PEER REVIEWED
Polypoidal Choroidal Vasculopathy
Jill Gottehrer, OD, FAAO
Abstract
Polypoidal choroidal vasculopathy (PCV) is one of the “pachychoroid spectra of diseases” of the retina. It often presents in the macula as orange nodules or subretinal hemorrhaging. Rarely, it will present in peripapillary or extramacular areas. PCV is commonly misdiagnosed as other retinal disorders that have different pathophysiology and require treatment plans, such as central serous chorioretinopathy and age-related macular degeneration. This teaching case report discusses a patient with previously misdiagnosed PCV. An accurate diagnosis was facilitated using ancillary testing, which is presented alongside a review of current evidence-based treatment guidelines.
Keywords
Background
Polypoidal choroidal vasculopathy (PCV) is believed to be a subtype of exudative age-related macular degeneration associated with an abnormal branching network of vessels with aneurysmal dilations referred to as polyps.1 Typically, it presents with orange nodules in the macular area or with a large (greater than 4-disc diameters) subretinal hemorrhage.2 Although less common, it may also occur in the peripapillary or extramacular areas. While PCV is a variation of choroidal neovascularization, it has a different pathophysiology compared to choroidal neovascularization related to exudative age-related macular degeneration.3
PCV has an estimated prevalence of 10-20% in a Caucasian population and as high as 22-62% in an Asian population.4 PCV occurs more often in males and bilaterally in a Caucasian population. In contrast, PCV occurs more often in females and unilaterally in an Asian population.5 It is most commonly diagnosed between the ages of 50-65 years old.6 Risk factors for PCV that have been identified include smoking, cardiovascular disease, hyperlipidemia and high body mass index.7-10
PCV is often misdiagnosed as central serous chorioretinopathy or exudative age-related macular degeneration. Multimodal imaging can help distinguish between these conditions. This teaching case report discusses a patient with PCV who was previously misdiagnosed with central serous chorioretinopathy. This case report presents the pathophysiology and evidence-based management guidelines for PCV. The target audience is third- and fourth-year optometry students, optometry residents and practicing optometrists.
Case Discussion
A 58-year-old Caucasian male presented to the eye clinic with a chief complaint of blurry vision in the left eye for the last 2 months. He further described it as constant darkness and waviness to his vision. He was last evaluated at the eye clinic 6 months prior. His systemic history was positive for hypertension, gastroesophageal reflux and sleep apnea, for which he was taking amlodipine besylate 10mg, losartan 50mg, aspirin 81mg and melatonin 3mg. A CPAP machine was used to manage his sleep apnea. No known medical allergies were reported. His ocular history was positive for dry eye syndrome, cataracts and simple hyperopia with presbyopia in both eyes. He was previously diagnosed with central serous chorioretinopathy in the left eye 7 years prior. The diagnosis was confirmed by a retina specialist. However, the patient elected to monitor without intervention. His ocular surgical history was negative and his family ocular history was negative.
At his current visit, his visual acuity in the right eye was stable at 20/25– and the left eye was reduced to 20/100 with no improvement upon refraction or pinhole compared to 20/30 6 months prior. Pupils were equal, round and reactive to light, motilities were full, and confrontation facial fields were full in each eye. His anterior segment in each eye was unremarkable. His intraocular pressure (IOP) by Goldmann Applanation Tonometry was 18mmHg in each eye at 2:05 PM. On posterior segment assessment, both eyes had 1+ nuclear sclerotic cataracts and vitreous syneresis, and both optic nerves had 0.25 round cupping. The macula in the right eye was within normal limits, while the left eye had a 1.5-disc diameter central area of hemorrhaging noted. The artery-to-vein ratio for both eyes were 2/3, and the periphery was within normal limits.
