PEER REVIEWED
Hyperopic Shift Following Rapid Glycemic Control in a Diabetic Adult
Brittney M. Brady, OD, FAAO
Abstract
Diabetes mellitus, the most common endocrine disease in the United States, is known to cause ocular complications, including retinopathy, macular edema, cataracts and refractive error changes, amongst other problems. Myopic shifts in diabetic patients are more common, resulting from lenticular changes or macular edema following poor glycemic control. Less commonly, hyperopic shifts may present following aggressive glycemic control. The purpose of this case report is to increase provider awareness and explore the pathophysiology behind hyperopic refractive shifts associated with diabetes. This article offers a comprehensive review of the etiology, clinical features and management of hyperopic shifts in diabetic patients.
Keywords
Background
Diabetes mellitus is a complex endocrine disease that can result in various ocular complications such as retinopathy, macular edema, ischemic optic neuropathy, ophthalmoplegia, cataracts, amongst other problems.1,2 Elevated blood glucose is the most common predictor of ocular complications while hemoglobin A1C levels provide the most reliable insight into long-term glycemic control.1 Transient bilateral refractive error changes are a well-documented phenomenon in diabetic patients.3–5 Myopic shifts secondary to diabetes are more commonly recognized by practitioners and result from poor glycemic control leading to lenticular swelling. Less commonly, hyperopic refractive changes may be seen and are typically associated with rapid glycemic control leading to a decreased lens thickness and lenticular refractive index.1,6
Case Description
A 50-year-old Hispanic male presented with complaints of painless decreased vision for the past three weeks in both eyes through his 2-year-old habitual correction. He reported being diagnosed with type 2 diabetes mellitus and acute kidney failure around the time of symptom onset. At the time of diagnosis, he had significantly elevated blood glucose, roughly 1200 mg/dL, for which he was immediately started on insulin in the emergency room. The patient did not know his hemoglobin A1c but reported his most recent blood glucose to be 117 mg/dL 6 days prior with daily use of insulin lispro 25/75, unknown dosage. His ocular history was remarkable for presbyopia, and his systemic history was unremarkable outside of the newly diagnosed diabetes. He denied the use of any other medications or relevant family medical history.

Table 1. Click to enlarge
Entering corrected visual acuity at distance was 20/100 in both eyes with a pinhole acuity of 20/50+2 in the right eye and 20/50-2 in the left eye. Entrance testing including pupils, confrontation visual fields, and motilities were unremarkable. Manifest refraction revealed a 3.00 and 3.50 diopter sphere hyperopic shift in the right eye and left eyes respectively (Table 1) which improved the vision to 20/20 in both eyes. The intraocular pressure was found to be 18 mmHg in both eyes via Goldmann applanation tonometry and the blood pressure was measured at 140/87 mmHg. Anterior segment exam including cornea, anterior chamber, iris, and intraocular lens was unremarkable. The dilated fundus examination was unremarkable without signs of diabetic retinopathy or macular edema. An optical coherence tomography of the macula was unremarkable and confirmed the absence of diabetic macular edema in either eye (Figure 1).
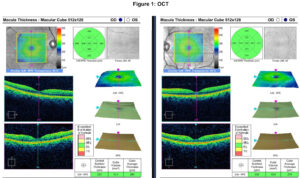
Figure 1. Click to enlarge
Drug induced hyperopia was considered, although the patient denied any use of medications outside of his recently prescribed insulin. Given the unremarkable anterior and posterior segment exams with recent aggressive treatment of significant hyperglycemia, the patient was diagnosed with a hyperopic shift secondary to diabetic induced lenticular changes. A follow-up in 1 month was advised to monitor the status of the refractive error.
Follow-up exam 1
The patient returned for follow-up 1 month later noting only slight improvement in vision since last exam. He had been using +3.00 diopter sphere over-the-counter spectacles for distance and near with some success. He reported good compliance with his insulin lispro, unknown dosage. His most recent blood sugar was reported as 144 mg/dL and his HbA1c was unknown. The entering corrected visual acuity at distance was 20/70-2 in both eyes with a pinhole acuity of 20/60+1 in the right eye and 20/60+2 in the left eye. Entrance testing including pupils, confrontation visual fields, and motilities were unremarkable. Manifest refraction revealed roughly 1 diopter less hyperopia since the previous exam which improved the vision to 20/20 in both eyes (Table 1). The anterior segment exam and undilated fundus examinations were stable and unremarkable without signs of diabetic retinopathy or macular edema. The patient was advised to follow up in 6 weeks to allow the refractive error additional time to stabilize.
