PEER REVIEWED
Neuroretinitis Due to Cat Scratch Disease
Anju Kanikunnel, OD, FAAO, and Sonali Singh, MD
Abstract
Neuroretinitis is an inflammatory condition that affects the optic nerve and the inner sensory retinal layers at the macula.1 Neuroretinitis can be an ocular manifestation of inflammatory or infectious conditions. Cat scratch disease is the most common etiology for infectious neuroretinitis.1,2 Cat scratch disease is self-resolving, and patients with infectious neuroretinitis secondary to cat scratch disease often have a good vision prognosis. This case report involves a presentation of unilateral optic disc edema as an ocular manifestation of cat scratch disease and reviews the treatment and management of patients with neuroretinitis.
Key Words: neuroretinitis, macular star, cat scratch disease, Bartonella henselae, fluorescein angiography, stellate maculopathy
Background
Neuroretinitis is the most common posterior segment complication of cat scratch disease. This condition presents as unilateral optic disc edema with macular exudation in a star-shaped pattern resulting in partial or complete stellate maculopathy. Macular star formation is usually seen within 2-4 weeks after the onset of optic disc edema.3 Cat scratch disease is the most common cause of infectious neuroretinitis. Serological evidence of cat scratch disease in patients with neuroretinitis was established in the late 20th century.4 Cat scratch disease is a chronic systemic bacterial infection that predominantly affects the lymphatic system, especially the lymph nodes around the head, neck and arms. It is a self-limiting condition; however, it can be life-threatening when the central nervous system is involved.5 Annually, approximately 22,000 cases of systemic cat scratch disease are estimated to occur in the United States, and approximately 2,200 patients are hospitalized for treatment.6,7
This report identifies a patient with optic disc edema and serous macular detachment as posterior segment findings of neuroretinitis due to cat scratch disease. The patient was treated with oral doxycycline 100 mg twice a day for 1 month, which resulted in complete vision recovery.
Case Description
A 55-year-old Hispanic male presented to the clinic complaining of “vision worsening in the right eye” with a dull frontal headache for 2 weeks. The patient reported longstanding reduced vision due to refractive amblyopia in the right eye of “20/100 since childhood.” He denied any symptoms of diplopia, amaurosis fugax, eye pain, recent weight loss, scalp tenderness, neck pain or jaw claudication. He also denied any recent history of tick bites. He reported having a fever of 101º Fahrenheit approximately 1 month ago but denied any other symptoms along with the fever. His blood pressure at presentation was 129/83 mmHg (right arm, sitting). His ocular history was unremarkable for trauma or ocular surgeries. The patient’s medical and family history was unremarkable and he denied taking any medications.
Best-corrected visual acuity (BCVA) measured 20/200 in the right eye and 20/20 in the left eye. Pupils were round and reactive with a grade 1 afferent pupillary defect (APD) in the right eye. Color vision testing with Hardy Rand Rittler pseudoisochromatic plates measured 4/12 in the right eye and 12/12 in the left eye. Results of extraocular motility testing, cover test and confrontation visual field testing were normal. Slit lamp examination was unremarkable. Intraocular pressure was 12 mmHg in each eye by Goldmann applanation tonometry. Dilated funduscopic examination of the right eye revealed a swollen elevated right optic nerve with obscuration of the optic disc margin that was worse along the temporal disc margin with associated neuroretinal rim hyperemia and hemorrhages. The cup-to-disc ratio of the right optic nerve was difficult to assess, and no evidence of Paton’s folds was seen. A small flame-shaped retinal hemorrhage was noted along the inferior temporal arcade in the right eye. Mild macular retinal pigment epithelial changes and retinal vessel tortuosity were also noted in the right eye (Figure 1A). Funduscopic examination of the left eye was unremarkable (Figure 1B).
