PEER REVIEWED
Adult-onset Foveomacular Vitelliform Dystrophy
Deepak Sharma, OD, FAAO, and Jonathan Hamilton, OD, FAAO
Abstract
Adult-onset foveomacular vitelliform dystrophy is a macular dystrophy that can present, typically in the fourth to sixth decades of life, with little to no symptoms. It presents with bilateral, asymmetric foveal or perifoveal subretinal lesions that are yellow, round and mildly elevated. Potential complications include choroidal neovascularization (CNV) and geographic atrophy. Ancillary ocular imaging is essential in aiding confirmation of the diagnosis. Treatment is based on whether CNV and subretinal fluid develop. Treatment may include anti-vascular-endothelial-growth-factor injections to help preserve remaining vision. This paper presents a case of adult-onset vitelliform dystrophy found incidentally after recent ocular trauma.
Key Words: adult-onset foveomacular vitelliform dystrophy, Best disease, age-related macular degeneration, anti-vascular-endothelial-growth-factor injections
Background
Adult-onset foveomacular vitelliform dystrophy (AOFVD) is one of the five autosomal pattern dystrophies.1 It typically presents in the fourth to sixth decades of life and is characterized by bilateral, asymmetric, solitary, round to oval subretinal lesions in the fovea or perifovea on fundoscopic evaluation.1,2 It can initially present asymptomatically or with mild blurred vision.1,2 Potential visual complications include choroidal neovascularization (CNV), geographic atrophy and outer retinal atrophy.3 Ancillary imaging modalities such as optical coherence tomography (OCT), fundus autofluorescence (FAF), indocyanine green angiography (ICGA) and fluorescein angiography (FA) are useful in confirming the diagnosis.2 Treatment and management depend on whether CNV is present and compromising central visual acuity and can include anti-vascular-endothelial-growth-factor (anti-VEGF) injections.1
This report describes the case of a patient who presented with symptoms of mild metamorphopsia and a history of previously diagnosed dry age-related macular degeneration (AMD) with subsequent discovery of a presumed AOFVD. Detection of AOFVD would allow a more proper treatment and management protocol and potentially reduce the need for unnecessary treatment burden with the correct diagnosis. For third- and fourth-year optometry students, optometry residents and practicing optometrists, this case report highlights the importance of exploring alternative diagnoses to AMD when presented with abnormal macular findings. It also explains ancillary testing used to confirm the diagnosis and treatment approach.
Case Description
A 69-year-old male presented with a chief complaint of blurred and wavy vision in his right eye (OD) after being scratched by his dog 3 days earlier. The patient had a pertinent medical history of hypertension and hyperlipidemia. Medications included simvastatin for the hyperlipidemia and an anti-hypertensive medication the name of which the patient could not recall. The patient had an ocular history of dry AMD in both eyes (OU), mild cataracts OU, a large chorioretinal scar OD and prior radial keratotomy for refractive correction OU. He was last seen 7 months prior at his annual eye exam. No family history of macular degeneration, glaucoma or other eye-related disease was reported. Social history was positive for alcohol use socially but negative for tobacco and recreational drug use.
The patient reported that he was experiencing blurred, wavy vision with or without spectacle correction, which did not change upon blinking and began 3 days prior OD only. He attributed the blurred and wavy vision to being scratched by his dog OD, which occurred the same day the vision problems began. The patient initially noted ocular pain and discomfort OD only; however, he noted that the ocular pain and redness had subsided by the time he came in for the visit. Entering visual acuity with his habitual spectacle prescription was 20/70 OD and 20/50 left eye (OS) with improvement on pinhole to 20/40+1 OD and 20/30 OS. His visual acuity 7 months prior was best-corrected to 20/30 OD and 20/25 OS, which was attributed to the dry AMD and cataracts. Confrontation visual fields were full to finger counting OU. Pupils were equal, round and reactive to light without an afferent pupillary defect. Extraocular muscle motilities were full without restriction, diplopia or pain OU. The patient reported metamorphopsia OD only on the Amsler grid. Anterior segment evaluation revealed normal appearance of the eyelids, lashes and conjunctiva OU. Both corneas were clear without any sign of acute corneal abnormality, including no abrasion and no infiltrate with or without fluorescein staining. Stable corneal scars from the previous radial keratotomy refractive surgery were noted. Intraocular pressure measured with Goldmann applanation tonometry was stable at 21 mmHg OD and 19 mmHg OS at 4:33 p.m.