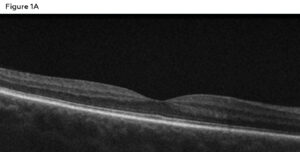
Figure 1A: Right eye macula OCT showing a normal foveal contour, inner/outer segment junction and RPE layer intact. Click to enlarge
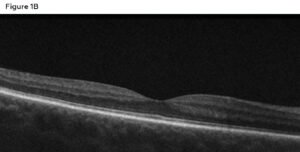
Figure 1B: . Left eye macula OCT showing a central macular hemorrhage (green arrow), double-layer sign (red arrow), and a thickened choroid (yellow arrow). Click to enlarge
Spectral-domain optical coherence tomography (SD-OCT) of the macula was performed without enhanced depth imaging. The right eye (Figure 1A) showed a normal contour with the inner segment/outer segment junction and retinal pigment epithelium layer intact. Comparatively, the left eye (Figure 1B) showed a subretinal hemorrhage, subretinal fluid, double-layer sign and a thickened choroid. Optos imaging was also obtained, showing no retinal pathologies in the right eye (Figure 2A) compared to a central macular hemorrhage in the left (Figure 2B).
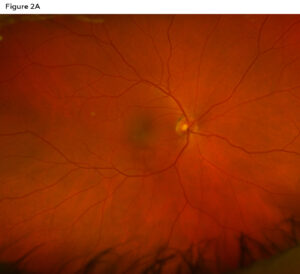
Figure 2A: . Optos imaging in the right eye showing no retinal pathology. Click to enlarge
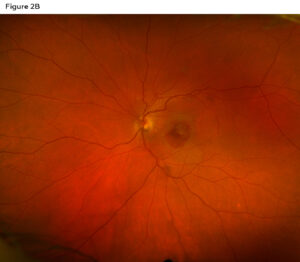
Figure 2B: . Optos imaging in the left eye showing a central macular hemorrhage. Click to enlarge
His diagnosis was changed from central serous chorioretinopathy to polypoidal choroidal vasculopathy in the left eye based on his clinical exam findings, including dilated fundus exam and OCT. He was referred to a retina specialist within a couple of weeks for further evaluation and management. The retina specialist confirmed the diagnosis and began treating the patient with anti-VEGF therapy.
Teaching Instructions
This teaching case report is most appropriate for third- and fourth-year students as well as residents. Appropriate assessments include presenting the case at a journal club at an optometry school, optometric rotation or residency site. Students and/or residents may work independently or in groups to answer the discussion questions.
Learning objectives
- Understand the signs and symptoms of PCV
- Understand the pathophysiology of PCV
- Understand the differential diagnoses of PCV
- Describe appropriate diagnostic testing for PCV
- Understand the treatment and management of PCV
- Understand the prognosis for PCV
Key concepts
- Identify the demographics and epidemiology of PCV
- Identify the clinical presentation of PCV
- Understand the different diagnoses of PCV
- Appropriate diagnostic testing for PCV
- Appropriate referrals, treatment and management for PCV
Discussion questions
Knowledge and understanding of the case and condition presentation
- What are the expected patient signs and symptoms in a case of PCV?
- What are the risk factors associated with PCV?
Differentials diagnoses
- What other conditions present in a similar clinical manner to PCV?
- How does a clinician differentiate between these similar conditions based on patient presentation and ancillary testing results?
Diagnostic testing
- Describe critical clinical testing for aiding the diagnosis of PCV and expected findings.
- What are the typical features of PCV noted on OCT, FA and ICGA?
Treatment and management
- What specialty referral would you consider making if you suspect PCV?
- What is the prognosis for PCV?