Follow-up exam 2
The patient was lost to follow-up for 3.5 months. Upon presentation, he reported his vision has seemed to stabilize, although he still noted blur with his habitual spectacles. The patient was unable to provide his HbA1c or fasting blood sugar but noted a recent improvement in his diet and exercise. The entering corrected visual acuity at distance was 20/70 in both eyes. Entrance testing including pupils, confrontation visual fields, and motilities was unremarkable. Manifest refraction revealed 1 diopter less hyperopia OD and 1.75 diopters less hyperopia OS since the previous exam (Table 1). The anterior segment exam and undilated fundus examinations were unremarkable without signs of diabetic retinopathy or macular edema. An optical coherence tomography of the macula confirmed the absence of macular edema in either eye and was stable to previous testing (Figure 2). A final spectacle prescription was released, and the patient was strongly advised to follow up with his physician for further management of his diabetes.
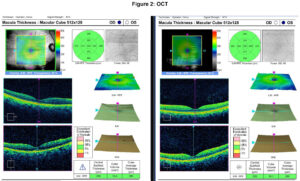
Figure 2. Click to enlarge
Education Guidelines
Learning Objectives
At the end of this case discussion, participants should be able to:
- Recognize the potential refractive error changes associated with diabetes mellitus
- List differential diagnoses of a hyperopic refractive error shift
- Describe the pathophysiology of a hyperopic refractive error shift in a diabetic patient
- Describe the treatment recommendations for refractive error shifts in diabetic patients
Key Concepts
- Refractive error changes secondary to diabetes mellitus
- Hyperopic shifts in the presence of diabetes mellitus typically follow rapid glycemic control
- Changes in lenticular refractive index and lenticular thickness are the primary mechanisms for refractive error changes associated with diabetes
- Recovery of baseline refractive error can take up to 11 weeks
Discussion Questions
- What is the most common refractive error change associated with diabetes mellitus?
- What causes a transient hyperopic shift following rapid glycemic control in diabetic patients?
- What ocular structure is the main driver of hyperopic shifts in diabetic patients?
- How long should practitioners wait before releasing a spectacle prescription following diabetic refractive error shifts?
Teaching Instructions and Learning Assessment
This case report is intended for optometry students, residents, and clinicians who have an understanding of ocular anatomy, optics, refractive error, diabetes mellitus and its impact on the ocular structures. This case has the potential to be taught in a variety of settings including formal academic classrooms, virtual education platforms, and small-group settings such as journal clubs or case discussions. Assessment of learning objectives may be instituted using various modalities including formal multiple-choice exam questions, open-ended discussion questions, or informal pop-quiz style questions in which feedback on tested concepts could be provided immediately following a correct or incorrect answer.
