Peripapillary Retinoschisis and Glaucoma Connection
Diana Mah, OD, FAAO, and Kristin Richwine, OD, FAAO
Abstract
Peripapillary and macular retinoschisis as well as serous retinal detachment are often associated with optic nerve head pits, colobomas, X-linked macular schisis and myopia. This report describes a case of peripapillary schisis and serous retinal detachment in a patient with advanced untreated open angle glaucoma but no other pathology. These findings may be unusual sequelae in patients with glaucomatous optic neuropathy. Although several case studies have been reported in the literature, the mechanism behind this disease process is not well-understood. Clinicians need to be aware of this atypical finding in order to manage the conditions appropriately.
Key Words: peripapillary schisis, serous retinal detachment, unusual sequelae, glaucomatous optic neuropathy
Background
Retinoschisis involving the peripapillary and macular regions is well-documented in the literature. The condition is commonly associated with optic nerve head pits, optic nerve colobomas, X-linked macular schisis and high myopia. Less often cited in literature are cases of retinoschisis associated with glaucoma or enlarged optic nerve cupping in the absence of other pathology, as described in this case.1-4,7 Although the underlying pathophysiology may not be completely understood, it is important to be aware of this atypical finding when managing glaucoma patients with retinoschisis because treatment options may differ from other causes of secondary retinoschisis. In addition, resolution of the retinoschisis can affect the presentation of glaucoma stability or progression. As discussed in this teaching case report, being cognizant of these co-existing conditions enables the optometrist to better manage patients.
This teaching case report highlights diagnostic tools and appropriate management of the patient with retinoschisis and glaucoma. It is intended for fourth-year optometry students actively involved in patient care. As this condition is not common to glaucoma-related findings, knowledge about the condition and the appropriate steps for management and treatment is essential for the practicing optometrist in any clinical setting.
Case Description
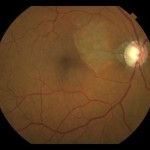
Figure 1A. Enlarged C/D and neurosensory retinal detachment temporal to the optic nerve head OD.
Figure 1A: click to enlarge
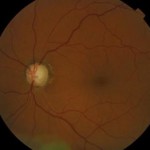
Figure 1B. Enlarged C/D secondary to glaucoma OS.
Figure 1B: click to enlarge
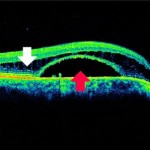
Figure 3. OCT showing retinoschisis (white arrow) and neurosensory retinal detachment (red arrow) OD.
Figure 3: click to enlarge
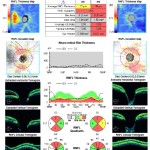
Figure 2. OCT of the retinal nerve fiber layer showing significant thinning OU. Note that the temporal quadrant OD appears thickened due to the macular schisis and neurosensory macular detachment.
Figure 2: click to enlarge
A 70-year-old Caucasian male presented to our clinic for an initial exam with the chief complaint of more difficulty with near vision over the past year. The patient had not had an eye exam in more than 25 years and was using over-the-counter reading glasses. His systemic history was positive for borderline hypertension and obesity. He was not taking any medications. His best-corrected visual acuity was 20/40 +2 OD (-0.50 -0.50 x 094) and 20/25 -2 OS (-0.75 -0.25 x 008). Slit lamp exam was remarkable for mild nuclear sclerotic and cortical cataracts OU. Intraocular pressure (IOP) was 20 mmHg OD and 20 mmHg OS at 2:40 p.m. Dilated fundus exam revealed a serous retinal detachment extending from the optic nerve towards the macula in the right eye (Figure 1A) and significant cupping with cup to disc ratio (C/D) of 0.8 OD and 0.9 OS (Figure 1B). Optical coherence tomography (OCT) of the optic nerve and macular region was performed OU (Figures 2 and 3).

Figure 4A. Humphrey 30-2 visual field test showing significant glaucomatous field loss OS.
Figure 4A: click to enlarge

Figure 4B. Humphrey 30-2 visual field test showing significant glaucomatous field loss OD.
Figure 4B: click to enlarge

Figure 5B. Decreased size and amplitude of retinoschisis and neurosensory retinal detachment OD after three years of treatment for glaucoma.
