PEER REVIEWED
Swan Syndrome Following Complicated RGP Lens Removal
Jenna D. Koskey, OD, FAAO, Dipl. ABO, ABCMO, and Emily M. Ulry, OD
Abstract
Swan syndrome is a rare condition of abnormal vascularization at a limbal wound site following cataract surgery. This case report involves a patient with suspected Swan syndrome confirmed by ocular history, exam findings and exclusion of other conditions. The case explores exam findings, differential diagnoses and treatment options and, due to the background and cause of Swan syndrome in this particular patient, addresses corrective lens options. This case is atypical for two reasons: 1) the rarity of the condition, which has been documented only a few times in the literature, and 2) the unique specifications of the glasses made for the patient when the condition resolved.
Key Words: Swan syndrome, hyphema, vitreous hemorrhage, aphakia, aniseikonia
Background
Swan syndrome is a rare condition first discovered in the 1970s. It occurs in patients with history of cataract extraction via scleral incision. Patients present with hyphema, vitreous hemorrhage and potentially angle neovascularization. Swan syndrome is often a diagnosis of exclusion, requiring careful case history including review of past surgeries, injuries and systemic disorders, and lab testing. Treatment and management of patients with Swan syndrome are highly dependent on severity of findings as well as recurrences. Patients should be managed closely to ensure proper treatment is initiated if necessary.
Case Description
History
An 84-year-old Caucasian male presented to clinic with decreased vision in a red, painful left eye (OS). The patient reported that symptoms began 4 days prior following difficult removal of a rigid gas permeable (RGP) contact lens OS. After unsuccessfully trying to remove the lens, the patient reported sleeping with the lens in the eye for 2 nights. His symptoms began on the third day after he finally succeeded in removing the lens. His symptoms of pain and redness persisted after he removed the contact lens, which prompted his visit to the eye clinic. He denied any additional ocular or head trauma.
Most notably, the patient’s ocular history OS included penetrating wire trauma requiring lens extraction and cryotherapy in 1980, which resulted in aphakia, chorioretinal atrophy and pannus and corneal edema secondary to RGP contact lens overwear. The patient’s ocular history included pseudophakia in the right eye (OD). He had controlled type 2 diabetes mellitus since 1998 with no history of diabetic retinopathy or clinically significant macular edema in either eye.
In addition to type 2 diabetes mellitus, the patient’s medical history was positive for hypertension, hyperlipidemia, chronic kidney disease, hypothyroidism, atrial fibrillation, stroke, nocturnal aortic stenosis, vitamin B12 deficiency, age-associated memory impairment, dysphasia, seizure disorder, obesity, insomnia, osteoarthritis, remission for alcohol dependence and tremor. Medication use included insulin, amlodipine besylate, clonidine, hydrochlorothiazide, metoprolol, rosuvastatin, ezetimibe, levothyroxine, apixaban, omeprazole, aspirin, cyanocobalamin, thiamine, iron, folic acid, levetiracetam and sertraline.
Examination findings
The patient presented with 20/25 visual acuity OD and light perception OS (no improvement with pinhole). His vision was previously correctable to 20/25-2 OS. Consistent with records from the patient’s last eye exam, the pupil OD was round and reactive to light, and the pupil OS was fixed (due to history of trauma). No afferent pupillary defect was present. Confrontational visual field testing results were full to finger counting OD and full to light perception OS. Extraocular motility was full in both eyes (OU), and tonometry (iCare) showed intraocular pressure (IOP) of 14 mmHg OD and 22 mmHg OS.
Slit lamp examination OD was remarkable for 2+ meibomian gland disease. Slit lamp examination OS was remarkable for 2+ meibomian gland disease, trace diffuse bulbar conjunctival injection, and trace bulbar conjunctival edema. The cornea had 3+ diffuse edema with guttata and 3 clock hours of pannus and neovascularization (Figure 1). The anterior chamber had a 1-mm tall hyphema (Figure 2) and anterior chamber reaction of 4+ cells and 1-2 flare. Clear and complete visualization of the iris was challenging due to the hyphema obstructing the inferior iris, 3+ corneal edema and 4+ cells and 1-2 flare anterior chamber reaction, making it difficult to completely rule out iris neovascularization. Posterior structures were also difficult to assess due to the corneal edema, hyphema and anterior chamber reaction. Therefore, a B-scan ultrasound was performed. No masses or retinal detachments were detected (Figure 3). Fundus examination and gonioscopy were performed only OD due to the presence of the hyphema OS. No abnormal vessels in the angles, angle neovascularization or other abnormalities were noted OD.
