PEER REVIEWED
Management of Corneal Abrasion Following Intravitreal Injection
Eric Ryan Harris, OD, FAAO
Abstract
Traumatic corneal abrasions typically cause acute eye pain. However, the insult may not be immediately obvious to the patient. This report highlights a case of corneal abrasion presumed to be from use of an eyelid speculum during an intravitreal injection of anti-vascular-endothelial-growth-factor medication for chronic cystoid macular edema. This report provides an overview of basic corneal epithelial anatomy and explains the pathophysiology of corneal epithelial damage from trauma. It also reviews multiple evidence-based treatment strategies for managing the tissue insult, promoting corneal wound healing, preventing infection and managing pain caused by this condition.
Key Words: cornea, corneal abrasion, ocular anatomy, eye pain, corneal epithelium
Introduction
A corneal abrasion is a disruption of the corneal epithelial integrity, usually due to external trauma, ocular foreign bodies, chemical burns or contact lenses.1 Corneal abrasions account for the largest proportion of eye-related complaints in emergency departments, with an incidence of three in 1,000 visits per year in the United States. Additionally, many patients with corneal abrasion report directly to optometrists, ophthalmologists and urgent care clinicians.2,3 Although many heal within a few days, abrasions can result in significant morbidity, suffering and loss of productivity for some patients.4,5 Knowledge of the tissue repair response within the human cornea is crucial to understanding a corneal abrasion patient’s best, safest and most effective treatment plan.
Case Description
Initial encounter
A 67-year-old male presented for a walk-in optometry clinic visit with acute, constant and intense right eye pain, redness, photophobia and tearing that began 1 day earlier. This patient had been seen several times in the previous 16 months by a retinal specialist in the clinic for a central retinal vein occlusion (CRVO) and chronic cystoid macular edema (CME) of the right eye. The patient was seen the day before the walk-in encounter by the retinal specialist, who administered the patient’s seventh intravitreal injection of aflibercept in the right eye. The procedure was documented as successful and without complication. However, upon arriving home, the patient experienced rapid onset of the symptoms described.
Other than the CRVO/CME, the patient’s ocular history consisted of emerging moderate nuclear sclerotic cataracts with vacuoles in both eyes, compound myopic astigmatism and presbyopia. His medical history was significant for type 2 diabetes, hypertension and hypercholesterolemia. The patient’s medication list consisted of metformin, metoprolol and atorvastatin. Best visual acuity was 20/70 in the right eye, which was decreased from his post-CRVO acuity (usually between 20/40 and 20/50). Visual acuity was a stable 20/30 in the left eye. Results of entrance testing, including confrontation visual fields, pupils and extraocular motilities, were unremarkable except for a 1+ afferent pupillary defect in the right eye, which was stable since the onset of the CRVO.
External examination, eyelids, eyelashes, conjunctiva, cornea and iris were all unremarkable during slit-lamp and anterior segment examination of the left eye. The anterior chamber was deep and quiet. An undilated view through the pupil revealed mild-to-moderate nuclear sclerosis of the crystalline lens with multiple small central vacuoles. Intraocular pressure measured by non-contact tonometry was 19 mmHg.
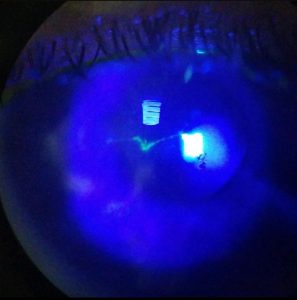
Figure 1. Right eye at initial encounter: V-shaped epithelial defect on the central cornea. Click to enlarge
In the patient’s right eye, external examination, eyelids and eyelashes were all unremarkable. The patient showed diffuse mild injection on the bulbar conjunctiva. There was a 3-mm round area of subconjunctival hemorrhage at the aflibercept injection site on the superior bulbar conjunctiva. Slit-lamp examination of the cornea revealed a 0.50 mm x 2.0 mm V-shaped epithelial defect on the central cornea, which stained with sodium fluorescein (Figure 1). No residual corneal epithelium requiring debridement was present around the wound border, and no foreign body was present during eyelid eversion. The anterior chamber was deep and quiet. An undilated view through the pupil revealed moderate nuclear sclerosis of the crystalline lens. The patient was diagnosed with a corneal abrasion of the right eye, presumed to be the result of eyelid speculum trauma. One drop of 0.5% proparacaine was instilled in-office in the right eye for initial comfort and cooperation with placement of a bandage contact lens (BCL) (senofilcon A). The patient was prescribed 0.5% moxifloxacin ophthalmic solution 4 times daily. Additionally, the patient was given a sample of Refresh Optive artificial tears to use as needed for irritation. Immediately after placement of the BCL, the patient noted a significant improvement in pain. The patient was told to return in 2 days for a follow-up examination. Ideally, he would have been seen the next day. However, he had transportation available only on certain days. He was given contact information for the on-call ophthalmology resident and instructed to report if his symptoms worsened in the interim. The retinal specialist was contacted about this encounter, and he was thankful for the communication.
Follow-up
The patient returned for follow-up as scheduled with no ocular complaints. He reported compliance with the topical medication regimen as prescribed. He reported no pain since the application of the BCL 2 days prior. He also reported that his vision was back to normal, and that photophobia and tearing were resolved. Best-corrected visual acuities returned to baseline at 20/50 in the right eye and 20/30 in the left eye. External examination, eyelids, eyelashes, anterior chamber and anterior segment findings were stable in the left eye.
External examination, eyelids, eyelashes and anterior chamber were stable in the right eye. At the aflibercept injection site, a 1-mm area of resolving subconjunctival hemorrhage remained. The BCL was still present. It was gently “floated” using artificial tears prior to removal to lessen the chance of new corneal epithelial damage. Following lens removal, corneal evaluation showed an intact epithelium with no sodium fluorescein staining. Intraocular pressure was measured as 19 mmHg in the right eye and the left eye using Tonopen. Because the corneal abrasion had healed, the patient was told to discontinue the 0.5% moxifloxacin. He was, however, told to frequently instill Refresh Optive artificial tears in the right eye at least 4 times daily for the next week. He was cautioned about the possibility and symptoms of recurrent corneal erosion. The patient was counseled on following up with his retinal specialist for ongoing intravitreal injections as scheduled.