Figure 5B: click to enlarge
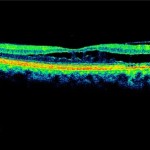
Figure 5A. Decreased size and amplitude of retinoschisis and neurosensory retinal detachment OD after eight months of treatment for glaucoma.
Figure 5A: click to enlarge
Based on the findings, the patient was diagnosed with advanced glaucomatous cupping OU and retinoschisis with neurosensory retinal detachment OD. The plan was to have the patient back in a week for an IOP check and visual field testing, and he was referred to the Retina Service for evaluation of the retinal findings. The patient was seen in the Retina Service two weeks later, and the retinoschisis and serous detachment were noted to be stable. IOP at that visit was 34 mmHg OD and 33 mmHg OS at 9:12 a.m. Due to the increased pressures and advanced cupping, treatment with travoprost ophthalmic solution 0.004% (Travatan Z) at night was initiated OU. The patient returned one month later for a follow-up and visual field testing (Figures 4A and 4B). The Humphrey 30-2 visual field test showed significant superior and inferior arcuate defects OU consistent with advanced glaucoma. Gonioscopy revealed wide-open angles with flat iris insertion OU. IOP on Travatan Z was 24 mmHg OD and 21 mmHg OS at 12:09 p.m. Due to the advanced field loss, dorzolamide/timolol (Cosopt) twice a day was added for both eyes. The patient returned one week later and IOP was 11 mmHg OD and 11 mmHg OS at 7:42 a.m.
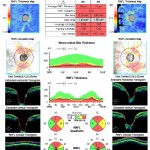
Figure 6. OCT of the retinal nerve fiber layer performed two years after initial presentation showing advanced thinning from glaucoma OU. Thickness in the temporal quadrant OD is decreased compared to the initial OCT due to resolution of the macular schisis and neurosensory retinal detachment.
Figure 6: click to enlarge

Figure 7B. Humphrey 10-2 OD performed 2.5 years after initial presentation showing significant but stable field loss encroaching fixation, OD>OS.
Figure 7B: click to enlarge

Figure 7A. Humphrey 10-2 OS performed 2.5 years after initial presentation showing significant but stable field loss encroaching fixation, OD>OS.
Figure 7A: click to enlarge
The patient continued to be followed over the next three years with pressure checks and dilated exams. Best-corrected visual acuity remained stable at 20/25- OD and 20/25+ OS. IOP remained consistently below 12 mmHg OU with Travatan Z at night and Cosopt twice a day OU. The schisis and sensory retinal detachment improved over time without any intervention except to control the intraocular pressure (Figures 5A and 5B). Two years after initial presentation, OCT of the retinal nerve fiber layer (RNFL) showed advanced thinning from glaucoma OU, but the temporal quadrant OD showed decreased thickness compared to the initial OCT due to resolution of the macular schisis and neurosensory retinal detachment (Figure 6). Humphrey visual fields OU remained severely depressed but stable over the next three years (Figures 7A and 7B).
Educator’s Guide
Key concepts
- Use of technology in diagnosing and documenting retinoschisis and sensory retinal detachment
- Conditions associated with retinoschisis
- Clinical findings and use of technology with glaucoma
- Treatment and management considerations with glaucoma and secondary retinoschisis
Learning objectives
At the conclusion of this case discussion, participants should be able to:
- Describe the signs and symptoms of different types of retinoschisis (i.e., inherited vs. acquired, presentation and location of each subtype)
- Understand the conditions commonly associated with the different types of retinoschisis
- Recognize the rare association between retinoschisis and glaucoma
- Describe typical visual field and OCT findings for glaucoma, primary retinoschisis and retinoschisis secondary to glaucoma
- Understand the natural course of the disease and appropriate management of glaucoma-related retinoschisis
- Provide proper patient education on management options and expectations following treatment
Discussion Questions
Retinoschisis
- Describe the clinical findings of retinoschisis
- Describe the different layers of the retina
- What conditions are commonly associated with retinoschisis
- Discuss the differential diagnosis for retinoschisis
- What ancillary testing can be used to confirm retinoschisis
- What is the natural course of retinoschisis
- What is the role of the primary care optometrist in the management of retinoschisis
Glaucoma
- What is the role of IOP in glaucoma
- Discuss risk factors for the development and progression of glaucoma
- Describe optic nerve head findings commonly seen in glaucoma
- What are typical visual field defects associated with glaucoma
- Discuss the differential diagnosis of glaucoma
- Describe the role of imaging (OCT, GDx, HRT) in diagnosing and following glaucoma
- Discuss the pharmaceutical treatment of glaucoma, including side effects and contraindications of medications
- What are various ways to determine glaucoma progression
- Discuss the staging of glaucoma and ICD-10 coding used for the various stages
Discussion
Only a handful of cases of a schisis and underlying serous detachment associated with glaucoma and optic nerve cupping in the absence of other pathology have been reported in the literature.1-4,7 The purpose of this case report is to help enable students to recognize the clinical findings of retinoschisis and glaucoma, identify the correlation between these co-existing diseases, become familiar with special testing to document and manage these diseases, and consider the appropriate treatment options.