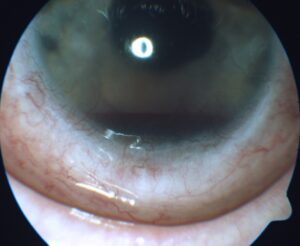 Figure 1. Hyphema, 3+ corneal edema, pannus and corneal neovascularization of the left eye at initial visit. Click to enlarge |
 Figure 2. Hyphema in the left eye present at initial visit. Click to enlarge |
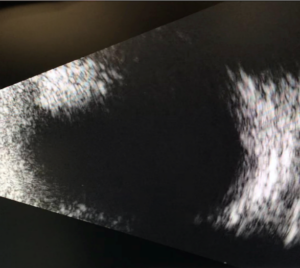 Figure 3. B-scan ultrasound of the left eye on the day of patient’s initial presentation to clinic. Note the absence of retinal detachment or masses. Click to enlarge |
Initial treatment
To quell the anterior chamber reaction, the patient was prescribed 1% prednisolone acetate to be used every hour OS and 1% atropine to be used 3 times per day OS. The patient was instructed to stay on bed rest with head elevation of at least 30 degrees at all times, avoid any type of straining, and wear a protective eye shield full-time OS to promote resolution and avoid re-bleeding of the hyphema. Contact lens wear OS was to be discontinued until further notice.
Out of an abundance of caution, a cornea culture was obtained OS. The patient’s recent blood test results were reviewed. Additional blood tests ― for protein C and protein S, herpes simplex virus (HSV) and sickle cell anemia ― were ordered to rule out systemic diseases with known ocular complications and to gain a better sense of the patient’s systemic health.
Follow-up
The patient was monitored closely during the following weeks, and the hyphema, anterior chamber reaction and corneal edema gradually resolved (Figures 4 and 5). The hyphema cleared 6 days after initial presentation, while the anterior chamber reaction cleared 10 days after initial presentation. When the anterior chamber was free of cells and flare, the topical steroids were tapered to prevent rebound inflammation. The patient went from one drop 1% prednisolone acetate every hour OS (~ 12x/day) to 8x/day for 1 week, 4x/day for 1 week, 3x/day for 1 week, 2x/day for 1 week, 1x/day for 1 week and then discontinuation. The patient returned to clinic at each 1-week interval to be evaluated for rebound inflammation before continuing taper of the steroid. Atropine 1% OS was also tapered when the anterior chamber reaction resolved, decreasing from one drop 3x/day to 2x/day for 1 week to 1x/day for 1 week and then discontinuation. Toward the end of the prednisolone and atropine tapering, approximately 6 weeks after symptom onset, the patient’s best-corrected visual acuity (BCVA) improved to 20/25-2 OS.
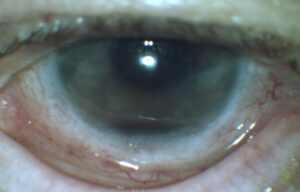 Figure 4. Hyphema in the left eye at the 2-day follow-up visit. Click to enlarge |
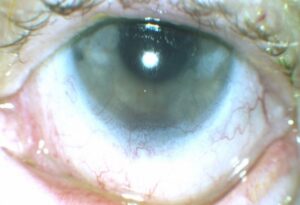 Figure 5. Hyphema in the left eye at the day-3 follow-up visit. Note the steady resolution of the hyphema.Click to enlarge |
Gross fundoscopic view of the posterior chamber was possible approximately 1 week from presentation and revealed a vitreous hemorrhage OS. The hyphema completely resolved 6 days after initial presentation; however, gonioscopy was not performed until 1 month after resolution of the hyphema to prevent triggering a re-bleed. Gonioscopy was performed OS and was open to ciliary body band in all four quadrants. No abnormal vessels in the angles, no angle neovascularization, or other abnormalities were noted.
Upon discovery of the vitreous hemorrhage, the differential diagnosis list expanded to include the following: Swan syndrome, ischemic proliferative diabetic retinopathy, ocular ischemic syndrome, central retinal vein occlusion, traumatic vitreous hemorrhage and spillover hyphema due to RGP lens removal, sickle cell retinopathy, herpetic eye disease and leukemic retinopathy. Although diabetic, sickle cell, herpetic and leukemic retinopathy may present with asymmetry, these conditions most often manifest bilaterally. As a result, these conditions moved lower on the differentials list. A clear view of the retina OS was possible 1 month after initial presentation, and no retinopathy was noted.