3 months later

Figure 2. Right eye 3 months after initial encounter and initial successful treatment: oval central corneal abrasion Click to enlarge
The patient presented to the optometry walk-in clinic in a similar fashion as before, 1 day after receiving an intravitreal injection of aflibercept in the right eye. He reported eye pain, tearing and photophobia beginning 2-3 hours following the injection. His visual acuity had worsened to 20/150. All other clinical findings were stable/unchanged compared to his initial urgent examination, and Tonopen-measured intraocular pressures were 18 mmHg in each eye. Following the examination, the patient was diagnosed with a 2.5 mm x 3.0 mm oval central corneal abrasion in the right eye following aflibercept injection (Figure 2). The mechanism was presumed to be due to the eyelid speculum. The patient was treated in-office with 1 drop of 0.5% proparacaine for comfort and cooperation with placement of a BCL (senofilcon A). He was prescribed 0.5% moxifloxacin ophthalmic solution to use 4 times daily and advised to use Refresh Optive artificial tears as-needed.
The patient responded favorably again and returned for follow-up 2 days later. He was symptom-free and vision returned to his baseline. All other ophthalmic findings and metrics were stable/unremarkable. The BCL was floated/removed, 0.5% moxifloxacin was discontinued, and the patient was told to use artificial tears 4 times daily for the next week. During a discussion with the treating retinal specialist, who was not comfortable assessing corneal findings, it was recommended that the patient be pressure-patched until the following morning for future injections. Following this intervention, the patient has not suffered from a new corneal abrasion after intravitreal injection or a spontaneous recurrent corneal erosion.
Education Guidelines
Key concepts
- The basic anatomy and kinetics of the corneal epithelium
- The cellular response to corneal epithelial trauma
- Making the diagnosis of corneal abrasion
- Appropriate treatment plans based on the clinical scenario
Learning objectives
- Recognize the clinical presentation of corneal abrasion including history, symptoms and signs
- List the potential differential diagnoses of corneal abrasion
- Initiate an effective and safe management plan for a patient with a corneal abrasion
- Explore different management strategies in complicated scenarios involving corneal abrasion or those involving patients with high risks
Discussion questions
1. Knowledge and concepts about the clinical case and condition
- What is the pathophysiology of corneal abrasion?
- What are the primary concerns of patients presenting with corneal abrasion?
- What complications can occur secondary to corneal abrasion?
2. Differential diagnosis and risk factors
- What other eye conditions can result in symptoms like those experienced with corneal abrasion?
- What other eye conditions can result in clinical signs like those seen with corneal abrasion?
- If a corneal abrasion is suspected, what are pertinent case history elements to explore to maximize safety in the treatment approach?
3. Management and the role of optometry in the care of patients with corneal abrasion
- What is the prognosis of a small- to medium-sized corneal abrasion without complications?
- How would you minimize the risk of permanent corneal damage in a patient with a corneal abrasion who received a bottle of topical tetracaine from the emergency department prior to seeing you?
- Name two ophthalmic fourth-generation fluoroquinolones and two ophthalmic second-generation fluoroquinolones
- How soon should a newly diagnosed 5-mm central corneal abrasion in a 15-year-old patient be seen for follow-up: 1 day, 5 days, 1 week or as-needed only?
4. Critical-thinking scenarios
- How might the addition of a topical corticosteroid or cycloplegic interfere with corneal epithelial wound healing in a patient with corneal abrasion and concurrent traumatic anterior uveitis?
- A patient with poor hand hygiene who chronically overwears and sleeps in contact lenses presents with a new corneal abrasion. Why is there a safety concern for the use of a BCL?
- A new patient presents to your office with a large, central, non-traumatic epithelial defect of the right cornea. This is 3 days after being treated by a local emergency department with ciprofloxacin and tetracaine eye drops, each 5 times daily. Since the emergency department visit, the patient’s pain has not improved, and vision is slightly worse. What differentials/complications should be considered in this patient?
Assessment of learning objectives
This teaching case is suitable for fourth-year optometry students and new practitioners in numerous settings. For learners who prefer independent study, this learning assignment can be completed in its entirety by the student alone, but should be reviewed with a practicing optometrist or ophthalmologist. However, a small-group setting, which can be in-person or virtual, is preferred. In this environment, students could be assigned the task of reading the case elements and the discussion prior to their meeting. Students should also be expected to complete and record the discussion questions prior to their group gathering. Ideally the group leader would be a faculty member, optometry resident or senior optometrist within their organization. Upon meeting, the abstract can be read to reset the tone for the discussion, and each of the discussion questions can be reviewed. All learners present should share their responses. A unique aspect of corneal abrasion is that it can be managed in multiple safe and effective ways. The variety of responses from the learners can help solidify this idea, which can aid the clinician in corneal abrasion cases that are not so straightforward.
Discussion
A corneal abrasion is a disruption of the corneal epithelial integrity, usually due to external trauma, ocular foreign bodies, chemical burns or contact lenses.1 The condition should be suspected in patients who present due to acute ocular pain, conjunctival injection, foreign body sensation or lacrimation following ocular trauma. Other symptoms may include blepharospasm, blurred vision and headache. Symptoms are often dependent on the size and location of the corneal epithelial defect.6 Traditionally, a diagnosis of corneal abrasion is based on a history of recent ocular trauma and the presence of a corneal epithelial defect observed with a slit-lamp biomicroscope with and without sodium fluorescein.7 However, differential diagnoses include recurrent corneal erosion, herpes simplex keratitis, herpes zoster keratitis, corneal foreign body and traumatic uveitis.