Clinical findings and differential diagnosis of retinoschisis
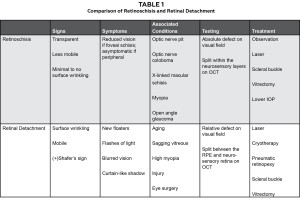
Retinoschisis is a splitting of the retina’s neurosensory layers, usually in the outer plexiform layer. Depending on the cause, it may be present in the macula, posterior pole or peripheral retina, most often inferior temporal retina. Depending on the location of the schisis, the patient may be asymptomatic or notice blurred vision or a scotoma. The primary differential diagnosis is a retinal detachment (Table 1). These conditions can be distinguished from each other clinically: a retinoschisis appears more transparent, is less mobile with less surface wrinkling, and has less underlying pigment epithelial atrophy than a full-thickness retinal detachment. In addition, the position of the cavity is constant and when the area is scleral depressed the inner retinal layer fails to collapse. If the student or clinician cannot determine whether the patient has a schisis or detachment, further diagnostic testing can be helpful. Visual field testing reveals an absolute defect with sharp borders in the case of a retinoschisis and a relative defect with a retinal detachment. OCT can also be used. In a retinal detachment, the separation of the layers will be between the neurosensory retina and the retinal pigment epithelium, whereas a schisis is a separation within the layers of the neurosensory retina.
Associated conditions
Retinoschisis and peripapillary serous retinal detachments are often seen in patients with congenital and acquired optic nerve pits, optic nerve colobomas, X-linked macular schisis and myopia (Table 2); therefore, these conditions need to be evaluated when a patient presents with retinoschisis and/or peripapillary serous retinal detachments. In optic disc pits, a small round hypopigmented or grey depression is seen funduscopically, typically on the temporal or central aspect of the nerve. These lesions are typically unilateral. Vision is often normal unless the pits are associated with a serous macular detachment. Optic nerve colobomas are congenital anomalies of the optic nerve resulting from incomplete closure of the embryonic fissure during development. These may be unilateral or bilateral and are typically accompanied by poor vision. The nerve is often large with an inferior defect and possible residual glial tissue. X-linked macular schisis is a retinal disorder associated with a variety of clinical findings. This condition usually occurs in males and results in impaired vision early in childhood. Foveal schisis is seen in almost all of these patients and appears as a stellate spoke-like maculopathy. OCT shows the split in the retina, and the split occurs across multiple layers.11 Clinically, pigmentary changes may occur at the macula over time as the schisis resolves. In addition, macular retinoschisis is not uncommon in highly myopic eyes with staphyloma.12 OCT shows the splitting of the retinal layers when this is present.12 Because this patient did not have this condition in childhood, did not show evidence of a spoke-like maculopathy, disc pit, nerve coloboma, or high myopia, it is presumed that the retinoschisis and serous detachment were secondary to his open angle glaucoma with elevated IOP.
