PEER REVIEWED
Optic Disc Drusen and Associated Complications
Raman Bhakhri, OD, FAAO, and Courtney Luce, OD
Abstract
Optic disc drusen (ODD) are congenital multi-lobed calcific deposits that are possibly due to a disturbance in axonal metabolism and slowed axoplasmic flow. Buried early in life, they become more superficial and visible as the patient ages. Although most patients are asymptomatic, visual acuity and visual field loss can occur. Rare complications can include peripapillary choroidal neovascular membrane and ischemic optic neuropathy. This case report reviews the epidemiology, pathophysiology, differential diagnosis and treatment options for ODD and ODD-associated complications.
Key Words: optic disc drusen, choroidal neovascular membrane, field loss, B-scan, fundus autofluorescence
Introduction
Optic disc drusen (ODD) are located in the optic nerve head of up to 2.4% of the population and are thought to be due to impaired axonal metabolism.1,2 In many cases, ODD are not visible due to being very small or poorly calcified or having a deeper location.3 They often give the appearance of optic disc elevation (pseudopapilledema), which can be confused with true papilledema caused by intracranial hypertension. Therefore, making a proper diagnosis is essential.1,4 Although most patients are asymptomatic, ODD can be associated with visual field defects in up to 87% of cases as well as visual acuity loss and infrequently choroidal neovascular membrane (CNVM) and non-arteritic ischemic optic neuropathy (NAION).2 This case highlights the rare development of visual acuity and visual field loss in addition to a peripapillary CNVM in a young female patient with multi-modal imaging aiding in the diagnosis and management.
Case Presentation
A 26-year-old Caucasian female presented for evaluation of blurry vision OS > OD, which was equal at distance and near. She had no other associated symptoms. Her ocular history was unremarkable. Her medical and family health history was also unremarkable. On initial examination, entering uncorrected distance visual acuities were 20/25-2 OD and 20/30 OS. Pupils were round and reactive to light with a mild afferent pupillary defect (AFD) present OS. Extraocular motility and confrontation fields were full OU. Manifest refraction revealed compound hyperopic astigmatism OU with best-corrected distance visual acuities of 20/20 OD and 20/25 OS. Intraocular pressure (IOP) measured with Goldmann applanation tonometry was 16 mmHg OU. Slit lamp biomicroscopy was unremarkable OD and OS. Blood pressure was measured at 110/67 mmHg right arm, sitting. Dilated fundus examination revealed a scalloped and raised optic nerve giving the appearance of indistinct and irregular disc margins OD and OS. This was more evident superior-temporally in both eyes (Figures 1 and 2). A spontaneous venous pulse was not noted in either eye. The cup-to-disc ratio was estimated to be 0.1/0.1 OD and OS. The left eye was also remarkable for a fibrovascular lesion superior-temporal to the optic nerve with surrounding pigment (Figure 2). No hemorrhaging or fluid was noted around the lesion. The presence of pigment around the lesion along with fibrovascular tissue indicated likely chronicity of the unknown lesion.5-7 The macula OD and OS was unremarkable. Fundus autofluorescence (FAF), with clear imaging and no artifacts present, revealed focal areas of hyper-autofluorescence on both optic nerves, OS > OD (Figures 3 and 4). FAF also showed mixed hyper- and hypo-autofluorescence of the fibrovascular tissue (hypo-autofluorescence surrounded by hyper-autofluorescence) with surrounding hypo-autofluorescence of the corresponding retinal pigment in the area juxtapapillary and superior-temporal to the optic nerve OS (Fig 4). An incidental operculated retinal hole, located superior-temporally in the periphery of the left eye, was also found at this visit with no evidence of retinal detachment. The periphery of the right eye was unremarkable. A baseline 24-2 SITA FAST Humphrey visual field test was performed in both eyes. Testing OD was reliable based on low amounts of fixation loss, false positive errors and false negative errors. Testing OS was of questionable reliability due to the blind spot not being plotted. Testing OD revealed a shallow inferior arcuate defect (Figure 5). Testing OS revealed nasal defects > inferiorly indicative of an arcuate defect with extension into the inferior-temporal quadrant (Figure 6).