Homeostatic balance of the human corneal epithelium depends on a critical cycle of cellular loss, renewal/proliferation, differentiation and migration. The basal cell layer of the corneal epithelium is a single layer of columnar cells that lies anterior to the basement membrane, connected via hemidesmosomes.8-12 Basal cells are the only corneal epithelial cells capable of division. These basal cells are renewed via stem cells near the limbus. Then, they gradually proliferate more centrally. Others differentiate and migrate anteriorly into the middle layers of corneal epithelium. These new wing cells have a concave posterior and a convex anterior surface. Desmosomes and gap junctions connect wing-cell-to-wing-cell and wing-cell-to-surface-cell.13
Surface cells are the most anterior layer of the corneal epithelium. The cellular height, usually 2 layers thick, is significantly shorter and more stratified than both basal and wing cells.14 Surface cells are nonkeratinized and squamous. Over time, surface cells mature, are gradually sloughed off, and are replaced by the underlying wing cells.15 In a normal cornea, the turnover from basal cell renewal through loss of surface cells takes about 7 days.16,17 This can take significantly longer in patients with corneoscleral/limbal disease or those with concurrent use of corticosteroids.18,19 Additionally, the corneal epithelium of patients with a history of diabetes mellitus has been shown to be thinner, of lower cell density, more fragile and have a reduced barrier function than the corneal epithelium of patients who do not have diabetes.18-26
During corneal epithelial homeostasis, the limbal epithelial stem cells divide occasionally in order to replace surface cells that slough off.27,28 However, in response to trauma, such as a corneal abrasion, the proliferation rate temporarily increases 8- to-9-fold in the limbus and approximately 2-fold in the periphery and central cornea.27,29,30 The wound healing process has been described in two distinct phases. Phase one involves cellular and subcellular reorganization, triggering epithelial cell migration at the edge of the wound.31,32 Phase two involves the cellular proliferation, differentiation and ultimately stratification to restore the damaged epithelial layer(s).32,33 These cellular pathways are mediated by various growth factors, cytokines,34,35 purinergic receptors,36 toll-like receptor 4,37 rho-associated protein kinase,38,39 proteinases such as matrix metalloproteinases,40-43 neuropeptides44,45 and extracellular matrix.35,46 In the event of corneal trauma, such as a corneal abrasion from a fingernail scratch, tissue repair may occur fairly quickly, in hours to days.16,17 However, if the traumatic event damages the basement membrane, renewal of this layer and hemidesmosomes can take several weeks or months.47,48
Following a corneal abrasion, a primary consideration for both the optometrist and the patient is pain management because significant ocular discomfort is expected. This is due to corneal epithelial damage near terminal branches of the sensory nerve fibers derived from the ophthalmic branch of the trigeminal nerve.49 Several treatment strategies exist to decrease pain in these patients. They include cycloplegics, topical non-steroidal anti-inflammatory drugs (NSAIDs), pressure-patching, topical anesthetics and BCLs.
Patients who have ciliary flush, anterior chamber cell and/or flare and photophobia suggestive of ciliary spasm could potentially benefit from topical cycloplegics such as cyclopentolate. However, cycloplegics have not been shown to be superior to placebo for managing pain in patients with corneal abrasion.50,51 There does not appear to be a delayed wound healing effect in corneal abrasion patients using cycloplegics. Many eyecare professionals reserve cycloplegics for larger corneal abrasions or when ciliary spasm or intraocular inflammation are present.51,52 The patient in this case report did not have an appreciable anterior chamber reaction, and his ocular pain subsided immediately upon BCL placement. For these reasons, a cycloplegic was not prescribed.
Multiple topical NSAIDs have been studied clinically for management of corneal abrasion pain. Subjectively, ketorolac was not shown to be superior to placebo in one 2001 study.53 However, the treatment group required less supplementary oral analgesia to control their pain. Studies of the efficacy of diclofenac for treating pain due to corneal abrasion have produced mixed results. One study did not find a statistically significant difference in pain management with diclofenac vs. placebo on day 1 of treatment, but did find that patients in the diclofenac group were less likely to rank their pain as moderate or severe on days 2 and 3.54 Another study found that topical diclofenac resulted in a 3-fold better improvement in pain compared to the control medication when assessed 2 hours after instillation.55 The diclofenac group also required less supplemental oral analgesia compared to the control group.
Other topical NSAIDs such as flurbiprofen and indomethacin have also been studied as potential pain-management strategies for patients with corneal abrasion. While the medications showed some promise, larger multicenter studies are needed for corroboration.56-58 One group performed a systematic review and meta-analysis of earlier works that examined pain management, corneal healing and complications for different treatment modalities for corneal abrasion. They found only two events considered to be major complications among 1,046 patients who received a topical NSAID.59 While there is an association between topical NSAIDs and corneal toxicity, impaired corneal sensation, persistent epithelial defects, infiltrative keratitis and corneal melts, it appears that these high-criticality events occur in low frequency.60-65 One study found no statistically significant difference in corneal wound healing by day 5 when comparing combined topical indomethacin/gentamicin to topical gentamicin alone.56
Topical anesthetic use for pain management is a controversial topic in optometry, ophthalmology and emergency medicine. Anecdotal reports of a new patient presenting with a corneal abrasion who brings along a bottle of tetracaine either stolen or received from an emergency department physician have been circulating for decades. A thorough literature review makes these reports sound plausible. Use of these medications outside of the clinical setting are largely discouraged in optometry and ophthalmology due to the risk of potentially sight-threatening topical anesthetic abuse keratopathy.66,67 Interestingly, emergency medicine has continued to investigate the use of topical proparacaine, amethocaine and tetracaine for corneal abrasion and other corneal trauma with very small patient sample sizes and variable results.68-70 As a result, for eyecare professionals, aside from the outpatient clinical setting, it is considered common practice to discontinue or confiscate these medications if a patient presents with one. The Wills Eye Manual, specifically marketed to both eyecare providers and emergency physicians, does not list topical anesthetics for corneal abrasion as a treatment option.1 Meta-analyses of patients receiving topical anesthetics for corneal abrasions showed a relatively low and fairly even number of complications when compared to the control groups.59
Pressure-patching is a non-medication pain management strategy for patients with corneal abrasion. The rationale rests on the inability of the eyelid to wipe across the wounded epithelial surface cells and exposed corneal nerve endings during blinks. Historically, pressure-patching was routinely embraced as part of the treatment process for corneal abrasion despite a lack of evidence for the practice.71,72 Most eyecare providers no longer perform patching due to the loss of binocularity, persistent foreign body sensation, inability to instill topical medications, microbial growth potential, the potential for reduced corneal oxygenation or impaired wound healing.73-76 Additionally, there is no strong evidence linking pressure-patching to appreciable improvements in pain or healing rates in patients with corneal abrasion.53,77 However, although better treatment modalities exist, pressure-patching is not contraindicated in all scenarios. The patient in this teaching case report received 5% povidone-iodine application prior to each aflibercept injection and a drop of 0.5% topical moxifloxacin immediately following each procedure. Therefore, the risk of endophthalmitis or infectious keratitis was not a significant concern with pressure-patching for 24 hours. In this scenario, patching was an effective plan for the patient and physician.