Laboratory test results
The cornea culture came back negative for white blood cells, organisms or growth in 72 hours. No abnormal hemoglobin or sickle cell traits were detected. Normal protein S and protein C levels and normal partial thromboplastin time (PTT) suggested a normal coagulation system. HSV-1 and HSV-2 were not detected.
Prothrombin time (PT) and international normalized ratio (INR) were elevated, indicating the patient’s blood takes longer than normal to clot, but this was confounded by his use of anticoagulants aspirin and apixaban. Elevated vitamin B12 ruled out pernicious anemia and was explained by the patient taking vitamin B12 supplements. Interestingly, the patient’s HbA1c rose from 6.3% to 8.7% in 6 months, which suggests poor control of type 2 diabetes.
Treatment and management
Swan syndrome surpassed diabetic retinopathy as a potential diagnosis when a clear view of the fundus was possible (~ 1 month after initial presentation) and no retinal hemorrhages, cotton wool spots or exudates were noted in either eye. The negative cornea culture and lab findings further supported the diagnosis by ruling out the remaining differentials. The diagnosis of Swan syndrome was confirmed after review of the lab results, the patient’s history of ocular trauma and aphakia, and the findings of vitreous hemorrhage and hyphema.
The patient’s hyphema and vitreous hemorrhage both completely resolved in 1 month. This was the first occurrence of hyphema and vitreous hemorrhage in the patient’s history. The plan was to continue to monitor him for re-bleeds or recurrences on a 3- 6-month follow-up schedule. The patient was thoroughly educated regarding cessation of contact lens wear and to report to clinic immediately if any signs or symptoms suggested re-bleeding. If recurrences do occur in this patient, referral for surgical intervention would be considered.
Refractive correction
Once the hyphema, anterior chamber reaction, corneal edema and vitreous hemorrhage resolved and was stable, approximately a month and a half after initial presentation, refraction was performed again. The updated refraction was OD: +1.00-0.25×090 with BCVA 20/25, and OS: +12.25-0.50×108 with BCVA 20/25-2.
The patient had been aphakic for more than 30 years and had a long history of both soft and RGP contact lens wear. In the past, he had worn a single-vision soft contact lens OS but switched to an RGP lens for better clarity and easier handling. His most recent correction consisted of bifocal glasses to correct distance vision OD and near vision OU, worn over an RGP contact lens for distance correction OS.
Due to the patient’s history of soft and RGP lens overwear, poor lid hygiene and concurrent ocular pathology, continued contact lens correction was avoided. Scleral lenses were considered but not a great option due to the diagnosis of Swan syndrome and the possibility of a pinched anastomosed vessel causing a re-bleed. Spectacle correction was therefore the only option, but it presented a unique challenge due to the marked anisometropia resulting from pseudophakia OD and aphakia OS. The patient also expressed a strong preference for a single pair of glasses as opposed to separate pairs for distance and near.
To minimize aniseikonia when placing the spectacle order for this patient, it was important to specify equal base curves and slab-off prism along with 1.74 high-index lens material. A request was made to the lab to place the bevel forward on the higher plus left lens and further back on the less plus right lens in order to manipulate vertex distance and reduce disparity in power factor magnification between the two lenses. In addition, the patient was encouraged to select smaller frames to minimize weight and lens edge thickness. The adjustments to lens, frame and material should mitigate negative effects of aniseikonia for the patient.
Education Guidelines
Learning objectives
At the end of the case discussion, participants should be able to:
- Recognize the key features of Swan syndrome
- List differential diagnoses for hyphema
- List differential diagnoses for vitreous hemorrhage
- Describe the epidemiology, key signs and risk factors associated with Swan syndrome
- Describe the treatment options for Swan syndrome
- Describe the treatment options for patients with aniseikonia due to aphakia
- Properly educate patients on prognosis and long-term management of Swan syndrome
Key concepts
- Identifying Swan syndrome in patients with recurrent hyphemas and/or vitreous hemorrhages
- Differential diagnoses for hyphema
- Differential diagnosis for vitreous hemorrhage
- Treatment options for patients with aniseikonia secondary to aphakia post trauma
- Delivering clear education to the patient regarding diagnosis, treatment and long-term management of Swan syndrome
Discussion points
1. Knowledge, facts and concepts required for critical review of this case
- Describe the epidemiology, pathophysiology and risk factors associated with Swan syndrome
- Describe the key clinical findings in patients with Swan syndrome
2. Differential diagnosis
- Know characteristic signs of Swan syndrome
- List appropriate clinical diagnoses for hyphema
- List appropriate clinical diagnoses for vitreous hemorrhage
3. Disease treatment and management
- What are the benefits of closely monitoring this patient?