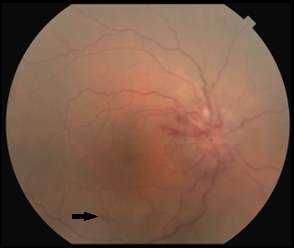 Figure 1A. Color fundus photograph of the right eye at initial presentation revealed obscuration of the optic disc margin, worse temporally, with associated neuroretinal rim hyperemia. No signs of Paton’s folds or overlying disc drusen were seen. A small flame-shaped retinal hemorrhage was seen along the inferior-temporal arcade (arrow). Click to enlarge |
 Figure 1B. Color fundus photograph of the left eye showed a normal optic nerve with distinct disc margin and normal macula. Click to enlarge |
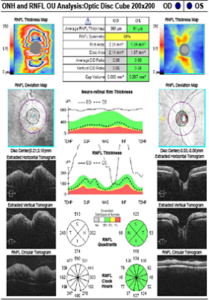 Figure 2A. OCT of the optic nerves showed increased thickness of the retinal nerve fiber layer in the right eye (368 μm) compared with the left eye (91 μm). Click to enlarge |
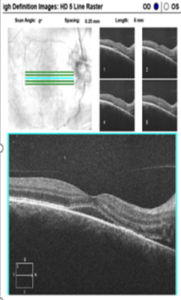 Figure 2B. HD OCT 5-line raster scan of the right macula showed serous macular detachment with areas of hyper-reflectivity in the inner sensory retinal layers. Click to enlarge |
Humphrey visual field central 30-2 threshold testing of the right and left eye was clear and reliable. Optical coherence tomography (OCT) of the peripapillary retinal nerve fiber layer showed significant thickening temporally consistent with the clinical appearance of optic disc edema (Figure 2A). OCT also confirmed the presence of serous macular detachment with scattered areas of hyper-reflectivity along the inner retinal layers (Figure 2B). B-scan ultrasonography of the optic disc was unremarkable for buried drusen of the optic nerve in the right and left eye.
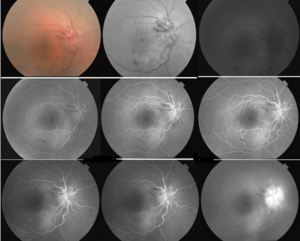
Figure 3. Fluorescein angiography of the right eye showed dye leaking at and around the optic nerve during the arteriovenous and late phases, which was suggestive of optic nerve head edema. No leakage was seen at the macula. Click to enlarge
Intravenous fluorescein angiography (FA) of the right eye showed leakage at and around the optic nerve head during the early and late phases. Higher fluorescein leakage during the late phase suggested optic nerve head edema (Figure 3).
Given the findings from the initial exam, the patient was diagnosed with optic disc edema with serous macular detachment due to unknown etiology OD. The differential diagnoses considered in the case at this point were:
- Malignant hypertension. Malignant hypertension (stage IV hypertensive retinopathy) presents with extremely elevated blood pressure typically greater than 140/90 mmHg. Retinal arteriole narrowing, copper- or silver-wire arteriole, arteriovenous crossing change, cotton wool spots, micoraneurysms, flame-shaped hemorrhages, hard exudates, macular star (acute) and Elschnig spots are seen on clinical examination.8,9
- Idiopathic intracranial hypertension. Due to elevated intracranial pressure, idiopathic intracranial hypertension can lead to bilateral optic disc edema, i.e., papilledema. It is commonly seen in overweight young females. A computerized tomography (CT) scan or magnetic resonance imaging (MRI) and lumbar puncture aid in the diagnosis.8
- Leber’s hereditary optic neuropathy (LHON). This condition presents initially as acute unilateral vision loss usually in males age 15 to 50. Contralateral eye involvement occurs within a week to a month in these patients.9,10 Because the condition is due to mitochondrial DNA mutation, a family history of vision loss or Leber’s optic neuropathy among maternal family members is seen.10 Visual acuity ranges from 20/100 to count fingers with an APD, abnormal color vision, reduced contrast sensitivity and subnormal electroretinography results.10 Visual field testing shows bilateral cecocentral or central scotoma. In LHON, the optic nerve may appear normal or pseudo-edematous. No leakage is seen at the optic nerve head on FA.1,9 Enhancement of the optic nerve and optic chiasm is seen on MRI.10 Visual prognosis in these patients is poor, and patients are considered legally blind due to severe bilateral visual field deficits.9,10
- Metabolic or toxic optic neuropathy. Metabolic or toxic optic neuropathy is often bilateral and presents as progressive painless vision loss due to poor nutrition or pernicious anemia. Certain medications, such as antimicrobials, immune modulators and suppressants, chemotherapy drugs, ethambutol, digitalis and chloroquine, can also play a role.11,12 Visual field testing shows bilateral cecocentral or central scotoma in these patients.