Given the lack of anterior segment findings correlating with the reported eye scratch and decreased vision, the pupils were dilated for further assessment. On dilated fundus exam, the cup to disc ratio measured 0.35 horizontally and vertically, and the optic nerves were pink and healthy without disc edema OU. In the macula OD, a large central yellow deposit and a separate yellow deposit inferior temporal to the optic nerve were noted (Figure 1A). OS, a small central yellow deposit in the macula and multiple yellow deposits in a 360-degree ring-like formation were noted (Figure 1B). There were no appreciable clinical signs of subretinal fluid or hemorrhages on funduscopic exam. The retinal periphery was intact 360 degrees without retinal holes, breaks or detachments OU. A presumed stable inactive toxoplasmosis scar was noted nasally OD.
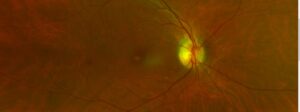 Figure 1A. Fundus image of the right eye documenting yellow deposits, including a central deposit, in the macular region. |
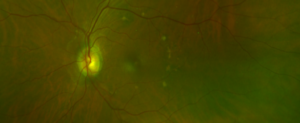 Figure 1B. Fundus image of the left eye documenting yellow deposits in the macular region. Note the ring-like appearance of some of the deposits. Click to enlarge |
Due to the slight decrease in vision along with newly noted metamorphopsia, OCT of the macula was performed the same day. The scans revealed bilateral large subretinal deposits centrally and a slightly attenuated photoreceptor integrity line and no evidence of intraretinal or subretinal fluid (Figures 2A, 2B and 3).
Given the patient’s decreased vision and onset of metamorphopsia without any other correlating ocular or retinal abnormality, the cause was presumed to be related to the progression of subretinal deposits. Given the OCT results and lack of drusen-like findings or family history, the presumptive diagnosis was bilateral AOFVD. The patient was referred to a local retina specialist for further evaluation, including possible FA and/or FAF, and was given an Amsler grid for home monitoring. He has not yet gone to see the retina specialist.
 Figure 2A. Five-line raster macular OCT scan of the right eye revealing large subretinal deposits centrally with a slightly attenuated photoreceptor integrity line and no evidence of intraretinal or subretinal fluid. Click to enlarge |
 Figure 2B. Five-line raster macular OCT scan of the left eye revealing large subretinal deposits centrally with a slightly attenuated photoreceptor integrity line and no evidence of intraretinal or subretinal fluid. Click to enlarge |
 Figure 3. Macular cube OCT scan of both eyes revealing large subretinal deposits centrally with a slightly attenuated photoreceptor integrity line and no evidence of intraretinal or subretinal fluid. |
Education Guidelines
Learning objectives
- Understand pertinent differential diagnoses of AOFVD
- Know signs and symptoms of AOFVD
- Describe useful diagnostic testing for AOFVD
- Understand treatment and management of AOFVD
- Understand the different types of pattern dystrophies
Key concepts
- Ocular signs and symptoms of AOFVD
- Diagnostic testing for AOFVD
- Treatment and management of AOFVD
Discussion questions
1. Critical knowledge and background information for critical review of this case
- What are the ocular signs and symptoms of AOFVD?
2. Differential diagnosis
- What are key differentials for AOFVD?
- How does AOFVD differ from Best disease?
3. Diagnostic testing
- Describe useful diagnostic testing for aiding diagnosis of AOFVD and expected findings
4. Treatment and management
- What treatment options are available for management of AOFVD?
Discussion
Teaching instructions
All participants should read each question and consider how they would respond before reading the appropriate text. This can be done individually or in small groups to facilitate critical thinking on each question. Assessments can be done based on participants’ responses.
What are the ocular signs and symptoms of adult-onset vitelliform dystrophy?