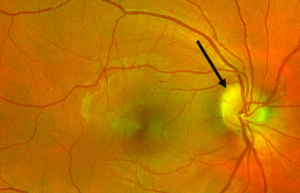 Figure 1. Ultra-widefield retinal imaging of the right eye showing a scalloped and raised presentation of the optic disc drusen giving the appearance of indistinct and irregular disc margins, more so superior-temporally (black arrow). |
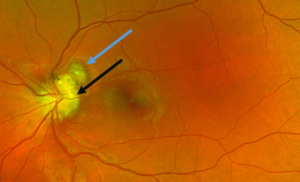 Figure 2. Ultra-widefield retinal imaging of the left eye showing a scalloped and raised presentation of the optic disc drusen giving the appearance of indistinct and irregular disc margins, more so superior-temporally (black arrow). A fibrovascular lesion with surrounding pigment, indicative of a choroidal neovascular membrane, is noted juxtapapillary to the optic disc (blue arrow). Click to enlarge |
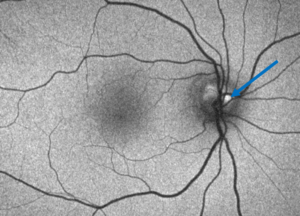 Figure 3. Fundus autofluorescence of the right eye showing hyper-autofluorescence of the optic disc, more so superior-temporally, indicating optic disc drusen (blue arrow). |
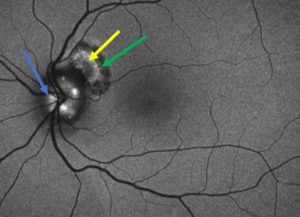 Figure 4. Fundus autofluorescence of the left eye showing hyper-autofluorescence of the optic disc indicating optic disc drusen (blue arrow). A mixed patch of hypo- and hyper-autofluorescence (hypo-autofluorescence surrounded by hyper-autofluorescence, yellow arrow) surrounded by hypo-autofluorescence (green arrow) is noted superior-temporal to the disc. This corresponds with the fibrovascular lesion with surrounding pigment disruption noted on fundus photos and is indicative of the inactive choroidal neovascular membrane. |
 Figure 5. Baseline 24-2 SITA FAST Humphrey visual field test of the right eye. Testing was deemed reliable based on low amounts of fixation loss, false positive errors and false negative errors. Testing revealed an inferior arcuate defect that corresponded to the disc appearance on fundus photos (Figure 1) and with optical coherence tomography scans that revealed retinal nerve fiber layer thinning superior-temporally > inferior-temporally (Figure 7). Click to enlarge |
 Figure 6. Baseline 24-2 SITA FAST Humphrey visual field test of the left eye. Testing was deemed questionable as the blind spot was not accurately plotted. Testing revealed an inferior arcuate defect that corresponded to the disc appearance on fundus photos (Figure 2), with optical coherence tomography scans that revealed retinal nerve fiber layer thinning superior-temporally > inferior-temporally and with large amounts of ganglion cell complex thinning noted superiorly on ganglion cell complex testing (Figure 8). |
Optical coherence tomography (OCT) of both optic nerves was also performed. The signal strength was adequate with no artifacts being noted indicating reliable test results. Small disc and rim areas were noted in both eyes along with very small to absent cup-to-disc ratios. The neuroretinal rim in both eyes was noted to be thicker than the normal range. Testing revealed significant retinal nerve fiber layer (RNFL) loss superior-temporally and inferior-temporally in both eyes (larger loss superior when compared to inferior), with larger amounts of thinning evident OS (Figure 7). These test results corresponded with the overall appearance seen on fundus photos. The OCT RNFL results also corresponded with the visual field defects (inferior arcuate defects OD/OS, deeper defect OS). The OCT scans, particularly the horizontal and vertical tomograms for each eye, revealed a lumpy bumpy internal contour of the optic nerves with no apparent or a very mild cup in addition to a normal retinal pigment epithelium (RPE)/Bruch’s membrane contour (Figure 7). Ganglion cell complex (GCC) analysis was obtained with OCT as well. The scan was judged reliable based on a strong signal strength and absence of artifact. Testing OD showed dense temporal thinning, while testing OS showed significant amounts of overall thinning, with larger amounts of thinning noted in the superior quadrants (Figure 8). Comparison of the GCC scans showed significant asymmetry with larger amounts of thinning noted OS. An OCT raster scan, reliable with no artifacts present, was performed through the juxtapapillary lesion OS with testing showing raised and hyper-reflective fibrosis (Figure 9). There was no evidence of any fluid within or around the lesion.
Based on exam and multi-modal imaging findings (hyper-autofluorescence of both discs, neuroretinal rim thickening, RNFL loss and appearance on OCT scans and fundus imaging) the patient was diagnosed with ODD OD < OS. The juxtapapillary lesion OS was thought to represent an inactive and chronic CNVM secondary to the ODD. This was based on its location adjacent to the ODD, lack of fluid in and around the lesion, lack of hemorrhaging, and presence of retinal pigment. The mild reduced acuity and positive APD OS were attributed to the significant RNFL and GCC thinning that were more prominent OS. Lastly, the patient was diagnosed with an operculated hole OS with no evidence of retinal detachment.