A second consideration for corneal abrasion management is the promotion of corneal healing. BCLs are frequently used in corneal surgery for this purpose.78 Favorable observations have been appreciated with conditions such as neurotrophic keratitis,79 ocular chemical injuries80 and ocular graft-vs.-host disease,81 as well as following refractive surgery82,83 and penetrating keratoplasty.84 They have been shown to improve Ocular Surface Disease Index scores, tear break-up time and comfort in dry eye syndrome patients immediately following cataract surgery.85,86 However, there is not enough evidence in the literature on the topic of BCLs specifically for treatment of corneal abrasion to either support or refute the benefits on healing or pain control. With that said, BCLs are still commonly used for these purposes. Currently, several high dK/t soft contact lens materials, including Air Optix® Night & Day Aqua (lotrafilcon A), Purevision® (balafilcon A) and Acuvue® Oasys (senofilcon A), have FDA approval for therapeutic use for a variety of corneal indications including pain relief, enhanced corneal healing, corneal protection, corneal sealing and drug delivery.87 Complications associated with BCLs are rare, but do exist with high-criticality scenarios such as infectious keratitis.88,89 Because of this, a thorough risk assessment of a patient’s potential for developing infectious keratitis must precede the use of BCLs.
The third consideration for corneal abrasion management is antibacterial prophylaxis. In the literature, there is an absence of compelling evidence to support routinely prescribing topical antibiotics for the prevention of bacterial keratitis. However, one study from Nepal found that only 4% of patients receiving topical chloramphenicol for traumatic corneal abrasion (n = 442) developed a corneal ulcer.90 Interestingly, the most significant determinant of bacterial keratitis development in these patients was found to be the time between the corneal trauma and the patient presenting for treatment. In this study, 109 patients presented for treatment 18-24 hours after the injury, and four (3.7%) developed ulceration. Of the 49 patients presenting 24-48 hours after trauma, 14 (29%) subsequently developed a corneal ulcer. In this study, due to ethics, there was not a control group that did not receive topical antibiotics. However, many eyecare providers and emergency physicians prescribe topical antibiotics to promote a sterile corneal epithelium during the healing stages of a corneal abrasion.7,91
The class of topical antibiotic selected, its preparation, dosing schedule and treatment period often vary depending on the prescriber’s preferences, which are determined by the abrasion size, location and other potential ocular surface biofilm risk factors. Pseudomonas aeruginosa, for example, has been shown to exhibit resistance to contact lens disinfectants.92,93 Additionally, this species has been shown to colonize contact lens materials during wear and survive in contact lens storage cases.94,95 Because of this, optometrists and other prescribers must proceed with caution when considering BCLs and selecting topical antibiotics for corneal abrasions in contact lens wearers who are non-compliant with their recommended daily lens care regimen. The Wills Eye Manual recommends a topical fluoroquinolone for corneal abrasion in contact lens wearers, as well as those that occur in other high-risk scenarios, such as fingernail or vegetative matter trauma.1 Of topical fluoroquinolones, highly virulent bacteria such as Pseudomonas and resistant forms of Staphylococcus aureus show the highest susceptibility to the fourth-generation preparations.96 One study found that 96.2% of gram positive cocci isolated from bacterial keratitis was susceptible to topical gatifloxacin, a fourth-generation fluoroquinolone, whereas only 60.4% were susceptible to topical ciprofloxacin, a second-generation fluoroquinolone.97 When it comes to methicillin-resistant S aureus, the literature has shown resistance that ranges from 68% – 85% for topical moxifloxacin and 71% – 85% for topical gatifloxacin, which is concerning given that these are both fourth-generation medications.98-100
The decisions of which formulation and class of topical antibiotic to prescribe for a corneal abrasion often depend on the patient’s ability to instill eye drops frequently, the age of the patient and the need for high-risk or broad-spectrum coverage. Aside from topical fluoroquinolones, there are additional classes of topical ophthalmic antibiotics to consider, including aminoglycosides, polymyxin B combinations and others. For a corneal abrasion that warrants an ophthalmic antibiotic ointment, Neosporin® (neomycin/polymyxin B/bacitracin) is one option that has broad-spectrum antibiotic activity. Tobramycin 0.3% is one of multiple topical ophthalmic antibiotics that is available as both a solution and an ointment. Erythromycin is a topical ophthalmic antibiotic that is only available as an ointment. Dosing for ophthalmic antibiotic ointments for corneal abrasion is often a decision based on the prescriber’s preference. One reason to prescribe an antibiotic ointment for a corneal abrasion is the longer contact time between the medication and the ocular surface compared to a solution or suspension. Optometrists should prescribe these medications with moderate consideration, balancing the efficacy of an ointment’s instillation frequency with the predictable side effect of blurred vision due to the viscosity of the ointment. Additionally, if a BCL is to be placed in conjunction with a topical ophthalmic antibiotic, formulations such as ophthalmic ointments or suspensions may destabilize the contact lens or cause prolonged blurred vision during treatment.
The decision of how to intervene with antibacterial prophylaxis, pain management and healing promotion often depends on the specifics of the case history and the clinical presentation. For instance, cases of suspected corneal abrasion that initially present much later after onset may have only minimal clinical signs that may only warrant a much more conservative approach, such as artificial tears. Other patients, for instance, with unclear history and/or dramatic slit-lamp findings, may warrant consideration of other treatment modalities, such as oral antiviral prophylaxis. Some family physicians recommend an initial follow-up of no more than 24 hours for most corneal abrasions.7 However, there is no consensus in the optometry and ophthalmology communities on timing of follow-up. This decision is dependent on multiple factors. Many eyecare professionals follow up with patients based on the key factors driving the encounter, such as the patient’s pain level, size and location of the corneal epithelial defect, and systemic and ocular comorbidities.
The patient in this case report suffered from a corneal abrasion twice following intravitreal injections administered by the same retinal specialist. The retinal specialist had no other patients calling to report pain symptoms or presenting to the same-day optometry clinic with an abrasion. There were several key questions to consider related to etiology. Was the etiology centered around patient-specific risk factors? Was this a technical problem at the hands of the retinal specialist, or the result of poor cooperation by the patient during the procedure? Was the trauma caused directly by an external source, such as an eyelid speculum or fingertip, or was it the result of hyperexposure of the ocular surface while the eyelid speculum was in place? These questions are challenging to answer after the fact, but it was crucial to at least consider each given this patient’s concurrent history of CRVO with CME and the long-term visual prognosis in the right eye in question. One meta-analysis showed that corneal abrasions have been reported as a major complication following intravitreal injections. However, the frequency was low, 46 cases after 44,734 injections over a 4-year period.101 Another retrospective study found slightly better results, but found that 0.60% of intravitreal injections resulted in an urgent follow-up visit, with corneal abrasions representing approximately 10% of those encounters.102
Conclusion
Intravitreal injections are one of the most common procedures performed by ophthalmologists in the United States. It is estimated that 5.9 million were performed in 2016, and the number is expected to rise yearly.103 As this continues, optometrists should be clinically prepared for the potential complications, such as corneal abrasion. With that said, it is more likely that optometrists would encounter patients with corneal abrasions due to other causes, such as fingernails, pet scratches, foreign bodies and chemical burns. Corneal abrasion is associated with multiple complications, such as recurrent corneal erosion,104,105 microbial keratitis106 and corneal scarring, which can result in varying degrees of vision loss. Pressure-patching is largely considered obsolete compared to more contemporary treatment approaches. However, it was successful as an ongoing post-intravitreal injection treatment for the patient in this case report. With the highest criticality complication of a corneal abrasion being infection with patching, this risk was mitigated using betadine prep prior to intravitreal injection and topical moxifloxacin following the procedure.