- Vascular etiologies. Vascular etiologies considered in this case included nonarteritic anterior ischemic optic neuropathy (NAION), arteritic anterior ischemic optic neuropathy (AION) and perioperative anterior ischemic optic neuropathy (PION). NAION typically presents in patients older than 50 years as acute painless vision loss usually upon waking. Patients often have a small cup-to-disc ratio (disc at risk) with an underlying systemic condition such as hypertension, diabetes or other vascular condition.9,13 An inferior altitudinal defect is the most common visual field defect seen, and patients have a poor visual prognosis. AION is commonly seen in patients older than 70 years. It is an acute unilateral painless vision loss due to giant cell arteritis.13 Patients experience headaches, jaw claudication, scalp tenderness, fatigue or weight loss. Erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) are significantly elevated. PION is acute, often unilateral, vision loss due to systemic hypotension or significant blood loss during head, neck, cardiac or spinal surgery, but it can be bilateral.
- Neoplastic and compressive etiologies. Potential neoplastic and compressive etiologies include conditions such as optic nerve glioma, optic nerve sheath meningioma or intracranial mass.14 They present as progressive vision loss or visual deficits that respect the vertical midline with an APD. The optic nerve could appear normal, pale or edematous. MRI of the brain and orbits are warranted.
- Inflammation. Potential inflammatory etiologies in this case included demyelinating diseases such as multiple sclerosis (MS). MS presents with acute unilateral often painful vision loss that worsens within 2 weeks.15 It is often seen in patients age 18 to 40 years. Visual field testing reveals a central scotoma. MRI shows an increased optic nerve signal and white matter lesions in the brain.15 MS patients have a good vision prognosis of 20/40 visual acuity or better. Ocular clinical findings in other inflammatory conditions, such as sarcoidosis, include bilateral optic neuropathy, conjunctival nodules, bilateral anterior granulomatous uveitis, sheathing over retinal veins (vasculitis and the appearance of “candle wax drippings”), neovascularization and cystoid macular edema.14 These patients have an elevated serum angiotensin converting enzyme (ACE) level and pulmonary involvement (chest X- ray).
- Infectious causes. Infectious etiologies considered in this case included syphilis, Lyme disease, tuberculosis (TB) and cat scratch disease. Syphilis can be either congenital or sexually transmitted. It is caused by the spirochete Treponema pallidum.16 Syphilitic optic neuropathy is often seen in stage 3 (secondary) and stage 5 (tertiary) of the disease.16 A macular star may be present. These patients have positive fluorescent treponemal antibody absorption (FTA-ABS) and rapid plasma reagent (RPR) (in untreated cases) test results.16,17 Lyme disease is caused by the spirochete Borrelia burgdorferi, which is transmitted through a tick bite. Some of the ocular signs of Lyme disease are conjunctivitis, uveitis, vitritis and optic neuropathy.16 TB is caused by Mycobacterium tuberculosis. Some of the ocular signs of TB are conjunctival nodules, granulomatous uveitis and optic neuropathy.9,16 This condition primarily affects the lungs; therefore, a chest X-ray and purified protein derivative panel (PPD) aid in diagnosis.16 Cat scratch disease is the most common cause of neuroretinitis.3,6 Serology testing with a Bartonella panel (anti-Bartonella henselae IgG, IgM) help confirm the diagnosis.4
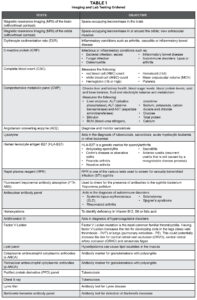
Table 1. Click to enlarge
In consultation with a neuro-ophthalmologist and retina specialist, a comprehensive workup for potential systemic and neurological etiologies of the patient’s optic nerve edema was performed. The workup included MRI of the brain and orbits with contrast. Laboratory studies included ESR, CRP, complete blood count, comprehensive metabolic panel, ACE, lysozyme, human leukocyte antigen B27, RPR, (FTA-ABS), antinuclear antibody panel, homocysteine, antithrombin III, factor V Leiden, serum protein, lipid panel, cytoplasmic antineutrophil cytoplasmic antibodies, perinuclear antineutrophil cytoplasmic antibodies, PPD panel, chest X-ray, Lyme and B. henselae antibody panel (Table 1).