AOFVD is an uncommon macular disease that generally presents in the fourth to sixth decades of life.1,5 It is part of the autosomal dominant pattern dystrophy group,1 which also includes butterfly-shaped pigment dystrophy, reticular dystrophy of the retinal pigment epithelium (RPE), multifocal pattern dystrophy simulating fundus flavimaculatus, and fundus pulverulentus (Table 1). Due to variable expression of a mutation in the peripherin-2 (PRPH2) gene, autosomal dominant pattern dystrophies differ in onset, impact on vision and rate of occurrence.1
Butterfly-shaped pigment dystrophy presents with yellow and melanin pigmentation in the fovea typically in a triradiate or butterfly wing-like pattern.1 Reticular dystrophy of the RPE presents with a network of pigmented lines in the posterior pole that can resemble a fishnet or chicken wire.1,3 Multifocal pattern dystrophy simulating fundus flavimaculatus is characterized by many irregular yellow lesions that are widely distributed in the arcades and posterior pole.1 This is a differentiating characteristic because fundus flavimaculatus and Stargardt disease are more dispersed, including outside the arcades.1 Fundus pulverulentus presents with macular pigment mottling.1
Patients with AOFVD typically complain of mild blurring of vision, small central or paracentral scotomas or mild metamorphopsia.1,2,9 The typical presentation is bilateral, asymmetric foveal or perifoveal subretinal lesions that are yellow, solitary, round to oval and elevated, often with central pigmentation from one-third to one-half disc diameter in size.2,6,9,10 The disease can quite often be confused with other macular diseases and is most often misdiagnosed as AMD or Best disease. The effect on visual acuity is generally mild with an average of 20/50.1 A study of 85 patients with AOFVD found that 43% of the eyes had visual acuity better than or equal to 20/80; however, only 20% of eyes maintained this level of acuity after 4 years of follow-up.9 Vision-threatening complications of AOFVD include CNV, geographic atrophy and outer retinal atrophy.1,9 Subfoveal CNV has been reported to occur in 11.7% of cases.9 AOFVD typically presents without subretinal fluid or hemorrhage when there is no CNV.1,9
What are key differential diagnoses for adult-onset vitelliform dystrophy?
AMD is a key differential as it is the most significant cause of irreversible vision loss among older adults worldwide.1,4 It typically presents after age 50, bilaterally with drusen, hyperpigmentation or hypopigmentation of the RPE, geographic atrophy, RPE detachment and/or CNV.2,4 In dry AMD, OCT shows sub-RPE deposits and alterations. Loss of RPE and photoreceptors is seen in more advanced stages such as geographic atrophy.1,2,3,4 In wet AMD, OCT shows subretinal or intraretinal fluid or pigment epithelial detachments PEDs.1,2 The PEDs in wet AMD can appear with variable internal reflectivity on OCT and have been described as either fibrovascular, drusenoid or serous in nature.1,2
FAF in dry AMD exhibits hypofluorescence due to RPE loss and lipofuscin leading to a region with a high-contrast transition between areas of viable and non-viable retina.2,5 FA can confirm the presence of CNV in wet AMD. In dry AMD, FA demonstrates hypofluorescence due to drusen masking the background fluorescence, or hyperfluorescence due to a window defect from the atrophy of the overlying RPE.3,5 The patient in the case described here had a history of dry AMD noted for at least the past 3 years.
Another main differential is Best disease or vitelliform macular dystrophy. Similar to AOFVD, Best disease occurs when lipofuscin accumulates in the central macula causing progressive vision loss.1 It differs from AOFVD in that Best disease presents early in life.1,6 Genotypically, Best disease is an autosomal dominant macular dystrophy linked to mutations in the bestrophin 1 (BEST1) gene.1,6 The exact mechanism for how BEST1 gene mutation leads to accumulation of lipofuscin has not been definitively proven.6 Best disease presents during childhood with bilateral yellow yolk-like macular lesions with a diameter ranging from a few hundred micrometers to a few millimeters.2,6 As the patient ages with Best disease, the lesion will eventually scar and atrophy leading to a more difficult diagnosis later in life.2 Multiple stages of evolution have been noted for Best disease.1,6,7,8 Stage 1 or pre-vitelliform stage is characterized by lower-than-expected electro-oculography (EOG) in an asymptomatic infant or child with a normal fundoscopic appearance.6,7 Stage 2 is vitelliform and occurs from infancy to early childhood and does not impede visual acuity.1,6,7 It presents with a circumscribed delineated macular lesion described as “sunny side up egg yolk” with size varying from 0.5-2 disc diameters.6,7 At this stage, the dystrophy may be unilateral or appear asymmetric between the two eyes.6,7,8 Stage 3 is pseudohypopyon described as “scrambled egg.”6,7,8 Stage 4 is vitelliruptive when the lesion breaks up and visual acuity declines.6,7 Stage 5 is atrophy when all pigment has disappeared leaving an atrophic scar of the RPE.6,7 A sixth stage can occur in which CNV may develop and lead to a subretinal bleed, subretinal fibrosis and further decline in visual acuity.6,7