References
- Bagheri N, Wajda B, Calvo C, Durrani AE. The Wills Eye Manual. 7th ed. Philadelphia, PA: Lippincott Williams and Wilkins; 2017.
- Vernon SA. Analysis of all new cases seen in a busy regional centre ophthalmic casualty department during 24-week period. J R Soc Med. Apr 1983;76(4):279-82. doi:10.1177/014107688307600408
- Ahmed F, House RJ, Feldman BH. Corneal abrasions and corneal foreign bodies. Prim Care. Sep 2015;42(3):363-75. doi:10.1016/j.pop.2015.05.004
- Wong TY, Lincoln A, Tielsch JM, Baker SP. The epidemiology of ocular injury in a major US automobile corporation. Eye (Lond). 1998;12 ( Pt 5):870-4. doi:10.1038/eye.1998.220
- Khodadoust AA, Silverstein AM, Kenyon DR, Dowling JE. Adhesion of regenerating corneal epithelium. The role of basement membrane. Am J Ophthalmol. Mar 1968;65(3):339-48. doi:10.1016/0002-9394(68)93082-1
- Wakai A, Lawrenson JG, Lawrenson AL, et al. Topical non-steroidal anti-inflammatory drugs for analgesia in traumatic corneal abrasions. Cochrane Database Syst Rev. May 18 2017;5(5):CD009781. doi:10.1002/14651858.CD009781.pub2
- Wipperman JL, Dorsch JN. Evaluation and management of corneal abrasions. Am Fam Physician. Jan 15 2013;87(2):114-20.
- Kruse FE. Stem cells and corneal epithelial regeneration. Eye (Lond). 1994;8 (Pt 2):170-83. doi: 10.1038/eye.1994.42
- Thoft RA, Friend J. The X, Y, Z hypothesis of corneal epithelial maintenance. Invest Ophthalmol Vis Sci. 1983 Oct;24(10):1442-3.
- Tseng SC. Concept and application of limbal stem cells. Eye (Lond). 1989;3 (Pt 2):141-57. doi: 10.1038/eye.1989.22
- Hogan MJ, Cornea AJT. In Hogan MJ, Alvarado JA, Weddell, JE, editors: Histology of the Human Eye. Saunders; 1971.
- Eugene Wolff’s Anatomy of the Eye and Orbit. 7th ed: Saunders; 1976. p. 30.
- Williams KK, Watsky MA. Dye spread through gap junctions in the corneal epithelium of the rabbit. Curr Eye Res. 1997 May;16(5):445-52. doi: 10.1076/ceyr.16.5.445.7039
- Nichols B, Dawson CR, Togni B. Surface features of the conjunctiva and cornea. Invest Ophthalmol Vis Sci. 1983 May;24(5):570-6.
- Pfister RR. The normal surface of corneal epithelium: a scanning electron microscopic study. Invest Ophthalmol. 1973 Sep;12(9):654-68.
- Hanna C, Bicknell DS, O’Brien JE. Cell turnover in the adult human eye. Arch Ophthalmol. 1961 May;65:695-8. doi: 10.1001/archopht.1961.01840020697016
- Hanna C, O’Brien JE. Cell production and migration in the epithelial layer of the cornea. Arch Ophthalmol. 1960 Oct;64:536-9. doi: 10.1001/archopht.1960.01840010538009
- Ho PC, Elliott JH. Kinetics of corneal epithelial regeneration. II. Epidermal growth factor and topical corticosteroids. Invest Ophthalmol. Aug 1975;14(8):630-3.
- Elliott JH. Epidermal growth factor: in vivo ocular studies. Trans Am Ophthalmol Soc. 1980;78:629-56.
- Pepose JS, Ubels JL. The Cornea. In Hart WM Jr, editor: Adler’s Physiology of the Eye: Clinical Application. 9th ed. Mosby; 1992.
- Saini JS, Khandalavla B. Corneal epithelial fragility in diabetes mellitus. Can J Ophthalmol. Apr 1995;30(3):142-6.
- Quadrado MJ, Popper M, Morgado AM, Murta JN, Van Best JA. Diabetes and corneal cell densities in humans by in vivo confocal microscopy. Cornea. 2006 Aug;25(7):761-8. doi: 10.1097/01.ico.0000224635.49439.d1
- Yusufoğlu E, Güngör Kobat S, Keser S. Evaluation of central corneal epithelial thickness with anterior segment OCT in patients with type 2 diabetes mellitus. Int Ophthalmol. 2023 Jan;43(1):27-33. doi: 10.1007/s10792-022-02384-5
- Chang SW, Hsu HC, Hu FR, Chen MS. Corneal autofluorescence and epithelial barrier function in diabetic patients. Ophthalmic Res. 1995 Mar-Apr;27(2):74-9. doi: 10.1159/000267600
- Gekka M, Miyata K, Nagai Y, et al. Corneal epithelial barrier function in diabetic patients. Cornea. Jan 2004;23(1):35-7. doi:10.1097/00003226-200401000-00006
- Göbbels M, Spitznas M, Oldendoerp J. Impairment of corneal epithelial barrier function in diabetics. Graefes Arch Clin Exp Ophthalmol. 1989;227(2):142-4. doi:10.1007/BF02169787
- Lehrer MS, Sun TT, Lavker RM. Strategies of epithelial repair: modulation of stem cell and transit amplifying cell proliferation. J Cell Sci. 1998 Oct;111 (Pt 19):2867-75. doi: 10.1242/jcs.111.19.2867
- Pellegrini G, Golisano O, Paterna P, et al. Location and clonal analysis of stem cells and their differentiated progeny in the human ocular surface. J Cell Biol. May 17 1999;145(4):769-82. doi:10.1083/jcb.145.4.769
- Cotsarelis G, Cheng SZ, Dong G, Sun TT, Lavker RM. Existence of slow-cycling limbal epithelial basal cells that can be preferentially stimulated to proliferate: implications on epithelial stem cells. Cell. Apr 21 1989;57(2):201-9. doi:10.1016/0092-8674(89)90958-6
- Lavker RM, Wei ZG, Sun TT. Phorbol ester preferentially stimulates mouse fornical conjunctival and limbal epithelial cells to proliferate in vivo. Invest Ophthalmol Vis Sci. Feb 1998;39(2):301-7.