Discussion
Signs and Symptoms of PCV
Patients often report blurry vision and a central scotoma/metamorphopsia. On dilated fundus exam, a small, medium or large orange-red elevated central macular lesion may be noted. Polypoidal lesions are commonly noted in the macula in 69.5% of cases and peripapillary involvement in 4.5% of cases.11 PCV lesions can be isolated and single or multiple and widespread.12
Pathophysiology of PCV
PCV has been categorized as part of the pachychoroid spectrum of retinal conditions. This group of conditions also includes central serous chorioretinopathy, pachychoroid pigment epitheliopathy and pachychoroid neovasculopathy.13 “Pachychoroid” refers to thickening of the choroid with pathognomonic dilation of blood vessels in Haller’s layer. These vessels are referred to as “pachyvessels” and are associated with an abnormal increase in choroidal permeability.14 Pachyvessels are also accompanied by thinning of the choriocapillaris and middle choroid layer vessels that overlie them. Vascular endothelial growth factor (VEGF) stimulates angiogenesis and increases vascular permeability. Elevated levels of VEGF have been found in the aqueous of eyes with PCV, though in lower levels compared to neovascular age-related macular degeneration.15
Spontaneous subretinal hemorrhages may occur due to the rupture of the thin-walled choroidal vessels. Therefore, PCV lesions can be classified clinically as quiescent, hemorrhagic or exudative. Quiescent refers to the presence of polyps but lack of subretinal or intraretinal fluid or hemorrhage. Hemorrhagic refers to any subretinal or sub-retinal pigment epithelium (RPE) hemorrhaging with or without other exudative changes. Lastly, exudative PCV is characterized by the absence of hemorrhaging but the presence of exudative changes such as neurosensory detachments, sensory retinal thickening, pigment epithelium detachment (PED) and subretinal lipid exudation.6
There is also an association between increased levels of C-reactive protein, a nonspecific inflammatory protein and PCV.16,17 Other pro-inflammatory and pro-angiogenesis factors have been implicated in the pathogenesis of PCV, including VEGF, IL-23 (interleukin-23), MCP-1 (monocyte chemoattractant protein-1), and tumor necrosis factor-a.18 MCP-1 is one of the key chemokines that regulate migration and infiltration of monocytes and macrophages at the site of inflammation. MCP-1 induces angiogenesis via upregulation of hypoxia-inducible factor 1 alpha gene expression, and VEGF, in turn, stimulates MCP-1, suggestive of a positive regulatory feedback loop.19 Studies have found higher levels of MCP-1 in PCV eyes, indicating that MCP-1 is a significant biomarker and a potential therapeutic target.20 Tumor necrosis factor-a is another pro-inflammatory cytokine that can increase vascular leakage, ultimately contributing to choroidal neovascularization.21 IL-23 has a particularly strong association with PCV. IL-23 is a pro-angiogenic cytokine that increases the inflammatory response, such as the upregulation of matrix metalloproteinase MMP9.22 Matrix metalloproteases (MMP) degrade extracellular matrix (ECM) proteins. In those with PCV, MMP2 and MMP9 have been found to be elevated, implying that MMPs may serve as a biomarker of ECM metabolism for patients with PCV.23 Elevated serum levels of MMPs may indicate that PCV may be a type of systemic vascular disease, such as atherosclerosis.24 IL-17 (interleukin-17) is also a pro-inflammatory cytokine.24 It promotes angiogenesis by enhancing choroidal endothelial cell migration and tube formation, ultimately promoting the abnormal vessel growth associated with PCV.25
Genetic variants within complement factor H have been identified that increase the risk of developing both age-related macular degeneration and PCV.26,27 Both I62V, a specific coding variant, and the ARMS2 gene have been found to be strongly associated with PCV.28,29
What are the differential diagnoses of PCV?
- Macular Degeneration: Also causes blurry vision and/or metamorphopsia in those ages 60 and up. This is commonly noted bilaterally, drusen will be present, and a thinner choroid on OCT will be noted.
- Central serous chorioretinopathy: Causes unilateral blurry vision and/or metamorphopsia in those ages 20-50. This is a sensory retinal detachment that self-resolves over months, unlike PCV.
- Pachychoroid Neovasculopathy: Both pachychoroid neovasculopathy and polypoidal chorioretinopathy will demonstrate a thicker choroid and dilated choroidal vessels (pachyvessels); however, PCV will have polypoidal lesions present. ICGA can help further distinguish the two.
- Pathological myopia: Clinical features that are present include atrophic lesions in the macula, lacquer cracks and Fuch’s spots.
- Choroidal tumors or metastases: PCV is generally slow growing with recurrent hemorrhages, while a choroidal tumor demonstrates rapid growth and vision loss. ICGA can help distinguish these two by showing the polypoidal lesions in PCV, compared with distinct vascular patterns and a large, solid mass in a choroidal tumor/metastasis.