Ancillary diagnostic testing is useful in Best disease. A patient with Best disease has an Arden or light to dark ratio of less than 1.5 with EOG testing, and electroretinogram (ERG) can sometimes show a reduced C-wave.1,6,8 Importantly, this is the only disease with a relatively normal ERG with an abnormal EOG.6,8 OCT reveals dome-shaped, hyper-reflective and homogenous lesions located below the hyper-reflective photoreceptor layer.3 In the earlier stages, visual acuity can remain good; however, as the lesion atrophies, vision typically drops to the 20/200 range.1 As with AOFVD, there is a rare chance of CNV development.1 Though this patient’s fundoscopic and OCT findings correlated to Stage 2 or the vitelliform stage of Best disease, Stage 5 or the atrophic stage would be expected if he had Best disease given his age of 69 years. Stage 5 of Best disease typically occurs after age 40.6
A third differential is multifocal vitelliform lesions without Best disease. This is when multifocal vitelliform lesions, similar to those in Best disease, are seen around the macular vascular arcades and optic disc.1 Differing from Best disease this occurs initially in adult life.1 In addition, EOG is normal and family history is negative.1 It is important to note that genetically confirmed Best disease can present with multifocal lesions.1
The fourth differential for AOFVD is multifocal pattern dystrophy. This presents with multiple scattered irregular yellow lesions or flecks like those in fundus flavimaculatus and typically presents between the fourth and sixth decades of life.1 OCT demonstrates multiple hyper-reflective areas between the RPE and ellipsoid zone.1,3 FA shows hyperfluorescence of the flecks, and the choroid would not be dark compared to fundus flavimaculatus.1
How does adult-onset vitelliform dystrophy differ from Best disease?
AOFVD differs from Best disease as AOFVD has a later onset, typically from age 30-50 years, presents with mild to moderate symptoms, and can have a normal or mildly subnormal EOG with an Arden ratio of less than 1.7.5 In Best disease, the onset is typically early, before age 30, and is associated with an abnormal EOG.6 The subretinal lesions tend to be larger in Best disease, from 1-5 mm compared to one-third to one-half disc diameters in AOFVD.6 AOFVD occurs when there is insult to the RPE with focal loss of photoreceptors in the area of atrophic RPE damage.6 Pigmented material can be found between the retina and Bruch’s membrane.9 Genetically, Best disease is caused by a mutation in BEST1. Some forms of AOFVD have been linked to mutations in BEST1 and PRPH2 (formally known as retinal degeneration slow [RDS]) though the causative gene for AOFVD has not been discovered.5,6,7
Describe useful diagnostic testing to aid in diagnosis of adult-onset vitelliform dystrophy and expected findings
Imaging that can be helpful includes macular OCT, FAF, FA and/or ICGA. Macular OCT can help differentiate the layer the lesion impacts, such as subretinal for AOFVD or sub-RPE for more drusenoid changes. OCT shows the vitelliform material as a highly reflective dome-shaped lesion located between the photoreceptor layer and the RPE.1,2,7 In this patient, the sub-RPE macular deposits seen on OCT were more suggestive of vitelliform lesions than drusen. OCT is also useful for classifying AOFVD into four different stages.9 The vitelliform stage is characterized by a dome-shaped subretinal lesion between the RPE and ellipsoid zone.9 In the pseudohypopyon stage, a hyporeflective area is seen with a hyper-reflective area containing the remaining vitelliform material corresponding to the disassociation of the lesion.9 In the vitelliruptive stage, the lesion is flattened, meaning the fluid has subsided and atrophy of the outer retina and RPE is made apparent.9 The vitelliruptive stage of AOFVD would demonstrate atrophy of the ellipsoid zone, outer nuclear layer and RPE.9 FAF is another useful imaging modality that can show increased autofluorescence within foveal yellow lesions due to accumulation of lipofuscin within the deposit.9 When atrophy occurs, the lesion presents with hypofluorescence.9 FA can demonstrate an early central hypofluorescence corresponding to the yellow lesion with a surrounding ring of hyperfluorescence.1,9 The hypofluorescence is due to the vitelliform material and pigment, which can be surrounded by an area of atrophic RPE.1,9 In late phases, the entire lesion shows hyperfluorscence, which can cause confusion with CNV. This may require ICGA for confirming the presence of CNV.9
What treatment options are available for management of adult-onset vitelliform dystrophy?