The patient was referred, non-urgently, to a retinal specialist for further evaluation of the presumed inactive CNVM as well as possible prophylactic treatment for the retinal hole. The patient was seen 2 weeks later by the retinal specialist. He opted not to perform fluorescein angiography (FA) and chose OCT angiography (OCT-A) instead. OCT-A scanning was performed and was deemed reliable with no apparent artifacts. Testing showed a net of blood vessels in the avascular zone without flow indicating an inactive CNVM. The retinal specialist also deemed it inactive based on a lack of adjacent fluid and hemorrhaging and due to the surrounding retinal pigment hyperplasia (Figure 10). Barricade laser was performed around the operculated retinal hole. The patient continues to be monitored every 6 months for possible progression of RNFL thinning and visual field defects. Her most recent visit revealed stable findings.
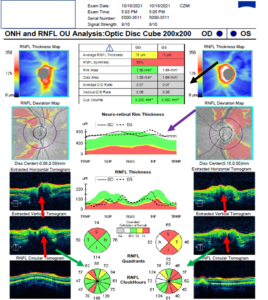 Figure 7. Optical coherence tomography (OCT) of both optic nerves. The signal strength was adequate with no artifacts being noted indicating reliable test results. Small disc and rim areas were noted in both eyes along with very small to absent cup-to-disc ratios (black arrow). The neuroretinal rim in both eyes was noted to be thicker than average (purple arrow). Testing revealed significant retinal nerve fiber layer (RNFL) loss superior-temporally > inferior-temporally in both eyes, with larger amounts of thinning evident OS (green arrows). These test results corresponded with the overall appearance of the discs seen on fundus photos. The OCT RNFL results also corresponded with the visual field defects (inferior arcuate defects OD/OS, deeper defect OS). The OCT scans, particularly the horizontal and vertical tomograms for each eye, revealed a lumpy bumpy internal contour of both optic nerves (red arrows) with no apparent or a very mild cup in addition to a normal retinal pigment epithelium/Bruch’s membrane contour, which was indicative of optic disc drusen. |
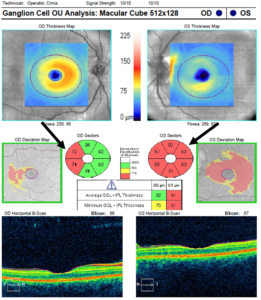 Figure 8. Ganglion cell complex analysis OD and OS. The scan was judged reliable based on a strong signal strength and absence of artifact. Testing OD showed dense temporal thinning, while testing OS showed significant amounts of overall thinning, with larger amounts of thinning noted in the superior quadrants (black arrows). The larger amounts of thinning OS correlated to the retinal nerve fiber layer thinning OS and to the reduced acuity and afferent pupillary defect noted OS. Click to enlarge |
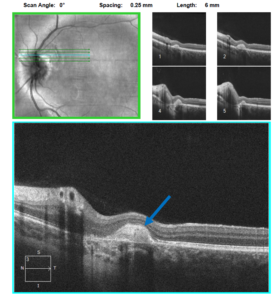 Figure 9. Spectral-domain optical coherence tomography raster scan of the inactive choroidal neovascular membrane OS. Fibrosis (blue arrow) is noted and no fluid is apparent. |
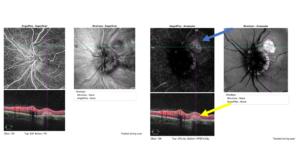 Figure 10. Optical coherence tomography angiography of the left optic nerve and retina, specifically the superficial enface (left) and the avascular enface (right). A hyper-reflective net representative of a choroidal neovascular membrane is present in the avascular layer (blue arrow). No blood flow is noted in the membrane indicating scarring and inactivity (yellow arrow). Click to enlarge |
Education Guide
Key concepts
- The basic anatomy, physiology and function of the optic nerve and its correlation to optic disc drusen
- The pathophysiology of optic disc drusen and associated complications
- Identifying and differentiating optic disc drusen from similar conditions
- Appropriate treatment and management plans including possible referrals and referral timelines
Learning objectives
- Recognize the clinical presentation of ODD including signs and symptoms
- List the potential differential diagnoses of ODD
- Create a management plan for a patient with and without complications from ODD
- Increase understanding of the value of ancillary testing in diagnosing and managing ODD
Discussion questions
1. Knowledge, understanding and facts about the clinical case and condition presentation
- What is the pathophysiology of ODD?
- Describe the typical appearance and presentation of ODD
- Discuss the complications that can occur secondary to ODD
2. Differential diagnosis
- What other condition(s) should be considered as differential diagnoses for ODD?
- How can ODD be differentiated from other similar conditions based on clinical appearance?
- What additional testing should be considered to help differentiate ODD from similar conditions?