- What is the standard of care for treating a patient with Swan syndrome?
- Discuss the most likely prognosis and possible complications following treatment of a patient with Swan syndrome
- What is the appropriate long-term management plan for a patient with Swan syndrome?
4. Patient education and communication
- How would you educate the patient regarding the suspected diagnosis of Swan syndrome?
- How would you educate the patient regarding the hyphema?
- How would you educate the patient regarding the vitreous hemorrhage?
- How would you educate the patient regarding long-term follow-up care and management?
5. Critical thinking
- How would you have managed this case? Justify your answer based on the findings
- What would have been a sign of poor prognosis?
Discussion
Teaching instructions
This case report is intended for optometry students, residents and clinicians with an intermediate knowledge base of ocular anatomy, anterior and posterior segment ocular health, ophthalmic optics, pathophysiology and pharmacology. The case report can be taught in a variety of settings including formal academic classrooms, virtual education platforms or small-group clinical settings. The learning objectives above can be assessed through multiple modalities, including multiple-choice TurningPoint or formal exam questions in an academic setting, multiple-choice pop-up questions in a virtual format, or open-ended discussion questions in small groups.
In a formal academic classroom setting, the case can be presented in full as a traditional PowerPoint lecture with detailed explanations of differential diagnoses, epidemiology, pathophysiology, signs/symptoms, risk factors, treatment and management options. Understanding can be evaluated through open-ended questions to the class or multiple-choice questions posed electronically via TurningPoint or written as an exam. Students can be encouraged to practice critical-thinking skills by suggesting alternative treatment or management of the case.
In a virtual education setting, the case can be presented in full as a traditional PowerPoint presentation with audience participation multiple-choice questions incorporated throughout to gauge comprehension and understanding. Questions would include key concepts such as risk factors, clinical findings, differential diagnoses, testing interpretation and treatment and management options. Students would be evaluated on ability to determine appropriate ancillary testing, testing and laboratory results, treatment and follow-up. Feedback would be provided immediately as detailed explanations of why the correct answers were correct and other answers were incorrect.
In a small-group clinical setting, the case can be discussed in sections to promote independent thinking and decision-making. For example, presenting the case as though the patient just walked into the student’s clinic with a red eye, decreased vision and hyphema, what are the appropriate tests to perform? Students can be encouraged to have an open discussion on how they would have handled the exam and determined appropriate testing, lab work and management of the patient. In addition, discussions could involve a management plan not addressed in the case but supported by other clinical testing and ideas.
Background
The term Swan syndrome refers to intraocular bleeding from abnormal limbal wound vessels secondary to crystalline lens or cataract extraction involving a scleral incision resulting in hyphema and/or vitreous hemorrhage. First described in the literature by Dr. Kenneth Swan in the 1970s, these instances of often-recurrent bleeding occur later in the postoperative period, months to years after surgery.
Diagnosis and clinical findings
Patients with Swan syndrome present with acute onset blurred or blocked vision, pain and blood in the front of the eye. Patients typically report no recent history of conspicuous ocular or head trauma. Clinical signs include blood in the anterior chamber, vitreous or anterior chamber angle.
The key clinical findings in patients with Swan syndrome include aphakia or history of cataract surgery involving a scleral incision, hyphema and vitreous hemorrhage. Gonioscopic examination of multiple patients with shared clinical history and presentation often exhibited focal areas of abnormal, leaky blood vessels near surgical incision sites, but these abnormal vessels may not be seen on gonioscopic examination of every patient. Postmortem histological analysis of eyes has revealed episcleral vessels extending deep into stromal wounds and terminating as fragile, leaky capillaries.1
A variety of triggers of bleeding episodes in cases of Swan syndrome have been reported, such as riding on a boat2 or vomiting.3 In some cases, there has been no apparent trigger.2,4 In the case of the patient in this case report, bleeding likely occurred due to the minor trauma of a difficult RGP lens removal due to poor dexterity.
Differential diagnosis
Swan syndrome is a diagnosis of exclusion; therefore, more common known causes of hyphema and vitreous hemorrhage must be ruled out with thorough patient history, clinical examination and blood testing.