Discussion
Refractive Error
Hyperopia occurs when light from a distant target focuses behind the retina. Hyperopic shifts can present anytime the retina is shifted anteriorly or if axial length is reduced as in cases of retrobulbar or choroidal tumors, macular edema, or central serous chorioretinopathy.2,7–10 Additionally, hyperopia can be induced following a decrease in ocular refractive power as in cases of corneal flattening, cataracts, posterior lens displacement, poor accommodation as seen in Adie’s syndrome, or following silicone oil injection.7,11,12 The list of anticholinergic drugs or drugs with anticholinergic properties that may induce a hyperopic shift are expansive. Shifts have been reported with cycloplegics, benzodiazepines, chloroquine, and antihistamines. Anticholinergics, such as dicyclomine and hyoscyamine, which are used in the management of inflammatory bowel disease, may also result in hyperopic shifts. Other anticholinergics drugs include antispasmodics, antimuscarinics, antidepressants, antipsychotics, antiemetics, muscle relaxants, antiarrhythmics, and muscle relaxants.2,8,13 Myopia is just the opposite and occurs when light from a distant target focuses in front of the retina. Therefore, myopia may be induced following axial elongation, as seen in congenital glaucoma, degenerative myopia, scleral buckles, or staphylomas, or with increased ocular refractive index, corneal steepening or increased lenticular refractive power.14
Transient refractive error changes in diabetic patients are a known occurrence with the term “Diabetic Myopia” first coined by Duke-Eder in 1925.15 These changes can present acutely and be transient with changes to glucose levels or be sustained despite normalization of glucose levels.1,16 Diabetic myopic shifts typically develop slowly and progressively, correlating with the gradual rise in blood sugar levels that occurs with the onset or poor management of the condition. Alternatively, hyperopic shifts typically occur more abruptly and with greater magnitude, often resulting from a rapid and substantial drop in blood sugar due to aggressive, fast-acting treatment.1, 6, 10, 15 The refractive power of the eye is dependent on the axial length, anterior and posterior curvature of both the cornea and lens, the anterior chamber depth, lens thickness, and refractive indices of the cornea, lens, aqueous humor, and vitreous.1,6,10 To understand the refractive changes associated with diabetes, the glycemic impact on these structures must be considered.
Anterior Segment
The tear film has the potential to be greatly impacted by diabetes resulting in lowered tear secretion, tear film instability, reduced goblet cell density, and decreased corneal sensitivity. Such changes are thought to be caused by reduced innervation to the lacrimal gland and subsequent conjunctival squamous cell metaplasia.1,17 The cornea plays an important role in the refractive abilities of the eye, contributing two-thirds of the overall refractive power. Diabetes can impact all corneal structures resulting in metabolic, morphological, and clinical changes such as weakened epithelial and endothelial structures, increased susceptibility to damage or trauma, reduced sensitivity, and delayed wound healing. Additionally, hyperglycemia can inhibit endothelial pumps resulting in corneal hydration and subsequent increased corneal thickness.17 It has been reported that intraocular pressure is higher in diabetic eyes and that anterior chamber depth decreases as severity of retinopathy increases, likely due to increased lenticular thickness. However, a study by Larsson et. al. found no clinically significant changes to aqueous humor dynamics associated with diabetes.18 Patients with diabetes may experience premature vitreous liquefaction due to increased glycation and abnormal crosslinking of collagen fibrils. Furthermore, angiogenic and metabolic vitreous changes observed in diabetic patients mimic retinal microvascular abnormalities associated with diabetic retinopathy leading to increased risk of proliferative disease and vitreous hemorrhage.17,19
The refractive power of the crystalline lens is dependent on the lens thickness, curvature, refractive index, and osmotic gradient across the lens capsule. A decrease in the refractive index of the lens results in light undergoing less refraction and induces a hyperopic shift. Alternately, an increase in refractive index of the lens results in light undergoing more refraction and in turn causes a myopic shift.2,4 In hyperglycemic states, glucose is converted to sorbitol which accumulates in the crystalline lens due to its poor membrane permeability. Rapid shifts from hyperglycemia to hypoglycemia lead to an osmotic pressure imbalance and an influx of water from the aqueous humor into the lens and a subsequent hyperopic shift.5,10 A prospective study by Okamoto et. al. found a hyperopic shift in diabetic patients after intensive glycemic control without observed change to lens thickness, corneal or lens curvature, axial length, or anterior chamber depth suggesting a change to lenticular refractive index as the cause for refractive error changes.10 These results were supported with a prospective study by Tai et. al. which concluded hyperopic shifts associated with diabetic treatments