Targeting Intraocular Pressure in Glaucoma: a Teaching Case Report
Andrew Kemp, OD, Marcus Gonzales, OD, FAAO, Joe DeLoach, OD, FAAO, and Zanna Kruoch, OD FAAO
Abstract
The setting of a target intraocular pressure is one of the most commonly performed procedures in glaucoma management. However, the setting of this marker is often made as an educated guess without a firm scientific basis. This manuscript discusses different strategies for targeting intraocular pressure backed by scientific research with a case example. Through the evaluation of multiple large-scale studies, a recommended standard of a 30% reduction for mild glaucoma, a 40% reduction for moderate glaucoma and a 50% or more reduction for severe glaucoma can be utilized to provide reliable and consistent glaucoma care.
Key Words: glaucoma, target pressure, staging, management
Background
Glaucoma is a range of conditions that causes a loss of retinal ganglion cell axons within the nerve fiber layer resulting in vision loss.1,2 Although many novel treatments for glaucoma are being investigated, the mainstay therapy is control and reduction of intraocular pressure (IOP).1,2 Upon diagnosis, the setting of an IOP target is the first step in the management of any glaucoma patient. This case report presents an example of one method for determining target IOP along with a brief review of the literature. This report is aimed for third- and fourth-year optometric students and practicing optometrists managing glaucoma.
Case Description
Referred by an outside physician, a 75-year-old African American male presented to the clinic for a glaucoma evaluation. The patient reported having no visual complaints, but he did report that his mother and maternal grandmother were diagnosed with severe glaucoma and that he had a history of high IOP. Best-corrected visual acuities were 20/20 in both eyes and all preliminary and anterior segment findings were normal except for mild nuclear sclerotic cataracts. Intraocular pressure measurements were 24 mmHg in the right eye and 27 mmHg in the left measured with Goldmann tonometry. Pachymetry revealed a corneal thickness of 525 µm in the right eye and 530 µm in the left eye. Anterior chamber angles were open to the ciliary body in all quadrants with minimal pigment and a flat iris insertion on gonioscopy in both eyes. Optic nerve head evaluation in the right eye revealed a round, distinct, well-perfused nerve with a vertical cup-to-disc ratio of 0.6 that followed the ISNT rule. According to the ISNT rule, the inferior rim of the optic nerve typically is the thickest, followed by the superior then nasal then temporal rim. The left optic nerve was also round, distinct and well-perfused, but the vertical cup-to-disc ratio was 0.8 and it did not follow the ISNT rule. The vertical disc diameter was 1.7 mm when viewed through a 78D lens in both eyes. The horizontal cup-to-disc ratio was equivalent to the vertical disc diameter. An inferior wedge defect was noted within the retinal nerve fiber layer on direct observation in the left eye and it was confirmed with optical coherence tomography (OCT). Primary open-angle glaucoma was diagnosed, and the patient was instructed to return for multiple follow-up visits for baseline visual field testing and repeat IOP measurements prior to treatment.
Upon multiple follow-ups, IOP remained in the 24-28 mmHg range in both eyes and a mild, superior/nasal step visual field defect was confirmed in the left eye. Prostaglandin therapy was initiated in both eyes and a target IOP of 18-20 mmHg was set to achieve a 30% reduction from the highest recorded measurement. Further glaucoma monitoring was initiated.