A positive history of crystalline lens extraction is a key element in making the diagnosis of Swan syndrome. It is also important to ensure that the patient has not undergone a recent significant major trauma, such as a blow to the head or globe. Careful fundus examination is necessary to confirm the absence of retinal breaks, retinal neovascularization and other significant retinopathy. Laboratory blood tests should be ordered to rule out the various hematologic disorders mentioned below.
The differential diagnoses of hyphema include trauma, uveitis-glaucoma-hyphema syndrome, juvenile xanthogranuloma, leukemia, child abuse, lens-extraction surgery sequela, Fuch’s heterochromic iridocyclitis, sickle cell disease and rubeosis irides.5,6
The differential diagnoses of vitreous hemorrhage include retinal break, trauma, retinal neovascularization, posterior vitreous detachment, ruptured microaneurysm, retinopathy of blood disorders, Valsalva retinopathy and Terson syndrome.5
Management and treatment
Patients with hyphema and vitreous hemorrhage, especially those with recurrences, must be made aware that it is critical to determine underlying etiology so appropriate follow-up care and treatment can be recommended. They should also be educated about side effects and potential risk factors for recurrence. Patients should expect to have thorough ocular health examinations, lab tests and frequent follow-up visits initially to monitor for changes and improvement of hyphema and vitreous hemorrhage.
Once a diagnosis of Swan syndrome is made, patients must be thoroughly educated about the potential for recurrences of hyphema and/or vitreous hemorrhage, which can be exacerbated by trauma or other factors, including those that contributed to the initial episode. Patients should know to seek care promptly for eye pain, redness or acute vision loss. Each patient should be made aware of the causative abnormal limbal wound vascularization, as well as treatment options for both acute and recurrent episodes of hyphema and vitreous hemorrhage.
A variety of treatments have been applied to patients with Swan syndrome. Goniophotocoagulation with argon laser has been performed in some cases with varying degrees of success. For example, some patients did not experience a re-bleed during follow-up periods ranging from 36 months to 5 years following a single photocoagulation treatment, but others required goniophotocoagulation re-treatments when vitreous hemorrhage or hyphema recurred.2,3 Other approaches, including re-opening of the surgical incision to perform cryopexy or diathermy on the leaky blood vessels or direct surgical excision of wound blood vessels, have been reported in the literature as well.1,7 Efforts to suture across the limbal incision site to close any remaining wound gap have also been attempted, but may introduce a large amount of astigmatism.4
Ultimately, multiple treatment methods may be required on a case-by-case basis. For instance, Chen and Kwon described a case of Swan syndrome in which the patient did not show a reduction in bleeding episodes after gonioscopic laser photocoagulation. Their next approach was to re-open the surgical incision so that limbal wound vessels could be cauterized. The patient continued to have frequent hemorrhages, however, and sutures were placed across the incision site to increase wound apposition. This reduced the frequency of the patient’s hemorrhages enough to warrant careful observation without further surgical intervention.4
Aside from direct treatment of wound neovascularization, hyphema and vitreous hemorrhage must also be managed in Swan syndrome patients. Patients with hyphema should be instructed to wear a protective eye shield, sleep with their head elevated at least 30 degrees and, if possible, avoid blood thinners until the hyphema resolves. Topical steroids and cycloplegic agents should be used to eliminate any accompanying anterior chamber reaction and stabilize the blood-aqueous barrier. More invasive clinical examination techniques such as gonioscopy and scleral depression should be avoided until at least 1 month after resolution to prevent a re-bleed.
If IOP is elevated due to hyphema or topical steroid use, IOP-lowering agents may be used. Pilocarpine and prostaglandin analogues are contraindicated due to their pro-inflammatory nature and destabilization of the blood-aqueous barrier.8 Additionally, patients with underlying sickle cell disease should not be treated with acetazolamide or other carbonic anhydrase inhibitors to lower IOP. These agents may promote sickling of red blood cells in the anterior chamber, potentially obstructing trabecular aqueous humor outflow, because they increase ascorbate concentration and lower the pH of the aqueous humor.6
The prognosis for vision recovery following hyphema is generally good with approximately 76% of patients achieving recovery of 20/50 or better (if not a total hyphema).9 However, patients must be monitored for future angle recession (if traumatic etiology) and red-cell/ghost-cell or uveitic glaucoma.5
As in hyphema management, instructing the patient to maintain head elevation, rest in bed and avoid anticoagulants when possible may promote settling and resolution of the vitreous hemorrhage. Pars plana vitrectomy should be considered for idiopathic vitreous hemorrhage persisting more than 6 months, a non-clearing hemorrhage in a diabetic patient lasting longer than 1 month, and in other situations involving an intractable elevation of IOP, reduced visual acuity in the fellow eye, or when a retinal break or detachment is suspected.5
Finally, protective eyewear with durable frames and impact-resistant lens material, such as polycarbonate, should be strongly recommended to patients with Swan syndrome to protect their eyes from trauma that may trigger re-bleeding.