result from a decrease in the lenticular refractive index as well as by an experimental study which reported a 3.2 diopter hyperopic shift following a 0.02 decrease in lenticular refractive index.20,21 Such changes have been shown to be transient and normalize within 14-84 days with a strong correlation between maximum hyperopic shift and recovery time.10 Research has shown that myopic shifts associated with hyperglycemia occur following increased lens thickness from decreased ciliary zonule tension. Not surprisingly, the opposite is true for hyperopic changes that occur following normalization of glucose levels due to increased zonular tension resulting in decreased lens thickness.22 Further evidence that a change in refraction associated with blood glucose levels are primarily related to lens changes was demonstrated by Gwinup and Villarreal. In this study, diabetic patients were given 50 milliliters of 50 percent glucose and the changes in refraction observed over a 90-minute period after which phakic patients were observed to undergo myopic changes while aphakic patients showed only slight hyperopic changes.23
Differential Diagnosis of Hyperopic Shifts in Diabetic Patients
- Flattening of the corneal curvature
- Trauma
- Refractive Surgery
- Adie’s Tonic Pupil
- Posterior dislocation of the intraocular lens
- Cortical cataract
- Drug induced hyperopia
- Chloroquine
- Benzodiazepines
- Antihistamines
- Cycloplegics
- Anterior dislocation of the retina
- Macular edema
- Central serous chorioretinopathy
- Choroidal Tumors
- Decreased axial length
- External globe compression by a mass
- Intraocular silicone oil
Treatment
Diabetic patients presenting with acute bilateral myopic or hyperopic refractive shifts must undergo a thorough ocular examination to rule out a pathological cause. Ancillary testing including corneal topography, axial length, posterior segment optical coherence tomography, or B-scan ultrasonography can provide useful information regarding potential anatomical changes to account for the hyperopic shift. If all anatomical findings are normal, updated A1c or fasting blood glucose measurements should be obtained. Updated spectacle prescriptions should not be released immediately in such patients. It has been documented that it can take 4 to 12 days for onset of hyperopia following intensive glycemic control with recovery of the initial refraction taking an average of 64 days. Therefore, practitioners should use caution when releasing a spectacle prescription for the first 8 to 10 weeks and consider options for interim management of the stabilizing refractive error.6 Diagnostic disposable soft contact lenses are an efficient, affordable option for temporary refractive correction. For patients unable to wear contact lenses, in-office optical labs may be used to provide quick turnaround lenses manufactured using similar powered stock lenses to allow sufficient visual acuity between visits. Patients should be followed regularly as lens power changes may be necessary as the refractive error stabilizes.6
Conclusion
Myopic changes associated with diabetes are more readily and more frequently recognized by practitioners. However, a clear description of hyperopic changes following intensive glycemic control has been documented with decreasing lenticular refractive index as the predominant factor. This phenomenon must be considered as a differential by students, residents, and clinicians identifying such refractive changes in their clinical care of diabetic patients. Additionally, practitioners should monitor the refraction and not release spectacle prescriptions for at least 8 weeks to allow for as much normalization as possible to occur.
Acknowledgements
Raman Bhakhri, OD, FAAO
Nancy Wong, OD, PhD, FAAO
References
- Kaštelan S, Gverović-Antunica A, Pelčić G, Gotovac M, Marković I, Kasun B. Refractive Changes Associated with Diabetes Mellitus. Semin Ophthalmol. 2018;33(7-8):838-845. DOI: 10.1080/08820538.2018.1519582
- Diel R, Stiff H, Kwon Y, Haugsdal J. Refractive changes in diabetes: not always what meets the eye [Internet]. Iowa City, IA: EyeRounds.org; 2020 [cited 2024 Mar 18]. Available from: https://eyerounds.org/cases/295-refractive-changes-in-diabetes.htm
- Klein BEK, Lee KE, Klein R. Refraction in adults with diabetes. Arch Ophthalmol. 2011 Jan;129(1):56-62. DOI: 10.1001/archophthalmol.2010.322
- Jain S, Gupta CP. Relationship between Refractive Error and Diabetes Mellitus. Journal of Mahatma Gandhi University of Medical Sciences and Technology. 2018;3(2). DOI:10.5005/jp-journals-10057-0074
- Li HY, Luo GC, Guo J, Liang Z. Effects of glycemic control on refraction in diabetic patients. Int J Ophthalmol. 2010;3(2):158-60. DOI: 10.3980/j.issn.2222-3959.2010.02.15
- Lin SF, Lin PK, Chang FL, Tsai RK. Transient hyperopia after intensive treatment of hyperglycemia in newly diagnosed diabetes. Ophthalmologica. 2009;223(1). DOI: 10.1159/000173714
- Majumdar S, Tripathy K. Hyperopia [Internet]. Treasure Island, FL: StatPearls Publishing; c2023 [cited 2024 Mar 18]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560716/
- Moore BD, Augsburger AR, Ciner EB, Cockrell DA, Fern KD, Harb E. Care of the Patient with Hyperopia. Optometric Clinical Practice Guideline. St. Louis, MO: American Optometric Association; 1997.