3. Patient management and role of the optometrist
-
-
- What is the prognosis of ODD without complications?
- How would you manage ODD if there were no complications?
- How does your treatment and management plan change based on potential complications?
- What patient education should be given to a patient with this condition?
-
4. Critical-thinking concepts
-
-
- When should you consider referring this patient to another eyecare professional?
- How quickly should the referral be made?
- How does patient education change if the patient was diagnosed as a child rather than as an adult?
-
Assessment of learning objectives
This teaching case report is suitable for use in multiple settings that can include formal classroom lecture, small-group learning/laboratory or online or distance-learning.
In a classroom setting, students can be given the case details and tasked with arriving at a formal diagnosis and management plan based on the presented findings and ancillary testing. Understanding and knowledge can be evaluated through open-ended questions to the class or through formal testing with multiple-choice questions utilizing platforms such as TurningPoint.
In a small-group setting such as laboratory or clinic, students can have an open discussion together once the case is presented. Groups can then request appropriate images and scans including fundus photos, OCT scans and visual fields that will be interpreted and formally entered into a lab assignment sheet or electronic medical record. This will enhance comprehension of the material and can help lead to appropriate differential diagnoses, a formal diagnosis and a treatment and management plan.
In an online format, the case can be presented along with appropriate ancillary testing and results. Comprehension and understanding can be gauged through multiple-choice questions that address key concepts such as pathophysiology, signs and symptoms, differentials, interpreting testing and treatment and management. Students can be presented multiple-choice questions that must be answered correctly before they can proceed to the next section. Immediate feedback can be provided as students can learn from their mistakes and build upon foundational concepts.
Discussion
ODD are aggregations of calcium phosphate and hyaline deposits that form and manifest early in life with a location anterior to the lamina cribrosa.8 They are clinically found bilaterally in most cases but can be seen in an asymmetric fashion.3,9 Males and females are equally affected with the prevalence being fairly rare at 1-2%.1,10 Caucasians tend to manifest the condition more so than other ethnicities.11 The prevalence of ODD varies between ethnicities. A lower prevalence is noted in those with Chinese or African heritage while Caucasians tend to have an increased prevalence. This could be due to optic disc diameter, which is known to be smaller in Caucasians comapred with other ethnicities.11
Although they are usually a standalone finding, ODD can also be found with other ocular and systemic conditions including retinitis pigmentosa (RP), pseudoxanthoma elasticum (PXE) and rarer conditions such as Alagille syndrome.2 Optic disc drusen in patients with RP are seen at an increased rate compared to standalone ODD (9%) with the ODD tending to be parapapillary rather than within the disc itself.2,10 The ODD are also seen more frequently with systemically associated forms of RP such as Usher syndrome type 1 (35%) according to one study.12 Increased frequency of ODD is also seen with PXE as studies have estimated its prevalence to be anywhere from 8.5% to 24%, much higher than in the general ODD population.13,14 The increased prevalence is thought to be due to a hypo-calcification of Bruch’s membrane, which may lead to increased calcification of ODD.10 Alagille syndrome is a rare genetic disorder that leads to damage of the heart and liver. These patients also present with higher frequencies of ODD with one study showing more than 90% of patients with Alagille syndrome having ODD.15 Another more recent study found the presence of ODD to be 52%.16
The exact cause of ODD is not known currently. Researchers postulate that formation is secondary to axonal disruption and swelling of mitochondria into the prelaminar extracellular space with dysregulation of calcium deposition.17 This theory has been supported by a recent histopathological study that reported the absence of macrophages in some specimens of ODD, suggesting chronic axonal transport obstruction.10 Research also suggests that ODD are secondary to an irregular dominant inheritance pattern.18 However, this pattern is shown to be incomplete and sporadic. A gene for ODD has yet to be identified.19 As reported by Mullie et al., it has also been hypothesized that ODD form more often in small, crowded optic nerve heads with smaller scleral canals.20 Optic disc area has been shown to be significantly smaller in eyes with ODD.21
Signs or indications that a patient may have ODD include a small and elevated disc with irregular (scalloped) or blurred disc margins. Clinicians should look for round yellow white refractile clusters in and around the disc.3,9,10 With age, drusen tend to go from buried to a more superficial state.1 Other signs include abnormal vessel branching including tortuosity and trifurcations. The nasal aspect of the disc tends to be most affected.1,2,5,11,22-24
Most patients with disc drusen tend to be asymptomatic; however, advanced drusen deposition has been known to lead to transient vision obscurations, vision loss and visual field defects.2 Vision loss is usually mild, while field defects are very common and can progress as the drusen start to surface.2,3 Limited data exist on the rate of field loss at this time; however, studies have shown stability of field loss in adulthood.24,25 Our patient fits the general epidemiological profile based on her ethnicity, disc appearance and presentation with mild vison loss accompanied by field loss.