Significant anisometropia with resultant aniseikonia complicates quality of vision in any patient. Patients with sufficient ocular surface health and gentle dexterity will likely get better reduction in aniseikonia symptoms with contact lens correction than with spectacle correction. This is because contact lenses have a small center thickness and vertex distance, which reduce differences in shape and power factors of magnification, respectively. However, for patients with symptomatic aniseikonia for whom contact lenses are not an appropriate option, strategic manipulation of the base curve, center thickness and vertex distance of each spectacle lens can help reduce the difference in magnification between the two lenses. This principle is known as iseikonic lens design.10 Additional treatment options for aniseikonia include refractive surgery, clear lens exchange, secondary intraocular lens placement or occlusion.11
Fortunately, surgical incision wound vascularization with subsequent hyphema and/or vitreous hemorrhage has not been reported in eyes that have undergone more modern cataract surgical techniques involving clear corneal incisions.3
Conclusion
The patient in this case report was diagnosed with Swan syndrome based on his ocular history of trauma resulting in aphakia, and exam findings of hyphema and vitreous hemorrhage with no other significant concurrent pathology. The case not only demonstrates a rare condition, but it also highlights the importance of pursuing multiple differential diagnoses to arrive at a clear diagnosis. Patients with suspected Swan syndrome should be monitored closely not only during initial diagnosis but routinely after resolution of hyphema and/or vitreous hemorrhage to determine whether additional treatment is necessary.
References
- Swan KC. Hyphema due to wound vascularization after cataract extraction. Arch Ophthalmol. 1973 Feb;89(2):87-90. doi: 10.1001/archopht.1973.01000040089001.
- Bene C, Hutchins R, Kranias G. Cataract wound neovascularization. An often overlooked cause of vitreous hemorrhage. Ophthalmology. 1989 Jan;96(1):50-3.
- Banitt MR, Rand A, Vann RR, Gedde SJ. Bilateral intraocular hemorrhage from vascularization of cataract wounds. J Cataract Refract Surg. 2009 Sep;35(9):1637-9. doi: 10.1016/j.jcrs.2009.05.007.
- Chen JJ, Kwon YH. Recurrent intraocular hemorrhage secondary to cataract wound neovascularization (Swan syndrome) [Internet]. Swan syndrome. University of Iowa Ophthalmology Grand Rounds. University of Iowa Carver College of Medicine Department of Ophthalmology & Visual Sciences; 2012 [cited 2021 Jun 28]. Available from: http://eyerounds.org/cases/153-Swan-syndrome.htm.
- Friedman NJ, Kaiser PK, Pineda R. The Massachusetts Eye and Ear Infirmary Illustrated Manual of Ophthalmology. 5th ed. Edinburgh: Elsevier; 2021.
- Demeritt MJ, Shectman DL, Reynolds SA. Spotlight on sickle cell retinopathy [Internet]. Review of Optometry. Jobson Medical Information LLC; 2014 [cited 2021 Jun 24]. Available from: https://www.reviewofoptometry.com/article/spotlight-on-sickle-cell-retinopathy.
- Watzke RC. Intraocular hemorrhage from wound vascularization following cataract surgery. Trans Am Ophthalmol Soc. 1974;72:242-52.
- Lee DA, Higginbotham EJ. Glaucoma and its treatment: a review. Am J Health Syst Pharm. 2005 Apr 1;62(7):691-9. doi: 10.1093/ajhp/62.7.691.
- Walton W, Von Hagen S, Grigorian R, Zarbin M. Management of traumatic hyphema. Surv Ophthalmol. 2002 Jul-Aug;47(4):297-334. doi: 10.1016/s0039-6257(02)00317-x.
- McNeill S, Bobier WR. The correction of static and dynamic aniseikonia with spectacles and contact lenses. Clin Exp Optom. 2017 Nov;100(6):732-734. doi: 10.1111/cxo.12516.
- Stokkermans TJ, Day SH. Aniseikonia. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK585108/.