Education Guidelines
Learning objectives
At the conclusion of this case report a student should be able to:
- Accurately/consistently set a target IOP for glaucoma patients with scientific basis
- Understand the staging of glaucoma and how it relates to target IOP
- Describe variables in patient care that can affect the target IOP
Key concepts
- Properly staging glaucoma aides in the determination of the level of IOP reduction needed
- Setting a target IOP that ensures the highest probability for arresting the progression of glaucoma based on scientific evidence
Discussion points
- Basic knowledge of the role of controlling IOP in glaucoma
- Understand different technologies and how to incorporate them into glaucoma management
- Understand when to treat and when not to treat glaucoma
- Understand why a patient may not respond to treatment in the predicted way
- Clinical management of IOP
- Combine objective and subjective testing to create a consistent staging system
- Utilize the staging system to establish the initial target IOP range
- Understanding different strategies for setting target IOP
- Understand when an IOP target is inadequate
- Patient education
- How to use the target IOP to improve patient education, improve compliance, and provide the patient with a tangible goal
- Understand the importance of patient education on patient compliance
Discussion
Before students will be able to set the target IOP in glaucoma management, they must first understand the purpose and definition of target IOP. Target IOP is defined by the World Glaucoma Association (WGA) as “an estimate of mean IOP at which the risk of decreased vision-related quality of life due to glaucoma exceeds the risk of treatment.”2,3 Although this is an excellent description of the purpose of lowering pressure in this very diverse condition, it leaves room for interpretation of how to determine the extent to which the IOP should be lowered. This discussion will briefly review a few major studies that investigated the effect of IOP lowering on glaucomatous changes with ideas and concepts to be used in determining each patient’s unique target IOP as in the example above. The faculty will be able to use the following studies to provide a foundation for which students can develop their own glaucoma management principles. These studies may be best presented through case examples but can be introduced initially through lecture presentation. Students should become increasingly independent in their decision-making in clinic while managing glaucoma patients. They should also be able to have higher-order discussions with attending doctors when discussing comprehensive glaucoma management. Course objectives will be met when students can stage glaucoma correctly and consistently while setting a target pressure for therapy that ensures the lowest risk of progression.
The first concept for students to grasp when diagnosing glaucoma and determining treatment is highlighted within the WGA definition of the target IOP stated above. The “risk of decreased vision-related quality of life due to glaucoma” must outweigh any risks of treatment. This statement implies a situation in which a patient may have glaucoma, but the risk of vision loss that would affect one’s quality of life is not high enough to warrant treatment. This relates to the patient’s age in the above example. The patient is 75 years old with mild glaucomatous visual field loss. Due to the patient’s age, it is likely that there would be noticeable vision loss within his lifetime, and thus treatment was initiated. However, if the patient were 90 years old with mild vision loss and a concurrent terminal cancer diagnosis, initiating glaucoma treatment may decrease the quality of life of the patient even though vision loss would not be expected within the patient’s’ lifetime. There is often no absolute correct answer for these situations and the risk-benefit profile must be explained in detail to the patient and his or her family in order for a decision to be made on the proper course of action. A discussion must occur between faculty and students so that they understand that proper glaucoma management includes when to initiate glaucoma treatment and when to withhold treatment.
There are multiple theories for calculating target IOP. These theories are best presented to students in a lecture setting prior to clinical management. A commonly used theory, and the one used in this case report, is determining a percentage reduction based on a diurnal collection of untreated IOP measurements.2 The percentage reduction needed for each patient can vary from 20-50% or greater depending on factors including the type of glaucoma, severity, family history and age.1,2 In the Ocular Hypertensive Treatment Study (OHTS), the patients in the treatment group achieved a reduction of 20% and the study resulted in a 50% reduction in the number of patients who converted to glaucoma in five years.4 This reduction, however, is limited to those patients with pressures defined as ocular hypertensive (greater than 21 mmHg). Although the patient in the case report above had ocular hypertension, it was decided that a 20% reduction was not adequate due to his family history of severe glaucoma and thin corneas (less than 555 µm). Therefore, a 30% reduction was designated. Corneal thickness is a risk factor for patients with ocular hypertension.4 Patients with thin corneas are at a higher risk of developing glaucoma. Beyond this risk, corneal thickness has limited application. Corneal thickness can alter IOP readings in thicker corneas potentially providing falsely high readings and thin corneas providing falsely low readings on applanation tonometry. While corneal thickness nomograms proposed an addition or reduction of the IOP based on these generalizations, the extent of the actual alteration in the IOP reading for individual patients is unknown. This is a good example to use with students to show that they must combine multiple variables and risk factors for each individual case and there is not one correct way to manage every single glaucoma patient.
