndrome.3 Click to enlarge
Radiological imaging studies play an important role in the diagnosis of the underlying cause of HS and ruling out pathologies that carry a substantial risk of morbidity and mortality (Figure 4).3 The provider should consider the sensitivity and specificity of the imaging technique, its availability, and the patient’s health status (kidney function when considering contrast media, ionizing radiation emitted during the procedure, etc.) when ordering a radiological imaging study.3 Studies should be ordered based on urgency of the symptoms and localizing signs.3 Timely diagnostic imaging is crucial to determining the primary cause of HS and directing further management of the underlying condition.3
When HS can be localized to the first-order neuron with associated neurological symptoms (ataxic hemiparesis or upper limb paresthesia), magnetic resonance imaging (MRI) of the brain and cervical and upper thoracic spinal cord should be performed. When the HS can be localized to the second- or third-order neuron, the computerized tomography angiogram (CTA) protocol should include the Circle of Willis and the orbits, down to the level of the aortic arch.3 These images process the head, neck, and lung apices.3 Patients with third-order neuron or painful HS, with or without localizing signs, should have CTA protocol emergently.3 HS without localizing signs, pain, trauma, or history of malignancy does not require urgent imaging. CTA protocol should be performed within 6 weeks.3
Treatment/management
Treatment options are based on the diagnosis and management of the underlying cause of HS. Timely diagnosis and referral to an appropriate specialist is of critical importance. This requires collaboration with an interprofessional healthcare team which may include:
- optometry
- ophthalmology (neuro-ophthalmology and/or oculoplastics)
- primary care
- pulmonology
- neurology
- neurosurgery
- vascular surgery
- radiology
- oncology
Differential diagnosis
The clinician must consider the extensive number of other potential causes of anisocoria and/or ptosis. Common causes that may present to an optometric practice and how their characteristics differ from that of HS are explained below.
Anisocoria21
- Physiologic anisocoria: Anisocoria is seldom greater than 0.8 mm. The difference in pupil size should remain equal in dim and bright illumination. The anisocoria remains stable over time.21
- Third-nerve palsy: This condition may cause pupillary dilation, although isolated pupillary dilation is not classically observed in third-nerve palsies. If pupillary dilation is seen in third-nerve palsies, it is usually accompanied by extraocular motility restriction and upper lid ptosis. Extraocular motility restriction is not seen in HS.
- Traumatic mydriasis: The patient may recall a history of trauma or surgery in the eye. Damage to the iris sphincter may be visualized in the slit lamp. The pupil may be irregular in shape due to sectoral damage of the iris sphincter muscle. The pupil in HS is typically round and without iris sphincter damage.21
- Pharmacologic mydriasis: The patient should be questioned about the recent use of vasoconstrictors (including antihistamines, decongestants, and stimulants), scopolamine patches, and herbals (such as jimson weed and belladonna). If diluted pilocarpine fails to constrict the pupil, the pupil is pharmacologically dilated.21
- Adie’s pupil: The dilated Adie’s pupil demonstrates light-near dissociation, where the pupil response is better to an accommodative stimulus than to light.21 Light-near dissociation is not present in HS.
Ptosis22
- Mechanical ptosis: This can be due to mechanical disturbance of the upper eyelid from causes such as tumors, infections, or inflammation. Levator dehiscence due to long-term rigid gas permeable contact lens wear should also be considered.22,23
- Myogenic ptosis: This results from the weakness of the levator palpebrae, which can be caused by various conditions including congenital myogenic ptosis, progressive external ophthalmoplegia, myotonic dystrophy, thyroid myopathy, or inflammation related to orbital myositis.22
- Myasthenia gravis: Variable ptosis, which may or may not include diplopia, can be related to an autoimmune disorder in myasthenia gravis. Ice test can be performed in-office to confirm the diagnosis. A positive result is noted if the eyelid rises 2 mm immediately after ice is applied for 2 minutes. A lab test for antibodies against the acetylcholine receptor can also be performed.22
Critical-thinking concepts
The purpose of this case report is to offer optometry students, residents, and eyecare providers a review of HS and a practical guide to investigate the cause of the condition. A discussion can be led with optometry students and residents in a classroom or clinic setting. Case history and exam findings should be presented in an organized manner (i.e., anterior segment findings to posterior segment findings). Critical-thinking should be encouraged to rule out other causes of anisocoria and work through differential diagnoses. A practical guideline to administer and interpret diagnostic testing, along with guidance on how to order subsequent radiologic studies should be reviewed. The importance of effectively communicating the assessment and plan to the patient should be emphasized.
The patient in this teaching case report presented with a generalized complaint of irritated eyes. While taking the patient’s case history, gross observation revealed an upper lid ptosis of the right eye. Therefore, case history questions were re-directed with the ptosis in consideration. The provider was able to elicit an acute history of right upper lid ptosis, which began 4 days after carotid endarterectomy on the right side about 1 month prior. The carotid endarterectomy procedure was performed 1 year prior on the contralateral side. The case history is ever-evolving and continues throughout the exam.
Due to the patient’s recent surgical history and upper lid ptosis, the pupils were carefully assessed. With the anisocoria being greater in the dark, HS was suspected and ultimately confirmed with topical apraclonidine 1% instilled in-office. The clinician should be able to differentiate other pupil anomalies that may present as anisocoria. The clinician should also be able to evaluate pupillary response in light and dim illuminations and be comfortable assessing light-near pupillary dissociation.
Due to high suspicion of iatrogenic HS, non-emergent radiologic imaging studies were ordered. MRI of the brain and brainstem and MRA of the head and neck were ordered to rule out other potential causes and assess the patient’s risk of stroke. The clinician should be aware of the potentially serious systemic effects of HS. The patient’s cardiologist and vascular surgeon were alerted of the condition, and the patient was instructed to follow-up with his healthcare team.