Follow-up #1 (5 days)
At his 5-day follow-up visit the patient reported slight improvement in his headaches but no changes in his vision since his last exam. BCVA remained 20/200 in the right eye and 20/20 in the left eye. Dilated fundus examination of the right eye showed slight improvement of the optic disc edema with more visible, yet still obscured, disc margins and no signs of improvement in the serous macular detachment compared with baseline. Evaluation of the macula of the right eye also showed possible macular star formation with a few new retinal hemorrhages and hard exudates inferior-temporal to the fovea. The patient was asked about any recent history of cat scratch and he reported having several flea bites while he was cleaning under his trailer more than a month ago. He also reported that his wife had brought home six stray kittens around the same time.
Results of the comprehensive workup revealed no neurological abnormalities that would account for optic nerve edema. MRI of the brain and orbits were unremarkable with no space-occupying lesions or areas of abnormal enhancement. The results of all serological studies were normal, except for a positive finding of B. henselae.
The diagnosis of neuroretinitis in the right eye secondary to cat scratch disease was made based on the findings of positive B. henselae serology, history of exposure to feral kittens and flea bites. The patient was started on oral doxycycline 100 mg twice a day for 4 weeks and instructed to return in 4 to 5 weeks for a follow-up visit.
Follow-up #2 (1 month)
At this visit the patient’s BCVA was 20/100 in the right eye and 20/20 in the left eye. Dilated fundus examination showed marked improvement of the optic disc edema and no active serous macular detachment. Evaluation of the macula of the right eye still showed a few hard exudates at the fovea (Figure 4). The patient reported that his headaches had resolved, and his right eye’s vision had returned to “what it used to be.”
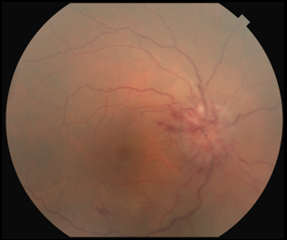 Figure 4A. Color fundus photography of the right eye at initial presentation revealed a blurred optic disc margin, worse along the temporal disc margin, and associated neuroretinal rim hyperemia. No signs of Paton’s folds or overlying disc drusen were seen. A small flame-shaped retinal hemorrhage was seen along the inferior temporal arcade. Click to enlarge |
 Figure 4B. Color fundus photography of the right eye after 4 weeks of treatment with oral doxycycline showed significant improvement in optic disc edema and the presence of a few hard exudates at the macula. Click to enlarge |
Education Guidelines
Learning objectives
- Become familiar with the ocular presentation of neuroretinitis due to cat scratch disease
- Become familiar with ophthalmic testing, neuroimaging and laboratory testing to be ordered for patients presenting with unilateral optic disc edema
- Become familiar with appropriate lab testing and imaging needed in cases of optic disc edema to rule out any life-threatening conditions
Key concepts
- Ocular manifestations of cat scratch disease
- Criteria for an accurate diagnosis of cat scratch disease
- Laboratory investigation needed to confirm the diagnosis of cat scratch disease
- Treatment and management of cat scratch disease
Discussion questions
- What is neuroretinitis and its most common cause?
- What are the signs and symptoms of cat scratch disease?
- What are the ocular manifestations of cat scratch disease?
- What findings are necessary for making an accurate diagnosis of cat scratch disease?
- What systemic treatment is indicated for patients with neuroretinitis due to cat scratch disease?
Discussion
What is neuroretinitis and its most common cause?
Neuroretinitis is inflammation of the optic nerve and inner sensory retinal layers at the macula resulting in stellate maculopathy. It is most commonly caused by cat scratch disease.3 The bacterium that causes this condition, B. henselae, has been identified in specimens taken from the lymph nodes of patients with cat scratch disease. B. henselae is an aerobic, oxidase-negative, gram-negative rod bacterium.4,6 In humans B. henselae invades the vascular endothelium initiating an acute inflammatory reaction resulting in lymphadenopathy and visceral organ involvement.7 It is not known why some patients develop only a localized infection while others develop a more widespread systemic infection.4 Cats are the primary carriers of B. henselae and it is prevalent in young felines, feral felines and flea-infected felines. The prevalence of the infection is greater in cats in warmer regions with higher annual rainfall because fleas thrive in those environmental conditions.3,18,19 The feline flea, Ctenocephalides felis, is involved in the transmission of B. henselae from cat to cat.4,6,7 The disease is transmitted to humans either through a cat scratch, cat bite or flea bite. Flea feces can transmit the disease via direct inoculation of open wounds or mucous membranes such as the conjunctiva or oral mucosa.2,18 There is no racial predilection for cat scratch disease and it commonly affects children and young adults (under age 21). Recurrence of cat scratch disease is uncommon. Cases in which several members of the same family were affected have been reported.2 Due to occupational exposure, veterinarians have a higher risk for this infection.3,18
What are the signs and symptoms of cat scratch disease?