The Collaborative Initial Glaucoma Treatment Study (CIGTS) determined that a target percentage reduction of 35% in newly diagnosed glaucoma showed the same reduction in risk of progression as more aggressive therapy (40% reduction or greater).5 However, in patients who already had advanced disease, larger reductions achieved with surgery significantly reduced the risk of progression compared to medicinal therapies.2,5 Surgery holds multiple benefits over medicinal methods that will not be discussed here, but it can be interpreted that a more aggressive approach to IOP management is most valuable in advanced disease. The severity of the glaucoma when initially presented can have significant importance when determining the target IOP.2,5 In this case report, the patient had a mild stage of glaucoma, thus a more aggressive target was not indicated. Student knowledge can be assessed when evaluating glaucoma severity. This will provide the faculty member with the knowledge of how well the student understands glaucoma fundamentals. Medical coding can also be included with this discussion, as the importance for proper staging has increased with the introduction of new coding principles.
Percentage reduction can also be used in conditions that do not have higher than statistically normal IOP. The Collaborative Normal Tension Glaucoma Study (CNTGS) showed benefits of a 30% reduction in IOP.6 However, with lower untreated IOP, it is sometimes difficult to reach high percentage reductions without surgical intervention. Expectations for treatment outcomes in different types of glaucoma can be addressed in the context of normal tension glaucoma. This provides a way to connect the mechanism of the disease and how that may change the expected outcome of treatment.
Through evaluation of multiple studies, it can be recommended that a staged percentage reduction can be considered. For mild, newly diagnosed glaucoma, a 30% reduction will significantly reduce the risk for progression. For more moderate and severe levels, a 40% and 50% reduction is likely needed.2,4-7 This provides the faculty with a scientifically based thought process for targeting IOP instead of explaining it as an educated guess. When setting the target, aiming for a range around the target IOP will take into account the variance between measurements. For example, if the untreated IOP ranges between 28 and 30 mmHg, a 30% reduction for mild glaucoma would set the target to 21, or 9 mmHg less than 30 mmHg. The target IOP range for this patient would be 19-21 mmHg. A standard procedure for setting target IOP can be very useful for clinical efficiency and ensuring continuity of care, but it has limitations. Each clinician must be aware that each glaucoma case may differ slightly and will need to tailor therapy on a case-by-case basis.1,2 In a clinical setting, students must understand common limitations to assessing intraocular pressure. IOP results can vary due to a number of variables including patient compliance, time of day and inter-clinician variability. This will help students understand that the target IOP is only an estimation and they should not be too quick to change treatment.
The percent reduction theory relies on the clinician having a scientifically based staging system for accurately assessing the severity of glaucoma. The standard for severity staging in glaucoma is mild, moderate and severe levels.8-11 A standard staging system will allow a clinician to be consistent from patient to patient for determining desired IOP reduction while reducing the amount of subjective bias. Staging each patient is a combination of optic nerve head assessment, retinal nerve fiber layer (RNFL) assessment and visual field defect assessment. Beginning with the optic nerve head, mild staging includes the narrowing of the optic nerve head rim tissue, asymmetry between the rim tissue of the two optic nerves and the presence of a disc hemorrhage. Transitioning into more moderate levels of atrophy, the moderate stage includes increased narrowing of the optic nerve rim tissue, central pseudopallor and localized notching of the rim tissue. Severe staging may include complete rim tissue loss in at least three quadrants, blood vessel bayoneting and bean-potting of the optic nerve head tissue.9,10 Only one of these characteristics is required to qualify the patient at that stage of glaucoma. Assessing a student’s knowledge of staging glaucoma incorporates a large number of concepts. The student must be able to understand the disease, assess the risk factors, interpret exam findings and come to a conclusion. This is an excellent exercise to assess a student’s ability for higher-order thinking that is crucial for becoming a successful clinician.