What ancillary testing is used to diagnose PCV?
Optical Coherence Tomography (OCT) can reveal hallmark signs indicative of PCV. A sharply peaked PED or notched “thumbprint” PED, thickened choroid and subretinal fluid are some examples. A sharply peaked PED denotes a polyp, and a thickened choroid helps exclude the diagnosis of macular degeneration, as a thinner choroid is more typical.30 The dual reflective layer, known as the “double layer sign,” is seen in 59% of eyes with PCV.31 This is seen as two highly reflective lines, one in the RPE layer and the other in Bruch’s membrane, indicating the location of the choroidal vascular network.29 PCV lesions also appear as chronic, multiple “serosanguineous” detachments of the RPE and/or neurosensory retina.32 According to the Asia-Pacific Ocular Imaging Society PCV Workgroup, a combination of the following three specific OCT findings supports the diagnosis of PCV: sub-RPE “ring-like” lesions, en face complexes of RPE elevations and sharply-peaked PEDS. Utilizing these three findings as diagnostic criteria led to an accuracy rate above 80%.33
Enhanced depth imaging OCT (EDI-OCT) can help identify features to distinguish PCV from exudative age-related macular degeneration. EDI-OCT will show increased choroidal thickness in eyes with PCV, whereas it is thinner in eyes with age-related macular degeneration. OCT angiography (OCT-A) can also aid in confirming the diagnosis of PCV. OCT-A can identify the polypoidal lesions and branching vascular network, which is not noted in exudative-age related macular degeneration.34
Fluorescein angiography (FA) favors the diagnosis and classification of macular degeneration over PCV due to the inability of the fluorescein to visualize RPE subtypes, such as polyps.35 Exudative lesions display uniform hypofluorescence in the early phase with pooling in the late phase. Hemorrhagic lesions are hypofluorescent secondary to blockage of dye due to blood, causing macular PCV to often be misdiagnosed as age-related macular degeneration based solely on fluorescein angiography findings.38 Similarly, PCV may appear similar to central serous chorioretinopathy on FA, as both ocular disorders will display a thickened choroid and RPE leakage.39
Indocyanine green angiography (ICGA) remains the gold standard for diagnosing PCV. This is because indocyanine green dye absorbs and emits near-infrared light that penetrates the RPE, allowing better imaging of the choroidal structures. Compared to fluorescein dye, indocyanine green dye does not leak from the choriocapillaris, allowing choroidal lesions to be more discernible. ICGA should be considered when a dilated fundus exam reveals orange subretinal nodules, notches, or hemorrhagic or serous PED, spontaneous, massive subretinal hemorrhage, or a lack of response to anti-VEGF therapy. During the early phase of ICGA, an abnormal choroidal branching vascular network filling is noted. The polyps in terminal vessels will become hyperfluorescent. During the mid-phase, leakage from the polyps is noted along with choroidal hyperfluorescence of the lesion. During the late phase, the reversal pattern of the dye is seen with the center of the lesion becoming hypofluorescent and the surrounding area becoming hyperfluorescent. Lastly, during the very late phase, a non-leaking PCV lesion will become “washed-out” whereas a leaking PCV lesion remains hyperfluorescent.38
According to evidence-based guidelines by Tan et al, diagnosis of PCV is defined as single or multiple focal nodular areas of hyperfluorescence from choroidal circulation within 6 minutes after injection of indocyanine green dye with one or more of the following features:
- nodular appearance of the polyps on stereoscopic examination
- hyperfluorescent halo surrounding the nodule(s)
- association with branching vascular network on ICGA
- pulsation of the polyps on dynamic ICGA
- orange, subretinal nodules on color fundus photography
- massive (> 4-disc diameters) submacular hemorrhage14
What is the treatment and management of PCV?