Symptoms occur within 1 to 2 weeks of the cat scratch, cat bite or flea bite. Common symptoms include redness and swelling of the skin, swollen lymph nodes around the head, neck and arms, fever, headaches, fatigue and loss of appetite.2 Cat scratch disease primarily affects the lymph nodes along the head, neck and arm, but the disease can affect the skin or cutaneous membrane near the inoculation site, visceral organs such as liver and spleen, the eyes, and the musculoskeletal and central nervous systems.5,20
What are the ocular manifestations of cat scratch disease?
Ocular manifestations are the second most common clinical presentation of cat scratch disease after lymphatic involvement.1 The most common ocular signs of cat scratch disease are Parinaud’s oculoglandular syndrome and neuroretinitis. Neuroretinitis occurs in approximately 2% of patients with cat scratch disease.1
Parinaud’s oculoglandular syndrome is the most common ocular finding in symptomatic cat scratch disease. Patients present with symptoms of unilateral red eye, discomfort, mild lid swelling and epiphora with serous discharge. In rare cases, purulent discharge is seen with the formation and rupture of abscess.2,19 Clinical signs of Parinaud’s oculoglandular syndrome consist of unilateral granulomatous conjunctivitis with regional lymphadenopathy involving the preauricular, submandibular or cervical lymph nodes.4,16 Conjunctival lesions consist of granulomatous nodules with an ulcerative epithelium and necrotic center involving either the bulbar or palpebral conjunctiva.2,16 Although the etiology of Parinaud’s oculoglandular syndrome is not fully understood, it is believed that direct inoculation of the conjunctiva with infected flea feces may be the route of infection.2
Neuroretinitis is the most common posterior segment complication of cat scratch disease, and cat scratch disease is the most common cause of infectious neuroretinitis.1,17 Infectious neuroretinitis is characterized by unilateral optic disc edema followed by formation of partial or complete macular star in 2 to 4 weeks.20 Formation of intraretinal or subretinal macular exudates (macular star) is usually seen in cases with severe optic disc swelling.19,21
Less common ocular manifestations are anterior and intermediate uveitis, vitritis, retinal vasculitis and a variety of multifocal retinochoroiditis findings.1 Multifocal retinochoroiditis manifestations include retinal white dot syndrome, retinal artery occlusions, retinal vein occlusions, focal choroidal infiltrates and subretinal angiomatous mass lesions.19
What findings are necessary for an accurate diagnosis of cat scratch disease?
Although signs and symptoms aid in the diagnosis of cat scratch disease, accurate diagnosis must include two or three of these findings:19
- history of cat exposure, cat scratch or bite or flea bite regardless of an inoculation site lesion2
- positive serology for B. henselae (IFA: indirect fluorescence assay or EIA: enzyme immunoassay) with a titer ratio ≥ 1:644,6
- positive Bartonella polymerase chain reaction assay, negative serology for other causes of adenopathy, sterile pus aspirated from a lymph node, and/or liver or spleen lesions seen on CT scan4
- positive Warthin-Starry silver stain or biopsy showing granulomatous inflammation consistent with cat scratch disease2,4,6
The patient in this case met criteria 1 and 2. He reported a recent history of flea bite with exposure to stray kittens, and his testing confirmed a positive serology result.
IFA serology is used in detecting anti-B. henselae IgG and is considered the gold standard in detecting antibodies to B.henselae.6 It has high specificity but low sensitivity. This test also has significant cross-reactivity between B. henselae and Bartonella quintana for IgG assays.4 IFA IgG titers < 1:64 indicate a negative Bartonella infection or a past infection. IFA IgG titers between 1:64 and 1:256 indicate a possible infection and repeat testing is recommended within 10 to 14 days. IFA IgG titers > 1:256 indicate an active infection.4, 6 EIA is used in detecting anti-B. henselae IgG and IgM. A positive IgM test indicates acute disease.6 The patient in this case had a B. henselae IgG titer > 1:2560 and a positive B.henselae IgM titer of 1:200 (normal is 1:100). He also had negative B. quintana IgG and IgM titers.
What systemic treatment is indicated for patients with neuroretinitis due to cat scratch disease?