Evaluating the RNFL is the next step in evaluating the stage of glaucoma. Mild glaucoma is consistent with light striations in the RNFL and a decrease in brightness of reflectance of the RNFL tissue.9 Moderate glaucoma shows an overall or sectional dimming of the reflectance with a loss of texture to the RNFL. Severe RNFL atrophy shows a dark reflex against the retina due to the minimal remaining RNFL.9,10
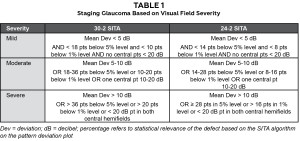
Lastly, evaluation of the visual field test results is necessary. Mild visual field defects begin with a nasal step or an isolated paracentral defect. On 24-2 SITA (Swedish Interactive Threshold Algorithm) testing, less than 14 points will be less than the 5% significance level and less than 8 will be below the 1%. The mean deviation will be less than 5 decibels (dB) and there will be no central points with a sensitivity less than 20 dB. The requirements are very similar on 30-2 SITA testing; however, more points are allowed below the 5% and 1% levels due to the increased total number of points tested. Moderate visual field defects are greater than the description in mild to one complete arcuate defect in one quadrant. There may be 14-28 points in the less than 5% significance level and 8-16 in the 1%. The mean deviation may be between 5 and 10 dB and a central point in one hemifield may be between 10-20 dB. Severe visual field defects include complete arcuate scotomas in both hemifields and 28 or greater points in the 5% significance level or greater than 16 in the 1%. The mean deviation may be >10 dB and there may be central points less than 20 dB in both hemifields. In the moderate and severe categories, the patient must meet only one of any of the characteristics listed above to be considered in that category of glaucoma. (Table 1)10 Table 1 is an excellent note for students to keep with them in clinic when they are first starting to manage glaucoma. Combining all of the above recommendations to categorize glaucoma patients into severity stages will aid in utilization of the percent reduction theory and ultimately better care for the patient.8,9,10
Another theory includes a fixed IOP threshold that is applied to all patients.2 The Advanced Glaucoma Intervention Study (AGIS) found that with an aggressive approach to IOP lowering (achieving a mean IOP of 12.3 mmHg or lower), no glaucomatous progression was noted.7 Supporters of the threshold theory often choose a fixed IOP of 12 or 13 mmHg to ensure that the risk for progression is the lowest possible for all patients.2,7 However, like with any study, AGIS has its limitations. The study focused on patients with advanced glaucoma only. Applying the same strategy to patients with mild or moderate stages could result in overtreatment, introducing increased risks for side effects and an unnecessary further decrease in quality of life. The threshold IOP theory has clinical uses, especially in patients who present with severe glaucomatous vision loss, but it does not apply to all glaucoma patients. Understanding how to set a target IOP can be a great teaching experience for students, showing them that they must gather data from research, clinical experience and from their education to establish how they will manage different conditions. They cannot do something simply because a previous attending or lecturer did it that way.
Once treatment is started and the initial target IOP is determined, it is also important to know when the target IOP is inadequate and needs to be adjusted. If glaucomatous progression is noted on objective and/or subjective testing, it is very important to determine the cause of the progression. One possible cause is an inadequate target IOP. In this situation, the target IOP will need to be lowered and an additional medication or surgery is indicated. Setting a new target depends on multiple factors including type of glaucoma, untreated IOP, the current treated IOP, the stage of glaucoma, and the expected rate of progression. The more severe and faster progressing glaucoma needs a more aggressive adjustment in target IOP. Following the percentage theory for target IOP, if a patient has shown mild progression while obtaining a 30% reduction in IOP, then it is possible only an additional 10% reduction may be necessary. However, if the patient has shown a severe change in objective or subjective testing, it may be necessary to add 20% more reduction to the previous IOP or to aim below a 12 mmHg threshold as determined by the AGIS study.7 It is very important to keep in mind limitations of topical therapy for controlling IOP including the difficulty of lowering IOP into single digits with medicinal therapy alone. This discussion allows for the faculty to introduce concepts to think about when adding additional therapy. It is still important to have goals set prior to changing or adding treatment and not to add treatment and gauge the goal in response to the results.
Another cause for progression of glaucoma is that the target IOP is adequate, but it is not consistently being met. This could result from the patient only using the prescribed medication consistently around the scheduled follow-ups, or sporadic use of the prescribed treatment regimen.1,12,13 In this situation, the IOP is within the target range at each visit, but progression is found due to the IOP not meeting the target during the time between visits. The patient may also be compliant but only reaching target IOP at appointment times and the treatment regimen is not properly controlling diurnal IOP. If the patient consistently has clinic appointments only in the morning, it is also possible that he or she is achieving the target IOP only in the morning and throughout the day the IOP is much higher.