Clinical diagnosis can be straightforward if the ODD are visible; however, early on, the drusen may be buried preventing a proper funduscopic view.25 Buried ODD can also appear clinically as an elevated or swollen nerve leading to an improper diagnosis of papilledema, the leading differential for ODD.2 Although papilledema does look similar to ODD, clinicians can differentiate the conditions with a proper history, sound clinical examination and adjunct testing such as OCT, B-scan and FAF. As there is not true elevation in intracranial pressure with ODD patients, they should lack symptoms such as headaches and tinnitus.1,2,5,26 Close exam of the optic nerve may reveal elevation of the nerve. However, lack of retinal vessel obscuration and RNFL thickening in ODD patients can be used to differentiate from patients with true papilledema.9 Furthermore, differentiating the conditions has improved with the addition of multi-modal imaging. Specifically, with enhanced depth imaging OCT (EDI-OCT), clinicians can visualize ODD, even if they are buried, due to increased signal penetration.1,27 One study noted EDI-OCT to have a higher detection rate compared to the previous gold standard, a traditional B-scan.28 EDI-OCT findings include the presence of ODD above the lamina cribrosa, a signal poor core, anterior hyper-reflective margins and hyper-reflective horizontal lines representing early ODD.2 Careful examination of the EDI-OCT scans should be undertaken as peripapillary hyper-reflective ovoid mass-like structures (PHOMS) can also be seen. These were originally thought to be ODD, but research has shown them to represent a non-specific OCT indicator of axoplasmic stasis that is also seen in papilledema, central retinal vein occlusions and acute demyelinating optic neuritis.8 Clinicians should also examine the RPE/Bruch’s complex as inward deflection or angulation is more indicative of papilledema. This finding is not seen with ODD and is due to elevated pressure in the subarachnoid space.29 B-scan ultrasonography, the traditional standard in diagnosing ODD, is still a viable option. The ODD will present as hyper-echoic and intensely reflective structures with posterior acoustic shadowing.2,4,5,30 If the ODD are buried, however, especially at a very young age, B-scan may not detect them. This is likely due to the buried drusen being in an uncalcified state.31,32 The autofluorescence properties of ODD can also be utilized by FAF imaging. This is due to large amounts of porphyrins, which are fluorophores, present in disc drusen, making FAF a viable option for proper diagnosis.33,34 The ODD appear as hyper-autofluorescent deposits.35 A weakness of this imaging modality is its inability to detect deeper or buried ODD.35 FA can also be used; however, it represents an invasive way to image ODD.2 Testing would show optic disc staining in the later stages of the FA.35,36 Lastly, ODD can be detected with computed tomography, but due to low sensitivity and lack of quick access, it is not recommended for diagnosis.1
Papilledema is the primary differential for ODD, but other differentials can include congenital anomalies such as crowded or small discs and a malinserted disc, which can both give the appearance of pseudopapilledema.30 Crowded discs resemble ODD due to their congested appearance and are secondary to a small posterior scleral foramen with a normal amount of retinal axons passing through it. However, they will lack hyper-autofluorescence on FAF and will not have hyper-echoic structures on OCT when compared to ODD.30 They are almost always seen in patients with hyperopia.27 Another differential is the malinserted disc. These are discs that show large amounts of nasal heaping or elevation secondary to a tilt on the vertical axis. At times this is seen as blurred or indistinct disc margins. However, these patients will also lack hyper-autofluorescence on FAF with OCT showing nasal heaping of the disc but no hyper-echoic structures.27
As mentioned earlier, ODD are largely benign with most patients having a good visual prognosis. However, as they progress from a buried to superficial state with increased age, complications can arise. Superficial ODD can lead to axonal damage resulting in peripapillary RNFL thinning and secondary visual field loss.23,37 Buried drusen are thought to be less likely to affect the optic nerve head due to their deeper location, lesser calcification and smaller size compared with superficial ODD. Visual field defects can include an enlarged blind spot in patients with buried drusen, while patients with superficial drusen can manifest nasal steps and arcuate defects corresponding to respective RNFL thinning.26
Macular thinning can also be seen, specifically in the ganglion cell/inner-plexiform layer (GCIPL). Casado et al. showed that as drusen severity increased, RNFL and GCIPL thinning were more likely.38 This was evident in our patient, with the left eye being affected more superiorly than inferiorly. The superior GCIPL thinning correlated to the significant superior-temporal RNFL thinning in the left eye as well. Both of these findings then corresponded to the inferior visual field defect in the left eye. The visual acuity loss and a corresponding APD were a result of the significant GCIPL thinning, which was more evident in the left eye than in the right eye. Unfortunately, at this time there is no treatment for the progression of ODD and possible RNFL, GCIPL and visual field loss.