Pathophysiology
The source of fluid and definitive pathogenesis of papillary schisis and detachment associated with optic disc anomalies, or in glaucomatous papillary detachments, as in this case, are unclear. One possible source of the fluid is the vitreous, which is thought to enter the optic disc through small holes or breaks in overlying membranes or neuroectoderm tissue.8 Vitreous traction may be associated with these breaks. Small changes in axial length accompanying IOP fluctuations may lead to vitreous traction in the inner retina that lead to schisis formation.3 In addition, animal studies have demonstrated that prolonged increase in IOP may lead to breaks in the inner limiting membrane with penetration of the vitreous into the retrolaminar space.4 It is also thought that fluctuations in IOP that lead to glaucoma-related structural defects in the nerve allow vitreous fluid to track through microholes in the thin tissue of the optic nerve leading to the schisis and serous retinal detachment.4,5 Fluid from the subarachnoid space may also be responsible. It has been suggested that the vitreous, subarachnoid and subretinal spaces may be at times interconnected because of the permeable nature of the optic nerve.2,10,14 A pressure differential must occur for fluid to move, and fluctuations in IOP can affect the pressure differential between the vitreous cavity and spaces outside the globe.8 Further studies need to be done to assess the mechanism behind this process but this is difficult given how few patients are typically seen.
Treatment/management
Once the student clinician achieves a diagnosis, he or she must determine whether the disease exceeds the treatment threshold. The decision to treat is influenced by consideration of whether the disease is vision- or life-threatening and the treatment benefits outweigh the risks. In this patient, significant glaucomatous cupping, elevated intraocular pressures and field loss warranted the initiation of pressure-lowering medication. Medical management of glaucoma consists of pressure-lowering medications to reduce aqueous production and/or improve aqueous outflow. Commonly used medications include topical beta blockers (e.g., timolol, betimol), alpha agonists (e.g., brimonidine), prostaglandin analogs (e.g., latanoprost, travoprost), carbonic anhydrase inhibitors (e.g. dorzolamide, brinzolamide) or a combination of these drugs. Factors determining the selection of drugs include ease of use, cost, side effects, contraindications, target pressure and severity of glaucoma. Many clinicians use a prostaglandin analog or timolol for first-line therapy. These medications typically reduce IOP by 30% and 25%, respectively, and are inexpensive to use.18
In this patient, the retinoschisis was deemed stable and not immediately vision-threatening. Typically in an asymptomatic and non-vision threatening retinoschisis, observation only in 6- to 24-month intervals is appropriate management as the condition is usually stable to slowly progressive for many years. Laser, scleral buckle or pars plana vitrectomy may be performed for vision-threatening retinoschisis.5,8,17 These procedures are not only invasive but also can lead to further complications and reduce patient quality of life. The peripapillary schisis reported here spontaneously resolved after glaucoma treatment was initiated. It is possible that lowering the IOP and minimizing fluctuations with topical glaucoma drops was responsible for this resolution as reported in similar cases in the literature.2,3,5,13 Therefore, if vision is not threatened, it may be prudent to lower IOP first and monitor closely for resolution of the retinal findings before considering more invasive strategies. When considering target pressures, pressures should be set according to visual field and/or optic nerve changes. Typical glaucomatous field patterns include arcuate defects that respect the horizontal meridian (e.g., Figure 4A), paracentral scotomas that are only part of the arcuate segment, and nasal step with a large sensitivity difference across the nasal horizontal meridian (e.g., Figure 7B). In addition, thinning of the peripapillary nerve fiber layer and ganglion cell complex can be estimated using OCT to support the diagnosis of glaucoma. However, it is crucial to correlate optic nerve findings with OCT measurements and visual field loss as structure should match function.
When assessing visual field and RNFL changes in patients with glaucoma and concurrent peripapillary retinoschisis, students should be aware that these patients will likely have nerve fiber layer changes that occur after resolution of the retinoschisis. When the retinoschisis is present, the RNFL appears falsely thick and sometimes will show a considerable rapid decrease after resolution which might lead one to believe that glaucoma has progressed.13 However, the visual field should not change in that area.17 This patient did show a decrease in RNFL thickness (Figures 2, 6) along with resolution of the retinoschisis without significant visual field progression. Therefore, when monitoring for progression of glaucoma, students should verify that a RNFL change on OCT corresponds with a change in visual field or in optic nerve appearance. In addition, retinoschisis associated with glaucoma may be overlooked because patients are often asymptomatic and retinoschisis can appear normal on fundoscopy alone. Therefore, if a sudden change in RNFL thickness is noted, consider ruling out an existing retinoschisis. If this cannot be confirmed, careful optic nerve examination as well as serial visual field testing should be used to confirm progression of glaucoma.