Cat scratch disease is a self-limiting condition and most patients with the typical systemic manifestation of the disease have a gradual resolution of symptoms without any antimicrobial therapy.22,23 As long as the immune system is intact, in the mild to moderate stages of the disease, patients weighing more than 100 pounds are treated with 500 mg of azithromycin for the first day then 250 mg for the next 4 days. Patients weighing less than 100 pounds (typically children) are treated with 10 mg/kg of azithromycin for the first day then 5 mg/kg for the next 4 days.19,23 Clarithromycin, rifampin, trimethoprim-sulfamethoxazole or ciprofloxacin can be used for patients intolerant to azithromycin.23
Cat scratch disease with liver and spleen involvement is typically treated with either azithromycin or gentamicin and rifampin for 10 to 14 days. Gentamicin dosing consists of an intravenous loading dose of 2 mg/kg then 1.5 mg/kg every 8 hours depending on normal renal function.2 Rifampin oral dosage consists of 300 mg twice a day in adults and 10 mg/kg every 12 hours with a maximum dose of 600 mg daily in children.2,3 Because doxycycline has better central nervous system penetration, it is typically used to treat patients older than 12 years with neuroretinitis and neurological manifestation. Doxycycline oral dosing consists of 100 mg twice a day for 4 to 6 weeks.2,22 In the severe stages of the disease patients are given intravenous antibiotics with or without oral rifampin for 2 to 4 weeks.2,22
Neuroretinitis is a self-limiting condition and treatment with antibiotics remains controversial.2 During the initial presentation of the condition, patients should be co-managed with a neuro-ophthalmologist and followed weekly with a dilated fundus examination until etiology for the neuroretinitis is determined. The patient can be followed every 3 to 4 weeks until resolution and then every 6 months or yearly thereafter.2,3
Several measures can be taken to prevent the spread and transmission of cat scratch disease. As irresistible as they are for many humans, avoiding handling or playing with felines can reduce the chances of a bite or scratch. It is important to immediately wash and disinfect any cat scratch or bite. Treating cats with topical parasiticides such as selamectin or dinotefuran/pyriproxyfen can prevent flea infestations and thereby prevent transmission of B. henselae from fleas to cats.7
Conclusion
Cat scratch disease is an important differential diagnosis in cases of unilateral optic disc edema. It is important to obtain a thorough case history and inquire about any recent flea bite, cat scratch or exposure to stray cats in addition to tick bites. In a patient presenting with unilateral disc edema, it is also important to rule out the numerous differential diagnoses for both unilateral and bilateral optic disc edema. Timely and appropriate lab testing and imaging need to be ordered to rule out possible life-threatening conditions. Cat scratch disease is a self-resolving condition and patients with infectious neuroretinitis secondary to cat scratch disease often have a good vision prognosis.
In summary, this patient’s BCVA at resolution was measured at 20/100 in the right eye due to refractive amblyopia. Findings from OCT and FA helped confirm optic disc edema. Macular star formation was not noted on the initial visit, but hard macular exudates were prominent in the follow-up visits. This raised the suspicion for infectious optic disc edema as the primary etiology for the ocular findings. However, it is important to rule out life-threatening causes of optic disc edema with MRI of the brain and orbits and other etiologies with necessary labs. The patient in this case showed slight improvement in clinical signs and symptoms before treatment was initiated. Per the recommendation of the on-staff neuro-ophthalmologist, the patient was treated with oral doxycycline 100 mg twice a day for 1 month.
References
- Ormerod LD, Skolnick KA, Menosky MM, Pavan PR, Pon DM. Retinal and choroidal manifestations of cat-scratch disease. Ophthalmology. 1998 Jun;105(6):1024-31. doi: 10.1016/S0161-6420(98)96003-1.
- Cunningham ET, Koehler JE. Ocular bartonellosis. Am J Ophthalmol. 2000 Sep;130(3):340-9. doi: 10.1016/s0002-9394(00)00573-0.
- Chrousos GA, Drack AV, Young M, Kattah J, Sirdofsky M. Neuroretinitis in cat scratch disease. J Clin Neuroophthalmol. 1990 Jun;10(2):92-4.
- Gulati A, Yalamanchili S, Golnik KC, Lee AG. Cat scratch neuroretinitis: the role of acute and convalescent titers for diagnosis. J Neuroophthalmol. 2012 Sep;32(3):243-5. doi: 10.1097/WNO.0b013e318233a0a6.