Conclusion
This teaching case report is intended to provide a comprehensive review of Horner syndrome for optometry students, residents, and eyecare providers. As frontline eyecare providers, it is important for optometrists to recognize the signs of HS and be proficient in using diagnostic pharmaceutical agents to make the diagnosis in-office. Furthermore, it is important to rule out more sinister causes of ptosis and anisocoria, especially if associated with pain. It is critical to assess the urgency with which radiologic imaging should be performed and work as part of a team of healthcare professionals to provide a comprehensive approach for managing the care of the HS patient.
References
- Kanagalingam S, Miller NR. Horner syndrome: clinical perspectives. Eye Brain. 2015 Apr 10;7:35-46. doi: 10.2147/EB.S63633.
- Weinstein JM, Zweifel TJ, Thompson HS. Congenital Horner’s syndrome. Arch Ophthalmol. 1980 Jun;98(6):1074-8. doi: 10.1001/archopht.1980.01020031064011.
- Davagnanam I, Fraser CL, Miszkiel K, Daniel CS, Plant GT. Adult Horner’s syndrome: a combined clinical, pharmacological, and imaging algorithm. Eye (Lond). 2013 Mar;27(3):291-8. doi: 10.1038/eye.2012.281.
- Khan Z, Bollu PC. Horner Syndrome. 2021 May 4. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-.
- Perry C, James D, Wixon C, Mills J, Ericksen C. Horner’s syndrome after carotid endarterectomy–a case report. Vasc Surg. 2001 Jul-Aug;35(4):325-7. doi: 10.1177/153857440103500414.
- Khan Z, Bollu PC. Horner Syndrome. 2022 May 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://pubmed.ncbi.nlm.nih.gov/29763176/.
- Reede DL, Garcon E, Smoker WR, Kardon R. Horner’s syndrome: clinical and radiographic evaluation. Neuroimaging Clin N Am. 2008 May;18(2):369-85, xi. doi: 10.1016/j.nic.2007.11.003.
- Alstadhaug KB. Ervervet Horners syndrom [Acquired Horner’s syndrome]. Tidsskr Nor Laegeforen. 2011 May 20;131(9-10):950-4. doi: 10.4045/tidsskr.10.0935.
- Sapalidis K, Florou M, Tsopouridou K, et al. Horner’s syndrome: an uncommon complication of thyroidectomy and selective lateral neck dissection. Curr Health Sci J. 2019 Jan-Mar;45(1):111-115. doi: 10.12865/CHSJ.45.01.15.
- Henry M, Johnson C, Ghadiali L, Raiji V. Horner’s syndrome following varicella vaccination. Neuroophthalmology. 2019 Feb 26;44(4):267-269. doi: 10.1080/01658107.2019.1583759.
- Rosenberg ML. The friction sweat test as a new method for detecting facial anhidrosis in patients with Horner’s syndrome. Am J Ophthalmol. 1989 Oct 15;108(4):443-7. doi: 10.1016/s0002-9394(14)73315-x.
- Trobe JD. The evaluation of Horner syndrome. J Neuroophthalmol. 2010 Mar;30(1):1-2. doi: 10.1097/WNO.0b013e3181ce8145.
- Thompson HS. Light-near dissociation of the pupil. Ophthalmologica. 1984;189(1-2):21-3. doi: 10.1159/000309380.
- Chen PL, Chen JT, Lu DW, Chen YC, Hsiao CH. Comparing efficacies of 0.5% apraclonidine with 4% cocaine in the diagnosis of Horner syndrome in pediatric patients. J Ocul Pharmacol Ther. 2006 Jun;22(3):182-7. doi: 10.1089/jop.2006.22.182.
- Bremner F. Apraclonidine is better than cocaine for detection of horner syndrome. Front Neurol. 2019 Jan 31;10:55. doi: 10.3389/fneur.2019.00055.
- Iopidine [package insert]. Forth Worth, TX: Alcon Laboratories, Inc.; 2001.
- Cooper-Knock J, Pepper I, Hodgson T, Sharrack B. Early diagnosis of Horner syndrome using topical apraclonidine. J Neuroophthalmol. 2011 Sep;31(3):214-6. doi: 10.1097/WNO.0b013e31821a91fe.
- Koc F, Kavuncu S, Kansu T, Acaroglu G, Firat E. The sensitivity and specificity of 0.5% apraclonidine in the diagnosis of oculosympathetic paresis. Br J Ophthalmol. 2005 Nov;89(11):1442-4. doi: 10.1136/bjo.2005.074492.
- Chu CJ, Ferris JD. Confirmation of Horner’s syndrome using apraclonidine eye drops. Postgrad Med J. 2011 Jun;87(1028):440. doi: 10.1136/pgmj.2010.113514.
- Kauh CY, Bursztyn LL. Positive Apraclonidine Test in Horner Syndrome Caused by Thalamic Hemorrhage. J Neuroophthalmol. 2015 Sep;35(3):287-8. doi: 10.1097/WNO.0000000000000222.
- Payne WN, Blair K, Barrett MJ. Anisocoria. 2022 Jun 20. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-.
- Díaz-Manera J, Luna S, Roig C. Ocular ptosis: differential diagnosis and treatment. Curr Opin Neurol. 2018 Oct;31(5):618-627. doi: 10.1097/WCO.0000000000000600.
- Katsoulos K, Rallatos GL, Mavrikakis I. Scleral contact lenses for the management of complicated ptosis. Orbit. 2018 Jun;37(3):201-207. doi: 10.1080/01676830.2017.1383475.