Although it is possible that the need for additional medication is required in some cases, the literature often shows that the first cause a clinician should rule out is medication adherence.12,13 In a recent study, 48.1% of patients self-reported that their medication compliance was poor and/or declining by year four of treatment.12 Medication adherence is a huge concern with many chronic diseases and is particularly troublesome in glaucoma due to the lack of symptoms. In regard to target IOP, it is possible that a patient has not been meeting the target due to medication adherence. Medication adherence in glaucoma can be influenced by a number a factors. These include cost of medication, lack of understanding of the condition, ocular irritation/side effects of treatment, or the inability to apply the medication.12,13 When adherence is poor, glaucomatous progression would be confirmed, but, unless the stage significantly changed, no change in the treatment plan would be required. Reinforcement of the established treatment plan is required and more frequent monitoring of the patient may be required to improve compliance. It is important for students to understand that there are multiple reasons for disease progression and each must be carefully addressed to avoid over-prescribing of medications or polypharmacy. Patient education can be a focused talking point when addressing patient adherence.
Conclusion
Although target IOP is in widespread use throughout the glaucoma management community, there is a lack of evidence that validates what target IOP is appropriate for each situation and how to calculate it. Many large associations such as the WGA or the American Academy of Ophthalmology recommend its use while understanding the limitations.1,2 In the end, target IOP is a prediction, and reviewing literature allows the clinician to make the most educated prediction possible. Achieving or not achieving target IOP does not define control of the disease. It is only structural or more commonly functional change that dictates the target IOP. Based on the review of literature, a staggered percentage reduction system (30%, 40%, 50%) can be recommended for mild, moderate and severe glaucoma respectively. At the current time, modifying IOP is the only way to preserve quality of life for glaucoma patients. As the understanding of glaucoma changes and the ability to monitor IOP over a 24-hour period becomes a viable option, the strategy of target IOP could change drastically.
References
- Weinreb RN, Aung T, Medeiros FA. The pathophysiology and treatment of glaucoma. JAMA. 2014;311(18):1901-1911.
- Colin C, Bhartiya S, Shaarawy T. New perspectives on target intraocular pressure. Surv Ophthalmol. 2014;59(6):615-626.
- Consensus Statement. The Netherlands, Kugler. World Glaucoma Association Consensus statement: Intraocular Pressure. 2007. Available from: https://www.oculist.net/downaton502/prof/ebook/glaucoma/AIGSIntraocularPressure.pdf.
- Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120(6):701-13, 829-30.
- Lichter PR, Musch DC, Gillespie BW, et al. Interim clinical outcomes in the Collaborative Initial Glaucoma Treatment Study comparing initial treatment randomized to medications or surgery. Ophthalmology. 2001;108(11):1943-53.
- Collaborative Normal-Tension Glaucoma Study Group. Comparison of glaucomatous progression between untreated patients with normal-tension glaucoma and patients with therapeutically reduced intraocular pressures. Am J Ophthalmol. 1998;126(1):487-97.
- AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS): the relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol. 2000;130(4):429-40.
- Spaeth GL, Lopes JF, Junk AK, Grigorian AP, Henderer J. Systems for staging the amount of optic nerve damage in glaucoma: a critical review and new material. Surv Ophthalmol. 2006;51(4):293-315.
- Quigley HA, Reacher M, Katz J, Strahlman E, Gilbert D, Scott R. Quantitative grading of nerve fiber layer photographs. Ophthal. 1993;100(12):1800-7.
- Brusini P, Johnson CA. Staging functional damage in glaucoma: review of different classification methods. Surv Ophthalmol. 2007;52(2):156-79.
- Balwantray C, Burgoune C. From Clinical Examination of the Optic Disc to Clinical Assessment of the Optic Nerve Head: A Paradigm Change. Am J Ophthalmol. 2013;156(2): 218-227.
- Newman-Casey PA, Blachley T, Lee PP, Heisler M, Farris KB, Stein JD. Patterns of glaucoma medication adherence over four years of follow-up. Ophthalmology. 2015;122(10):2010-21.
- Alan R, Grover DS. Compliance and adherence in glaucoma management. Indian Journal of Ophthalmology. 2011;59(1):S93-S96.