There is debate about whether treatment exists for standalone ODD. Studies have attempted to validate the use of IOP-lowering drops with or without an elevated IOP. Studies of patients with ODD with and without ocular hypertension have shown a greater prevalence of field loss in those with elevated IOP.39-41 However, careful analysis shows these to be retrospective or case report studies. A more recent study of 34 patients with bilateral ODD concluded that a decrease in IOP, with topical brimonidine, resulted in delayed optic neuropathy progression. However, the authors themselves note limitations such as small sample size and short time of observation that could have affected the results.42 Controlled clinical research is still needed for validated results on IOP and its relationship with ODD. At this time, primary or standalone ODD can be followed without treatment as most patients have a favorable visual prognosis.
Complications secondary to ODD can also occur. They include peripapillary hemorrhaging secondary to the compressive effects of ODD on adjacent peripapillary vessels. These tend to be benign and resolve without treatment.43 More serious complications can include CNVM, such as in our patient, or NAION. Peripapillary CNVM can be seen in younger or adult patients but younger patients tend to be more affected.9 CNVM tends to occur more nasally on the disc, likely due to the higher incidence of ODD on the nasal aspect of the disc. The etiology of peripapillary choroidal neovascularization is not known, but researchers postulate that enlarging drusen may compress and damage the vascular supply to the peripapillary retina resulting in an area of ischemia. This would lead to the release and upregulation of vascular endothelial growth factor (VEGF) and the development of a CNVM.5,44 This theory would correlate to younger patients presenting with CNVM.5,9 Although it is not used as frequently in the diagnosis of CNVM, when compared to FA, OCT and OCT-A, FAF may be a reasonable initial option as well. Subretinal fluid from active CNVM usually presents as hyper-autofluorescence, while disciform scarring or fibrosis presents as uneven autofluorescence of the lesion surrounded by hyper-autofluorescence (as in this case).45 Hemorrhaging and exudation from active CNVM, if present, usually present as hypo-autofluorescence along with RPE atrophy and RPE hyperplasia (as in this case).45 Anti-VEGF medications are standard treatment, and an urgent referral for treatment is indicated, especially when the CNVM threatens the macula. Most ODD patients require fewer injections for CNVM than do patients with age-related macular degeneration.5 If the CNVM is not near or threatening the macula, they can potentially be observed for self-resolution.2,5,9 The patient in this case report is therefore fortunate as the CNVM was located superior-temporal to the optic nerve and did not encroach near the fovea, which spared her central visual acuity.
Although extremely rare, another visually devasting complication that can occur secondary to ODD is NAION. Both NAION and ODD occur in eyes with an absent or small cup-to-disc ratio with a crowded appearance, namely a disc at risk. NAION in particular is known to be caused by a decrease in perfusion to the optic nerve leading to the resultant ischemic event.46 In terms of NAION secondary to ODD, it is thought that patients with ODD can have a resultant NAION as the ODD crowd the disc even more, leading to a further decline in perfusion pressure.46 Interestingly, cases of ODD-associated NAION tend to occur more so in younger patients with outcomes (vision and field loss) being more favorable when compared to traditional cases of NAION.22 Other signs and symptoms that can be seen with ODD-associated NAION include a sudden onset of vision loss along with disc pallor. None of these was noted by our patient or observed in her examination. Unfortunately, there are no treatments available for any type of NAION at this time. However, urgent referral to other eyecare professionals should be made if NAION is suspected.
Clinicians should follow ODD patients on a semi-annual to annual basis, with multi-modal imaging, to monitor for any progressive visual field and/or RNFL loss. Younger patients tend to progress more in terms of field loss and presenting with complications than older patients and should therefore be educated appropriately on this.5,22,28 Unfortunately, there are no treatments for ODD and treatment is reserved for possible complications arising from ODD.
This case highlights standard and rare complications that can been seen with ODD. Although most cases are benign with no visual sequalae, clinicians should be aware of the potential complications that can arise including vision and field loss. This can be due to normal ODD progression or be a consequence of conditions associated with ODD such as peripapillary CNVM or NAION. Patients, especially younger patients, should be educated on the natural progression of ODD and on identifying potential signs and symptoms associated with rarer complications. Clinicians should consider more frequent examinations in younger patients while also considering more frequent examinations in cases with ODD complications. Multi-modal imaging, such as OCT and FAF in addition to standard visual field testing, is strongly suggested in the management of patients with ODD and ODD-associated complications.
References
- Hamann S, Malmqvist L, Costello F. Optic disc drusen: understanding an old problem from a new perspective. Acta Ophthalmol. Nov 2018;96(7):673-684.