The assessment of the learning objectives for this case report can be accomplished in several ways. Students presented with fundus photos should be able to describe the photos, including normal and abnormal findings of the optic nerve, macula and retina. Once abnormal findings are identified, students should be queried on what additional testing should be performed to determine the underlying disease and any associated conditions. In this case, OCT scans were important in the diagnosis and management, so students can be evaluated for their knowledge of the different layers of the retina in a normal scan and be able to describe abnormal scans. Furthermore, students should also be assessed on their ability to describe abnormal visual fields and prescribe appropriate medical treatment for glaucoma. Proper patient education and management expectations can be accomplished by role-playing.
Conclusion
In rare instances, glaucoma may lead to peripapillary retinoschisis and underlying serous retinal detachment in the absence of other conditions mentioned in the differential diagnosis. Although the mechanism by which this occurs is not fully understood, awareness that this is a potential cause of secondary retinoschisis with serous detachment can lead to better patient outcomes. Students should be aware that these patients can be treated conservatively, by initiating glaucoma treatment and minimizing IOP fluctuations, rather than referred for potential invasive surgical intervention.
References
- Zhao M. Macular retinoschisis associated with normal tension glaucoma. Graefes Arch Clin Exp Ophthalmol. 2011;249(8):1255-8.
- Farjad H. Peripapillary schisis with serous detachment in advanced glaucoma. Optom Vis Sci. 2010;87(3):E205-17.
- Kahook M. Peripapillary schisis in glaucoma with narrow angles and increased intraocular pressure. Am J Ophthalmol. 2007;143:697-699.
- Hollander D. Macular schisis detachment associated with angle closure glaucoma. Arch Ophthalmol. 2005;123(2):270-2.
- Zumbro DS. Macular schisis and detachment associated with presumed acquired enlarged optic nerve head cups. Am J Ophthalmol. 2007;144(1):70-74.
- Pokroy R. Bilateral optic pit like maculopathy with normal optic nerve heads. Can J Ophthalmol. 2010;45(4):415-6.
- Mavrikakis E. Macular schisis and detachment secondary to large optic nerve head cup: a newly recognized syndrome amenable to vitrectomy. Acta Ophthalmol. 2011;89(1):95-6.
- Johnson TM, Johnson MW. Pathogenic implications of subretinal gas migration through pits and atypical colobomas of the optic nerve. Arch Ophthalmol. 2004;122:1793-1800.
- Rhee D, Pyfer M. The Wills Eye Manual third edition. Philadelphia, PA: LWW, 1999.
- Irvine AR, Crawford, JB, Sullivan JH. The pathogenesis of retinal detachment with morning glory disc and optic pit. Retina. 1986;6:146-150.
- Manschot WA. Pathology of hereditary juvenile retinoschisis. Arch Ophthal. 1972;88(2):131-138.
- Benhamov N, Massin P, Haouchine B, Erginay A, Gaudric A. Macular retinoschisis in highly myopic eyes. Am J Ophthalmol. 2002;133(6):794-800.
- Lee EJ, Kim TW, Kim M, Choi YJ. Peripapillary retinoschisis in glaucomatous Eyes. PLoS One. 2014;9(2):e90129.8.
- Zumbro DS, Jampol LM, Folk JC, Olivier MM, Anderson-Nelson S. Macular schisis and detachment associated with presumed acquired enlarged optic nerve head cups. Am J Ophthalmol. 2007;144:70-74.
- Lewis H. Peripheral retinal degenerations and the risk of retinal detachment. Am J Ophthalmol. 2003;136(1):155-60.
- Molday RS, Kellner U, Weber BH. X-linked juvenile retinoschisis: clinical diagnosis, genetic analysis, and molecular mechanisms. Prog Retin Eye Res. 2012;31(3):195-212.
- Song IS, Shin JW, Shin YW, Uhm, KB. Optic disc pit with peripapillary retinoschisis presenting as a localized retinal nerve fiber layer defect. KJO. 2011;25(6):455-458.
- Melton R, Thomas R. 2016 Clinical Guide to Ophthalmic Drugs. Review of Optometry. 2016;May 15(suppl):36-43.