- Brazis PW, Stokes HR, Ervin FR. Optic neuritis in cat scratch disease. J Clin Neuroophthalmol. 1986 Sep;6(3):172-4.
- Suhler EB, Lauer AK, Rosenbaum JT. Prevalence of serologic evidence of cat scratch disease in patients with neuroretinitis. Ophthalmology. 2000 May;107(5):871-6. doi: 10.1016/s0161-6420(00)00002-6.
- Zangwill KM, Hamilton DH, Perkins BA, et al. Cat scratch disease in Connecticut. Epidemiology, risk factors, and evaluation of a new diagnostic test. N Engl J Med. 1993 Jul;329(1):8-13. doi: 10.1056/NEJM199307013290102.
- Heidary G, Rizzo JF 3rd. Use of optical coherence tomography to evaluate papilledema and pseudopapilledema. Semin Ophthalmol. 2010 Sep-Nov;25(5-6):198-205. doi: 10.3109/08820538.2010.518462.
- Alexander L. Primary care of the posterior segment. 3rd ed. New York: McGraw-Hill Professional; 2002.
- Howell N. LHON and other optic nerve atrophies: the mitochondrial connection. Dev Ophthalmol. 2003;37:94-108. doi: 10.1159/000072041.
- Kerrison JB. Optic neuropathies caused by toxins and adverse drug reactions. Ophthalmol Clin North Am. 2004 Sep;17(3):481-8; viii. doi: 10.1016/j.ohc.2004.05.005.
- Sadun AA. Metabolic optic neuropathies. Semin Ophthalmol. 2002 Mar;17(1):29-32. doi: 10.1076/soph.17.1.29.10290.
- Hayreh SS, Zimmerman MB. Optic disc edema in non-arteritic anterior ischemic optic neuropathy. Graefes Arch Clin Exp Ophthalmol. 2007 Aug;245(8):1107-21. doi: 10.1007/s00417-006-0494-0.
- Chan JW. Optic nerve disorders. 2nd ed. New York: Springer Publishing; 2014.
- The clinical profile of optic neuritis. Experience of the Optic Neuritis Treatment Trial. Optic Neuritis Study Group. Arch Ophthalmol. 1991 Dec;109(12):1673-8. doi: 10.1001/archopht.1991.01080120057025.
- Golnik KC. Infectious optic neuropathy. Semin Ophthalmol. 2002 Mar;17(1):11-7. doi: 10.1076/soph.17.1.11.10293.
- Ghauri RR, Lee AG. Optic disk edema with a macular star. Surv Ophthalmol. 1998 Nov-Dec;43(3):270-4. doi: 10.1016/s0039-6257(98)00038-1.
- Berger JR. The interface of infectious disease and neuro-ophthalmology. J Neuroophthalmol. 2012 Sep;32(3):195-6. doi: 10.1097/WNO.0b013e318263e275.
- Reed JB, Scales DK, Wong MT, Lattuada CP Jr, Dolan MJ, Schwab IR. Bartonella henselae neuroretinitis in cat scratch disease. Diagnosis, management, and sequelae. Ophthalmology. 1998 Mar;105(3):459-66. doi: 10.1016/S0161-6420(98)93028-7.
- Purvin V, Sundaram S, Kawasaki A. Neuroretinitis: review of the literature and new observations. J Neuroophthalmol. 2011 Mar;31(1):58-68. doi: 10.1097/WNO.0b013e31820cf78a.
- Longmuir RA, Lee A. Cat-scratch neuroretinitis (ocular bartonellosis): 44-year-old female with non-specific “blurriness” of vision, left eye (OS) [Internet]. Iowa City, IA: EyeRounds.org., University of Iowa; 2005 Mar 31 [cited 2017 Apr 17]. Available from: http://webeye.ophth.uiowa.edu/eyeforum/cases/36-catscratchbartonella.htm.
- Bhatti MT, Lee MS. Should patients with bartonella neuroretinitis receive treatment? J Neuroophthalmol. 2014 Dec;34(4):412-6. doi: 10.1097/WNO.0000000000000166.
- Rolain JM, Brouqui P, Koehler JE, Maguina C, Dolan MJ, Raoult D. Recommendations for treatment of human infections caused by Bartonella species. Antimicrob Agents Chemother. 2004 Jun;48(6):1921-33. doi: 10.1128/AAC.48.6.1921-1933.2004.