- Kuwabara T, Perkins DG, Cogan DG. Sliding of the epithelium in experimental corneal wounds. Invest Ophthalmol. Jan 1976;15(1):4-14.
- Crosson CE, Klyce SD, Beuerman RW. Epithelial wound closure in the rabbit cornea. A biphasic process. Invest Ophthalmol Vis Sci. Apr 1986;27(4):464-73.
- Anderson RA. Actin filaments in normal and migrating corneal epithelial cells. Invest Ophthalmol Vis Sci. Feb 1977;16(2):161-6.
- Lu L, Reinach PS, Kao WW. Corneal epithelial wound healing. Exp Biol Med (Maywood). Jul 2001;226(7):653-64. doi:10.1177/153537020222600711
- Yu FS, Yin J, Xu K, Huang J. Growth factors and corneal epithelial wound healing. Brain Res Bull. Feb 15 2010;81(2-3):229-35. doi:10.1016/j.brainresbull.2009.08.024
- Lee A, Derricks K, Minns M, et al. Hypoxia-induced changes in Ca(2+) mobilization and protein phosphorylation implicated in impaired wound healing. Am J Physiol Cell Physiol. May 15 2014;306(10):C972-85. doi:10.1152/ajpcell.00110.2013
- Eslani M, Movahedan A, Afsharkhamseh N, Sroussi H, Djalilian AR. The role of toll-like receptor 4 in corneal epithelial wound healing. Invest Ophthalmol Vis Sci. Sep 02 2014;55(9):6108-15. doi:10.1167/iovs.14-14736
- SundarRaj N, Kinchington PR, Wessel H, et al. A Rho-associated protein kinase: differentially distributed in limbal and corneal epithelia. Invest Ophthalmol Vis Sci. Jun 1998;39(7):1266-72.
- Yin J, Lu J, Yu FS. Role of small GTPase Rho in regulating corneal epithelial wound healing. Invest Ophthalmol Vis Sci. Mar 2008;49(3):900-9. doi:10.1167/iovs.07-1122
- Daniels JT, Limb GA, Saarialho-Kere U, Murphy G, Khaw PT. Human corneal epithelial cells require MMP-1 for HGF-mediated migration on collagen I. Invest Ophthalmol Vis Sci. Mar 2003;44(3):1048-55. doi:10.1167/iovs.02-0442
- Blanco-Mezquita T, Martinez-Garcia C, Proença R, et al. Nerve growth factor promotes corneal epithelial migration by enhancing expression of matrix metalloprotease-9. Invest Ophthalmol Vis Sci. Jun 04 2013;54(6):3880-90. doi:10.1167/iovs.12-10816
- Petznick A, Madigan MC, Garrett Q, Sweeney DF, Evans MD. Contributions of ocular surface components to matrix-metalloproteinases (MMP)-2 and MMP-9 in feline tears following corneal epithelial wounding. PLoS One. 2013;8(8):e71948. doi:10.1371/journal.pone.0071948
- Mauris J, Woodward AM, Cao Z, Panjwani N, Argüeso P. Molecular basis for MMP9 induction and disruption of epithelial cell-cell contacts by galectin-3. J Cell Sci. Jul 15 2014;127(Pt 14):3141-8. doi:10.1242/jcs.148510
- Gallar J, Pozo MA, Rebollo I, Belmonte C. Effects of capsaicin on corneal wound healing. Invest Ophthalmol Vis Sci. Oct 1990;31(10):1968-74.
- Yang L, Di G, Qi X, et al. Substance P promotes diabetic corneal epithelial wound healing through molecular mechanisms mediated via the neurokinin-1 receptor. Diabetes. Dec 2014;63(12):4262-74. doi:10.2337/db14-0163
- Suzuki K, Tanaka T, Enoki M, Nishida T. Coordinated reassembly of the basement membrane and junctional proteins during corneal epithelial wound healing. Invest Ophthalmol Vis Sci. Aug 2000;41(9):2495-500.
- Gipson IK, Spurr-Michaud S, Tisdale A, Keough M. Reassembly of the anchoring structures of the corneal epithelium during wound repair in the rabbit. Invest Ophthalmol Vis Sci. 1989 Mar;30(3):425-34.
- Gipson IK, Spurr-Michaud SJ, Tisdale AS. Hemidesmosomes and anchoring fibril collagen appear synchronously during development and wound healing. Dev Biol. 1988 Apr;126(2):253-62. doi: 10.1016/0012-1606(88)90136-4
- Ljubimov AV, Saghizadeh M. Progress in corneal wound healing. Prog Retin Eye Res. Nov 2015;49:17-45. doi:10.1016/j.preteyeres.2015.07.002
- Thiel B, Sarau A, Ng D. Efficacy of topical analgesics in pain control for corneal abrasions: a systematic review. Cureus. Mar 27 2017;9(3):e1121. doi:10.7759/cureus.1121
- Meek R, Sullivan A, Favilla M, Larmour I, Guastalegname S. Is homatropine 5% effective in reducing pain associated with corneal abrasion when compared with placebo? A randomized controlled trial. Emerg Med Australas. Dec 2010;22(6):507-13. doi:10.1111/j.1742-6723.2010.01346.x
- Al-Saleh GS, Alfawaz AM. Management of traumatic corneal abrasion by a sample of practicing ophthalmologists in Saudi Arabia. Saudi J Ophthalmol. 2018;32(2):105-109. doi:10.1016/j.sjopt.2017.10.007
- Goyal R, Shankar J, Fone DL, Hughes DS. Randomised controlled trial of ketorolac in the management of corneal abrasions. Acta Ophthalmol Scand. Apr 2001;79(2):177-9. doi:10.1034/j.1600-0420.2001.079002177.x
- Jayamanne DG, Fitt AW, Dayan M, Andrews RM, Mitchell KW, Griffiths PG. The effectiveness of topical diclofenac in relieving discomfort following traumatic corneal abrasions. Eye (Lond). 1997;11 (Pt 1):79-83. doi: 10.1038/eye.1997.15
- Szucs PA, Nashed AH, Allegra JR, Eskin B. Safety and efficacy of diclofenac ophthalmic solution in the treatment of corneal abrasions. Ann Emerg Med. Feb 2000;35(2):131-7. doi:10.1016/s0196-0644(00)70132-6
- Alberti MM, Bouat CG, Allaire CM, Trinquand CJ. Combined indomethacin/gentamicin eyedrops to reduce pain after traumatic corneal abrasion. Eur J Ophthalmol. 2001;11(3):233-9. doi:10.1177/112067210101100304
- Patrone G, Sacca SC, Macri A, Rolando M. Evaluation of the analgesic effect of 0.1% indomethacin solution on corneal abrasions. Ophthalmologica. 1999;213(6):350-4. doi:10.1159/000027453
- Brahma AK, Shah S, Hillier VF, et al. Topical analgesia for superficial corneal injuries. J Accid Emerg Med. May 1996;13(3):186-8. doi:10.1136/emj.13.3.186
- Yu CW, Kirubarajan A, Yau M, Armstrong D, Johnson DE. Topical pain control for corneal abrasions: a systematic review and meta-analysis. Acad Emerg Med. Aug 2021;28(8):890-908. doi:10.1111/acem.14222
- Nguyen KD, Lee DA. Effect of steroids and nonsteroidal antiinflammatory agents on human ocular fibroblast. Invest Ophthalmol Vis Sci. Aug 1992;33(9):2693-701.