- Palmer E, Gale J, Crowston JG, Wells AP. Optic nerve head drusen: an update. Neuroophthalmology. Dec 2018;42(6):367-384.
- Flores-Rodríguez P, Gili P, Martín-Ríos MD. Ophthalmic features of optic disc drusen. Ophthalmologica. 2012;228(1):59-66.
- Rebolleda G, Kawasaki A, de Juan V, Oblanca N, Muñoz-Negrete FJ. Optical coherence tomography to differentiate papilledema from pseudopapilledema. Curr Neurol Neurosci Rep. 2017 Aug 17;17(10):74.
- Akıdan M, Bulut M, Yaprak L, Erol MK, Suren E. Optic coherence tomography angiography findings of bilateral choroidal neovascularization associated with optic disc drusen treated with intravitreal aflibercept injection. Case Rep Ophthalmol Med. 2020 Mar 10;2020:8398054.
- Ishikawa K, Kannan R, Hinton DR. Molecular mechanisms of subretinal fibrosis in age-related macular degeneration. Exp Eye Res. 2016 Jan;142:19-25.
- Friedlander M. Fibrosis and diseases of the eye. J Clin Invest. 2007 Mar;117(3):576-86.
- Fraser JA, Sibony PA, Petzold A, Thaung C, Hamann S. Peripapillary hyper-reflective ovoid mass-like structure (PHOMS): an optical coherence tomography marker of axoplasmic stasis in the optic nerve head. J Neuroophthalmol. Dec 1 2021;41(4):431-441.
- Chang MY, Pineles SL. Optic disk drusen in children. Surv Ophthalmol. 2016 Nov-Dec;61(6):745-758.
- Auw-Haedrich C, Staubach F, Witschel H. Optic disk drusen. Surv Ophthalmol. 2002 Nov-Dec 2002;47(6):515-32.
- Hoehn R, Butsch C, Zwiener I, Blankenberg S, Pfeiffer N, Mirshahi A. optic disc drusen: prevalence, ocular and systemic associations in the Gutenberg Health Study. Invest Ophthalmol Vis Sci. 2012;53(14):4885-4885.
- Edwards A, Grover S, Fishman GA. Frequency of photographically apparent optic disc and parapapillary nerve fiber layer drusen in Usher syndrome. Retina. 1996;16(5):388-92.
- Pipelart V, Leroux B, Leruez S, et al. A study of optic nerve head drusen in 38 pseudoxanthoma elasticum (PXE) patients (64 eyes). Location of optic nerve head drusen in PXE. J Fr Ophtalmol. Mar 2019;42(3):262-268.
- Munteanu M, Chercotä V. Drusenul papilar şi striile angioide în pseudoxantomul elastic [Optic nerve drusen and angioid streaks in pseudoxanthoma elasticum]. Oftalmologia. 2007;51(1):99-102.
- El-Koofy NM, El-Mahdy R, Fahmy ME, El-Hennawy A, Farag MY, El-Karaksy HM. Alagille syndrome: clinical and ocular pathognomonic features. Eur J Ophthalmol. Mar-Apr 2011;21(2):199-206.
- da Palma MM, Igelman AD, Ku C, et al. Characterization of the spectrum of ophthalmic changes in patients with Alagille syndrome. Invest Ophthalmol Vis Sci. 2021;62(7):27-27.
- Tso MO. Pathology and pathogenesis of drusen of the optic nervehead. Ophthalmology. Oct 1981;88(10):1066-80.
- Serpen JY, Prasov L, Zein WM, et al. Clinical features of optic disc drusen in an ophthalmic genetics cohort. J Ophthalmol. 2020 Oct 6;2020:5082706.
- Backhaus B, Lorentzen SE. Prevalence of pseudoexfoliation in non-glaucomatous eyes in Denmark. Acta Ophthalmol (Copenh). 1966;44(1):1-4.
- Mullie MA, Sanders MD. Scleral canal size and optic nerve head drusen. Am J Ophthalmol. Mar 15 1985;99(3):356-9.
- Jonas JB, Gusek GC, Guggenmoos-Holzmann I, Naumann GO. Optic nerve head drusen associated with abnormally small optic discs. Int Ophthalmol. Dec 1987;11(2):79-82.
- Fraser JA, Rueløkke LL, Malmqvist L, Hamann S. Prevalence of optic disc drusen in young patients with nonarteritic anterior ischemic optic neuropathy: a 10-year retrospective study. J Neuroophthalmol. 2021 Jun 1;41(2):200-205.
- Malmqvist L, Kyhnel A, Hamann S. Substantial Visual Field Loss Associated With Giant Optic Disc Drusen. JAMA Ophthalmol. Dec 2017;135(12):e174778.