Treatment of PCV is based on the location of the lesion, active leakage and whether it is affecting visual acuity. Approximately 50% of lesions are self-limiting and are not visually significant.39 Consider co-management with a retina specialist when the etiology is uncertain to ensure optimal visual outcome. PCV is considered to be active if any of the following findings are present: OCT and/or angiographic findings characteristic of PCV contributing to a decline in vision of at least 5 letters (EDTRS), subretinal fluid with or without intraretinal fluid, PED or subretinal hemorrhage, or angiographic evidence of leakage.6 Focal laser was used as monotherapy to ablate extrafoveal polyps; however, due to scarring and recurrence, this therapy is not used as often anymore.40 Photodynamic therapy (PDT) was also used to treat PCV; however, complications of this therapy include subretinal hemorrhage, choroidal infarction and RPE tear.41 Extrafoveal lesions tend to have complete resolution after being treated with laser photocoagulation; however, subfoveal and juxtafoveal PCV lesions are considered sight-threatening, and treatment is based on guidelines established by the EVEREST, EVEREST II and PLANET studies.42
The EVEREST study was a multicenter, double-masked, prospective study analyzing symptomatic PCV patients treated with verteporfin PDT combined with ranibizumab (Lucentis) vs. PDT alone vs. ranibizumab monotherapy. At 6 months, 71.8% treated with verteporfin PDT monotherapy had complete regression with BCVA improvement of 7.5 letters, compared to 77.8% had complete regression with PDT verteporfin combined with ranibizumab, with a BCVA improvement of 10.9 letters. Those receiving ranibizumab monotherapy achieved a complete regression rate of 28.6% and BCVA improvement of 9.2 letters. Based on these results, subfoveal and juxtafoveal PCV should be treated with either ICGA-guided verteporfin PDT or a combination of verteporfin PDT and three 0.5-mg ranibizumab intravitreal injections at monthly intervals.39 The authors recommended that combination therapy should be considered in cases of large amounts of subretinal fluid or exudation associated with PED, leakage from branch vascular network and polyps, ICGA features that are unclear between PCV and choroidal neovascularization, and/or if the lesions are a combination of PCV and choroidal neovascularization. It is recommended to monitor the subfoveal of juxtafoveal PCV 3 months after initial treatment, and if leakage is still noted on OCT/FA but lacks complete regression on ICGA, retreatment with ranibizumab is recommended. If there is incomplete regression of polyps, retreatment with verteporfin PDT monotherapy or combined with ranibizumab is recommended.6
The follow-up study, EVERST II, a multicenter, double-masked randomized trial, investigated the efficacy and safety of ranibizumab monotherapy vs combination therapy with ranibizumab and PDT in treating PCV. Participants were randomized to either ranibizumab plus PDT or ranibizumab with sham PDT and monitored over 24 months. In the combination therapy group, the average BCVA improved to 9.6 letters, and 56.6% of polyp lesions had complete regression compared to 5.5 letters in the monotherapy group, and 26.7% of polyp lesions had complete regression (consistent with the findings in the EVEREST trial). These results suggested that ranibizumab with PDT remained a superior option compared to ranibizumab monotherapy.43
The PLANET study, a randomized double-masked trial, compared intravitreal aflibercept (Eylea) monotherapy to aflibercept and rescue photodynamic therapy. All participants received treatment with intravitreal aflibercept for the first 3 months, then were randomized into either an aflibercept monotherapy group or a PDT “rescue” group, which was subdivided into a sham PDT group and a rescue PDT group. After 52 weeks, the mean BCVA improvement for the aflibercept monotherapy group was 10.7 letters and 33.1% of polyp lesions had complete regression. Comparatively, the mean BCVA improvement was 9.1 letters in the aflibercept plus PDT rescue group and 29.1% had polyp regression. This study concluded that aflibercept monotherapy was just as effective for PCV patients as combination therapy, and additional treatment with rescue PDT did not offer any significant benefit.44
What is the prognosis of PCV?
Generally, small lesions less than one disc diameter have a good visual prognosis. Factors that lead to poor prognosis/vision loss include large lesions, PED, cluster polyps, lesion recurrence and Caucasian race.45,46 Larger polyps or polyps near the macula may lead to complications such as a serous or hemorrhagic PED, subretinal hemorrhaging or exudative retinal detachment.47,48 Long-term follow-up is recommended for eyes with PCV, even in eyes with inactive lesions, as they can worsen in the future, even if stable for years. Severe vision loss can be found in about one-third of eyes with PCV due to choroidal ischemia, inflammation, RPE damage and breaks in Bruch’s membrane.49,50 Understanding the prognosis of PCV may prompt a referral for low vision rehabilitation services if the condition becomes visually significant to enhance the patient’s quality of life.