- Hsu JK, Johnston WT, Read RW, et al. Histopathology of corneal melting associated with diclofenac use after refractive surgery. J Cataract Refract Surg. Feb 2003;29(2):250-6. doi:10.1016/s0886-3350(02)01702-9
- Qu M, Wang Y, Yang L, Zhou Q. Different cellular effects of four anti-inflammatory eye drops on human corneal epithelial cells: independent in active components. Mol Vis. 2011;17:3147-55.
- Lindstrom R. The pharmacologic and pathophysiologic rationale for using NSAIDs in ocular inflammatory disease and ocular surgery. Int Ophthalmol Clin. 2006;46(4):7-11. doi:10.1097/01.iio.0000212131.98760.a9
- Guidera AC, Luchs JI, Udell IJ. Keratitis, ulceration, and perforation associated with topical nonsteroidal anti-inflammatory drugs. Ophthalmology. May 2001;108(5):936-44. doi:10.1016/s0161-6420(00)00538-8
- Hersh PS, Rice BA, Baer JC, et al. Topical nonsteroidal agents and corneal wound healing. Arch Ophthalmol. Apr 1990;108(4):577-83. doi:10.1001/archopht.1990.01070060125062
- Rosenwasser GO, Holland S, Pflugfelder SC, et al. Topical anesthetic abuse. Ophthalmology. Aug 1990;97(8):967-72. doi:10.1016/s0161-6420(90)32458-2
- Rosenwasser GO. Complications of topical ocular anesthetics. Int Ophthalmol Clin. 1989;29(3):153-8. doi:10.1097/00004397-198902930-00005
- Ball IM, Seabrook J, Desai N, Allen L, Anderson S. Dilute proparacaine for the management of acute corneal injuries in the emergency department. CJEM. Sep 2010;12(5):389-96. doi:10.1017/s1481803500012537
- Ting JY, Barns KJ, Holmes JL. Management of Ocular Trauma in Emergency (MOTE) Trial: a pilot randomized double-blinded trial comparing topical amethocaine with saline in the outpatient management of corneal trauma. J Emerg Trauma Shock. Jan 2009;2(1):10-4. doi:10.4103/0974-2700.44676
- Waldman N, Densie IK, Herbison P. Topical tetracaine used for 24 hours is safe and rated highly effective by patients for the treatment of pain caused by corneal abrasions: a double-blind, randomized clinical trial. Acad Emerg Med. Apr 2014;21(4):374-82. doi:10.1111/acem.12346
- Tintinalli JE, Kelen GD, Stapczynski JS. Emergency Medicine: A Comprehensive Study Guide. 5th edition. McGraw-Hill. 2000.
- Wu G. Ophthalmology for Primary Care. WB Saunders; 1997.
- Turner A, Rabiu M. Patching for corneal abrasion. Cochrane Database Syst Rev. Apr 19 2006;(2):CD004764. doi:10.1002/14651858.CD004764.pub2
- Arbour JD, Brunette I, Boisjoly HM, Shi ZH, Dumas J, Guertin MC. Should we patch corneal erosions? Arch Ophthalmol. Mar 1997;115(3):313-7. doi:10.1001/archopht.1997.01100150315001
- Donnenfeld ED, Selkin BA, Perry HD, et al. Controlled evaluation of a bandage contact lens and a topical nonsteroidal anti-inflammatory drug in treating traumatic corneal abrasions. Ophthalmology. Jun 1995;102(6):979-84. doi:10.1016/s0161-6420(95)30926-8
- Kirkpatrick JN, Hoh HB, Cook SD. No eye pad for corneal abrasion. Eye (Lond). 1993;7 (Pt 3):468-71. doi: 10.1038/eye.1993.95
- Wilson SA, Last A. Management of corneal abrasions. Am Fam Physician. Jul 01 2004;70(1):123-8.
- Mohammadpour M, Shakoor D, Hashemi H, Aghaie Meybodi M, Rajabi F, Hosseini P. Comparison of bandage contact lens removal on the fourth versus seventh postoperative day after photorefractive keratectomy: a randomized clinical trial. J Curr Ophthalmol. Jun 2017;29(2):103-107. doi:10.1016/j.joco.2016.08.008
- Sun YZ, Guo L, Zhang FS. Curative effect assessment of bandage contact lens in neurogenic keratitis. Int J Ophthalmol. 2014;7(6):980-3. doi:10.3980/j.issn.2222-3959.2014.06.12
- Baradaran-Rafii A, Eslani M, Haq Z, Shirzadeh E, Huvard MJ, Djalilian AR. Current and upcoming therapies for ocular surface chemical injuries. Ocul Surf. Jan 2017;15(1):48-64. doi:10.1016/j.jtos.2016.09.002
- Inamoto Y, Sun YC, Flowers ME, et al. Bandage soft contact lenses for ocular graft-versus-host disease. Biol Blood Marrow Transplant. Nov 2015;21(11):2002-7. doi:10.1016/j.bbmt.2015.07.013
- Grentzelos MA, Plainis S, Astyrakakis NI, et al. Efficacy of 2 types of silicone hydrogel bandage contact lenses after photorefractive keratectomy. J Cataract Refract Surg. Dec 2009;35(12):2103-8. doi:10.1016/j.jcrs.2009.07.015
- Edwards JD, Bower KS, Sediq DA, et al. Effects of lotrafilcon A and omafilcon A bandage contact lenses on visual outcomes after photorefractive keratectomy. J Cataract Refract Surg. Aug 2008;34(8):1288-94. doi:10.1016/j.jcrs.2008.04.024
- Aquavella JV, Shaw EL. Hydrophilic bandages in penetrating keratoplasty. Ann Ophthalmol. Oct 1976;8(10):1207-19.