- Malmqvist L, Kyhnel A, Hamann S. Substantial visual field loss associated with giant optic disc drusen. JAMA Ophthalmol. 2017 Dec 14;135(12):e174778.
- Spencer TS, Katz BJ, Weber SW, Digre KB. Progression from anomalous optic discs to visible optic disc drusen. J Neuroophthalmol. Dec 2004;24(4):297-8.
- Wilkins JM, Pomeranz HD. Visual manifestations of visible and buried optic disc drusen. J Neuroophthalmol. Jun 2004;24(2):125-9.
- Chiang J, Wong E, Whatham A, Hennessy M, Kalloniatis M, Zangerl B. The usefulness of multimodal imaging for differentiating pseudopapilloedema and true swelling of the optic nerve head: a review and case series. Clin Exp Optom. 2015 Jan;98(1):12-24.
- Merchant KY, Su D, Park SC, et al. Enhanced depth imaging optical coherence tomography of optic nerve head drusen. Ophthalmology. Jul 2013;120(7):1409-14.
- Kupersmith MJ, Sibony P, Mandel G, Durbin M, Kardon RH. Optical coherence tomography of the swollen optic nerve head: deformation of the peripapillary retinal pigment epithelium layer in papilledema. Invest Ophthalmol Vis Sci. Aug 22 2011;52(9):6558-64.
- Freund P, Margolin E. Pseudopapilledema. [Updated 2022 Aug 8]. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK538291/.
- Antcliff RJ, Spalton DJ. Are optic disc drusen inherited? Ophthalmology. Jul 1999;106(7):1278-81.
- Petrushkin H, Ali N, Restori M, Adams GG. Development of optic disc drusen in familial pseudopapilloedema: a paediatric case series. Eye (Lond). Aug 2011;25(8):1101-2.
- Schmitz-Valckenberg S, Holz FG, Bird AC, Spaide RF. Fundus autofluorescence imaging: review and perspectives. Retina. Mar 2008;28(3):385-409.
- Krishnamurthy PC, Du G, Fukuda Y, et al. Identification of a mammalian mitochondrial porphyrin transporter. Nature. Oct 5 2006;443(7111):586-9.
- Tuğcu B, Özdemir H. Imaging methods in the diagnosis of optic disc drusen. Turk J Ophthalmol. 2016 Oct;46(5):232-236.
- Chen JJ, Flanagan EP, Bhatti MT, et al. Steroid-sparing maintenance immunotherapy for MOG-IgG associated disorder. Neurology. Jul 14 2020;95(2):e111-e120.
- Malmqvist L, Wegener M, Sander BA, Hamann S. Peripapillary retinal nerve fiber layer thickness corresponds to drusen location and extent of visual field defects in superficial and buried optic disc drusen. J Neuroophthalmol. Mar 2016;36(1):41-5.
- Casado A, Rebolleda G, Guerrero L, et al. Measurement of retinal nerve fiber layer and macular ganglion cell-inner plexiform layer with spectral-domain optical coherence tomography in patients with optic nerve head drusen. Graefes Arch Clin Exp Ophthalmol. Oct 2014;252(10):1653-60.
- Grippo TM, Shihadeh WA, Schargus M, et al. Optic nerve head drusen and visual field loss in normotensive and hypertensive eyes. J Glaucoma. Mar 2008;17(2):100-4.
- Spalding JM. Visual-field loss with optic nerve drusen and ocular hypertension: a case report. Optometry (St Louis, Mo). Jan 2002;73(1):24-32.
- Morris RW, Ellerbrock JM, Hamp AM, Joy JT, Roels P, Davis CN, Jr. Advanced visual field loss secondary to optic nerve head drusen: case report and literature review. Optometry (St Louis, Mo). Feb 2009;80(2):83-100.
- Pojda-Wilczek D, Wycisło-Gawron P. The effect of a decrease in intraocular pressure on optic nerve function in patients with optic nerve drusen. Ophthalmic Res. 2019;61(3):153-158.
- Lee KM, Hwang JM, Woo SJ. Hemorrhagic complications of optic nerve head drusen on spectral domain optical coherence tomography. Retina. Jun 2014;34(6):1142-8.
- Law DZ, Yang FP, Teoh SC. Case report of optic disc drusen with simultaneous peripapillary subretinal hemorrhage and central retinal vein occlusion. Case Rep Ophthalmol Med. 2014;2014:156178.
- Ly A, Nivison-Smith L, Assaad N, Kalloniatis M. Fundus autofluorescence in age-related macular degeneration. Optom Vis Sci. 2017 Feb;94(2):246-259.
- Hayreh SS. Ischemic optic neuropathy. Prog Retin Eye Res. Jan 2009;28(1):34-62.