- Gupta A, Tripathy K. Central Serous Chorioretinopathy Continuing Education Activity [Internet]. Treasure Island, FL: StatPearls Publishing; c2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK558973/?report=printable
- Okamoto F, Sone H, Nonoyama T, Hommura S. Refractive changes in diabetic patients during intensive glycaemic control. Br J Ophthalmol. 2000 Oct;84(10):1097-102. DOI: 10.1136/bjo.84.10.1097
- Thompson J, Whitford S. Take Your Eye off the Ball, Gedde, SJ, editor [Internet]. San Francisco, CA; c2013 Oct [cited 2025 Feb 25]. p. 45–6. Available from: https://www.aao.org/eyenet/article/morning-rounds-take-your-eye-off-ball
- Sarao MS, Elnahry AG, Sharma S. Adie Syndrome [Internet]. Treasure Island, FL: StatPearls Publishing; c2023 Jul. Available from: https://www.ncbi.nlm.nih.gov/books/NBK531471/?report=printable
- Vanegas-Arroyave N, Caroff SN, Citrome L, Crasta J, McIntyre RS, Meyer JM, et al. An Evidence-Based Update on Anticholinergic Use for Drug-Induced Movement Disorders. CNS Drugs. 2024 Apr;38(4):239-254. DOI: 10.1007/s40263-024-01078-z
- Subudhi P, Agarwal P. Myopia [Internet]. Treasure Island, FL: StatPearls Publishing; c2023 Mar. Available from: https://www.ncbi.nlm.nih.gov/books/NBK580529/?report=printable
- Duke-Elder WS. Changes in Refraction in Diabetes Mellitus. Br J Ophthalmol. 1925 Apr;9(4). DOI: 10.1136/bjo.9.4.167
- Yarbağ A, Yazar H, Akdoğan M, Pekgör A, Kaleli S. Refractive errors in patients with newly diagnosed diabetes mellitus. Pak J Med Sci. 2015 Nov-Dec;31(6)1481-4. DOI: 10.12669/pjms.316.8204
- Calvo-Maroto AM, Perez-Cambrodí RJ, Albarán-Diego C, Pons A, Cerviño A. Optical quality of the diabetic eye: A review. Vol. 28, Eye (Lond). 2014 Nov;28(11):1271-80. DOI: 10.1038/eye.2014.176
- Larsson LI, Pach JM, Brubaker RF. Aqueous humor dynamics in patients with diabetes mellitus. Am J Ophthalmol. 1995 Sep;120(3):362-7. DOI: 10.1016/s0002-9394(14)72166-x
- Sebag J. Abnormalities of human vitreous structure in diabetes. Graefes Arch Clin Exp Ophthalmol. 1993 May;231(5):257-60. DOI: 10.1007/BF00919101
- Tai MC, Lin SY, Chen JT, Liang CM, Chou PI, Lu DW. Sweet hyperopia: Refractive changes in acute hyperglycemia. Eur J Ophthalmol. 2006 Sep-Oct;16(5):663-6. DOI: 10.1177/112067210601600501
- Planten JT. Changes of refraction in the adult eye due to changing refractive indices of the layers of the lens. Ophthalmologica. 1981;183(2):86-90. DOI: 10.1159/000309145
- Furushima M, Imaizumi M, Nakatsuka K. Changes in refraction caused by induction of acute hyperglycemia in healthy volunteers. Jpn J Ophthalmol. 1999;43(5):398-403. DOI: 10.1016/s0021-5155(99)00098-2
- Gwinup G, Villarreal A. Relationship of serum glucose concentration to changes in refraction. Diabetes. 1976 Jan;25(1):29-31. DOI: 10.2337/diab.25.1.29