- Chen X, Yuan R, Sun M, Lin S, Ye J, Chen C. Efficacy of an ocular bandage contact lens for the treatment of dry eye after phacoemulsification. BMC Ophthalmol. Jan 09 2019;19(1):13. doi:10.1186/s12886-018-1023-8
- Shi DN, Song H, Ding T, Qiu WQ, Wang W. Evaluation of the safety and efficacy of therapeutic bandage contact lenses on post-cataract surgery patients. Int J Ophthalmol. 2018;11(2):230-234. doi:10.18240/ijo.2018.02.08
- Lim L, Lim EWL. Therapeutic contact lenses in the treatment of corneal and ocular surface diseases-a review. Asia Pac J Ophthalmol (Phila). Dec 2020;9(6):524-532. doi:10.1097/APO.0000000000000331
- Salz JJ, Reader AL, Schwartz LJ, Van Le K. Treatment of corneal abrasions with soft contact lenses and topical diclofenac. J Refract Corneal Surg. 1994;10(6):640-6.
- Buglisi JA, Knoop KJ, Levsky ME, Euwema M. Experience with bandage contact lenses for the treatment of corneal abrasions in a combat environment. Mil Med. Apr 2007;172(4):411-3. doi:10.7205/milmed.172.4.411
- Upadhyay MP, Karmacharya PC, Koirala S, et al. The Bhaktapur eye study: ocular trauma and antibiotic prophylaxis for the prevention of corneal ulceration in Nepal. Br J Ophthalmol. Apr 2001;85(4):388-92. doi:10.1136/bjo.85.4.388
- Dollery W. Towards evidence based emergency medicine: best BETs from the Manchester Royal Infirmary. Antibiotics and corneal abrasion. J Accid Emerg Med. Sep 1998;15(5):352. doi:10.1136/emj.15.5.352
- Szczotka-Flynn LB, Imamura Y, Chandra J, et al. Increased resistance of contact lens-related bacterial biofilms to antimicrobial activity of soft contact lens care solutions. Cornea. Sep 2009;28(8):918-26. doi:10.1097/ICO.0b013e3181a81835
- Lakkis C, Fleiszig SM. Resistance of Pseudomonas aeruginosa isolates to hydrogel contact lens disinfection correlates with cytotoxic activity. J Clin Microbiol. Apr 2001;39(4):1477-86. doi:10.1128/JCM.39.4.1477-1486.2001
- Szczotka-Flynn LB, Pearlman E, Ghannoum M. Microbial contamination of contact lenses, lens care solutions, and their accessories: a literature review. Eye Contact Lens. Mar 2010;36(2):116-29. doi:10.1097/ICL.0b013e3181d20cae
- McLaughlin-Borlace L, Stapleton F, Matheson M, Dart JK. Bacterial biofilm on contact lenses and lens storage cases in wearers with microbial keratitis. J Appl Microbiol. May 1998;84(5):827-38. doi:10.1046/j.1365-2672.1998.00418.x
- Mather R, Karenchak LM, Romanowski EG, Kowalski RP. Fourth generation fluoroquinolones: new weapons in the arsenal of ophthalmic antibiotics. Am J Ophthalmol. Apr 2002;133(4):463-6. doi:10.1016/s0002-9394(02)01334-x
- Parmar P, Salman A, Kalavathy CM, et al. Comparison of topical gatifloxacin 0.3% and ciprofloxacin 0.3% for the treatment of bacterial keratitis. Am J Ophthalmol. Feb 2006;141(2):282-286. doi:10.1016/j.ajo.2005.08.081
- Asbell PA, Colby KA, Deng S, et al. Ocular TRUST: nationwide antimicrobial susceptibility patterns in ocular isolates. Am J Ophthalmol. Jun 2008;145(6):951-958. doi:10.1016/j.ajo.2008.01.025
- Deramo VA, Lai JC, Fastenberg DM, Udell IJ. Acute endophthalmitis in eyes treated prophylactically with gatifloxacin and moxifloxacin. Am J Ophthalmol. Nov 2006;142(5):721-5. doi:10.1016/j.ajo.2006.05.044
- Hwang DG. Fluoroquinolone resistance in ophthalmology and the potential role for newer ophthalmic fluoroquinolones. Surv Ophthalmol. Mar 2004;49 Suppl 2:S79-83. doi:10.1016/j.survophthal.2004.01.004
- Ramos MS, Xu LT, Singuri S, et al. Patient-reported complications after intravitreal injection and their predictive factors. Ophthalmol Retina. Jul 2021;5(7):625-632. doi:10.1016/j.oret.2020.09.024
- Miller A, Wilneff MA, Yazji A, et al. Analysis of urgent follow up visits and complications after intravitreal injections: a retrospective cohort study. Int J Retina Vitreous. Jan 18 2022;8(1):8. doi:10.1186/s40942-021-00358-w
- Williams GA. Intravitreal injections: health policy implications [Internet]. Jobson Medical Information LLC, Review of Ophthalmology. Available from: https://www.reviewofophthalmology.com/article/ivt-injections-health-policy-implications2014
- Hykin PG, Foss AE, Pavesio C, Dart JK. The natural history and management of recurrent corneal erosion: a prospective randomised trial. Eye (Lond). 1994;8 (Pt 1):35-40. doi:10.1038/eye.1994.6
- Suri K, Kosker M, Duman F, Rapuano CJ, Nagra PK, Hammersmith KM. Demographic patterns and treatment outcomes of patients with recurrent corneal erosions related to trauma and epithelial and bowman layer disorders. Am J Ophthalmol. Dec 2013;156(6):1082-1087.e2. doi:10.1016/j.ajo.2013.07.022
- Grixti A, Sadri M, Watts MT. Corneal protection during general anesthesia for nonocular surgery. Ocul Surf. Apr 2013;11(2):109-18. doi:10.1016/j.jtos.2012.10.003