Treatment and management of AOFVD is dependent on whether CNV or subretinal fluid develops.1,9 There is no treatment available to prevent the development of a vitelliform lesion.9 Several studies evaluated photodynamic therapy (PDT) or anti-VEGF therapy for patients with AOFVD without CNV and did not yield favorable results.9 Therefore, these treatments are not recommended in cases without CNV. Gene therapy is being investigated for possible prevention of vitelliform lesion development.9 If CNV develops from AOFVD, anti-VEGF treatment can be used.9 Similar efficacy has been noted with bevacizumab and ranibizumab injections in AMD and AOFVD patients with CNV.9 PDT may result in RPE atrophy and poorer visual outcomes in AOFVD associated CNV and is generally avoided in favor of anti-VEGF injections.9 When vision loss caused by AOFVD interferes with a patient’s ability to perform activities of daily living, referral to a low vision specialist can be useful.
The patient in this case report had relatively good visual acuity, no family history of macular disorders, and no personal history of macular or retinal abnormality prior to the past 3 years. This suggested late onset and supported the presumptive diagnosis of AOFVD rather than the original diagnosis of AMD. In addition, his macular deposits were sub-RPE, which is more consistent with AOFVD than with AMD. EOG could be beneficial in differentiating Best disease from AOFVD; however, the patient’s advanced age, the level of retinal presentation and the near normal visual acuity suggested AOFVD rather than Best disease.
Conclusion
This case report highlights AOFVD as an uncommon macular disorder that should be considered in patients with macular lesions that do not follow the typical AMD appearance or course. The conditions have different treatment and management approaches and can influence family members’ ocular health risk. Clinicians should also be aware of the importance of differentiating AOFVD from Best disease due to potential genetic counseling that would be pertinent in Best disease. In addition to dilated fundus examination, ancillary imaging such as OCT, FAF, FA or ICGA is critical in differentiating AOFVD from other macular disorders. If CNV develops as a complication of AOFVD, standard of care involves intravitreal injection of anti-VEGF agents to prohibit angiogenesis and vascular permeability. Whenever a patient with AOFVD encounters difficulty performing activities of daily living, low vision examination and/or rehabilitation is appropriate for determining whether a higher reading add, magnifiers, telescopes, video magnifiers or auditory devices may be necessary for meeting vision-related goals.
References
- Salmon J, Bowling B. Kanski’s clinical ophthalmology. 9th ed. Elsevier Limited; c2020. Chapter 14, Acquired macular disorders; p. 555-613.
- Goldman DR, Waheed NK, Duker JS. Atlas of retinal OCT: optical coherence tomography. 1st ed. Elsevier Inc.; c2018. Section 11, chapter 11.1, Vitelliform dystrophy; p. 72.
- Sadda SR, Schachat AP, Wilkinson CP, Hinton DR, Wiedemann P, Freund KB, Sarraf D, editors. Ryan’s Retina. 7th ed. Elsevier; c2023. Section 1, Retinal degenerations and dystrophies; p. 953-96.
- Ferri FF, editor. Ferri’s Clinical Advisor 2023. Elsevier, Inc.; c2023. Macular degeneration; p. 934-934.
- Friedman NJ, Kaiser PK, Pineda R II. The Massachusetts Eye and Ear Infirmary illustrated manual of ophthalmology. 5th ed. Elsevier Inc.; c2021. Chapter 10, Retina and choroid; p. 337-524.
- Tripathy K, Salini B. Best Disease (updated 2022 Feb 21) [Internet]. Treasure Island, FL: StatPearls Publishing. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537290/
- Chowers I, Tiosano L, Audo I, Grunin M, Boon CJ. Adult-onset foveomacular vitelliform dystrophy: A fresh perspective. Prog Retin Eye Res. 2015 Jul;47:64-85. doi: 10.1016/j.preteyeres.2015.02.001
- Johnson AA, Guziewicz KE, Lee CJ, Kalathur RC, Pulido JS, Marmorstein LY, Marmorstein AD. Bestrophin 1 and retinal disease. Prog Retin Eye Res. 2017 May;58:45-69. doi: 10.1016/j.preteyeres.2017.01.006
- Carnevali A, Al-Dolat W, Sacconi R, et al. Diagnosis, management and future treatment options for adult-onset foveomacular vitelliform dystrophy. Expert Review of Ophthalmology. 2018 June 7;13(3):161-69. doi: 10.1080/17469899.2018.1483722
- Do P, Ferrucci S. Adult-onset foveomacular vitelliform dystrophy. Optometry. 2006 Apr;77(4):156-66. doi: 10.1016/j.optm.2006.01.020