Conclusion
Polypoidal choroidal vasculopathy is an infrequent, distinct retinal disorder that is commonly misdiagnosed. Utilizing multimodal imaging can aid in appropriate diagnosis. Proper diagnosis allows patients the best opportunity for successful treatment and outcome. It is important for optometrists to understand the pathophysiology and progression of PCV to properly diagnose the condition, provide appropriate co-management and counsel patients on its prognosis.
References
- Wong CW, Yanagi Y, Lee WK, et al. Age-related macular degeneration and polypoidal choroidal vasculopathy in Asians. Prog Retin Eye Res. 2016 Jul;53:107-139. DOI: 10.1016/j.preteyeres.2016.04.002
- Yannuzzi LA, Sorenson J, Spaide RF, Lipson B. Idiopathic polypoidal choroidal vasculopathy (IPCV). Retina. 1990;10(1):1-8.
- Lim TH, Laude A, Tan CSH. Polypoidal choroidal vasculopathy: an angiographic discussion. Eye (Lond). 2010 Mar;24(3):483-90. DOI: 10.1038/eye.2009.323
- Chan WM, Lam DS, Lai TY, et al. Photodynamic therapy with verteporfin for symptomatic polypoidal choroidal vasculopathy: one-year results of a prospective case series. Ophthalmology. 2004 Aug;111(8):1576-84. DOI: 10.1016/j.ophtha.2003.12.056
- Kim JB, Nirwan RS, Kuriyan AE. Polypoidal Choroidal Vasculopathy. Curr Ophthalmol Rep. 2017 Jun;5(2):176-186. DOI: 10.1007/s40135-017-0137-0
- Koh AH; Expert PCV Panel; Chen LJ, et al. Polypoidal choroidal vasculopathy: evidence-based guidelines for clinical diagnosis and treatment. Retina. 2013 Apr;33(4):686-716. DOI: 10.1097/IAE.0b013e3182852446
- Cackett P, Yeo I, Cheung CM, et al. Relationship of smoking and cardiovascular risk factors with polypoidal choroidal vasculopathy and age-related macular degeneration in Chinese persons. Ophthalmology. 2011 May;118(5):846-52. DOI: 10.1016/j.ophtha.2010.09.026
- Laude A, Cackett PD, Vithana EN, et al. Polypoidal choroidal vasculopathy and neovascular age-related macular degeneration: same or different disease? Prog Retin Eye Res. 2010 Jan;29(1):19-29. DOI: 10.1016/j.preteyeres.2009.10.001
- Goldhardt R, Rosen BS. Polypoidal Choroidal Vasculopathy. Curr Ophthalmol Rep. 2019 Mar;7(1):66-72. DOI: 10.1007/s40135-019-00201-4
- Xu N, Xu H, Zhao M, Xu Y, Huang L. Associations of systemic, serum lipid and lipoprotein metabolic pathway gene variations with polypoidal choroidal vasculopathy in China. PLoS One. 2019 Dec 26;14(12):e0226763. DOI: 10.1371/journal.pone.0226763
- Hou J, Tao Y, Li XX, Zhao MW. Clinical characteristics of polypoidal choroidal vasculopathy in Chinese patients. Graefes Arch Clin Exp Ophthalmol. 2011 Jul;249(7):975-9. DOI: 10.1007/s00417-010-1575-7
- Tan CS, Ngo WK, Lim LW, Lim TH. A novel classification of the vascular patterns of polypoidal choroidal vasculopathy and its relation to clinical outcomes. Br J Ophthalmol. 2014 Nov;98(11):1528-33. DOI: 10.1136/bjophthalmol-2014-305059
- Yannuzzi LA, Freund KB, Goldbaum M, et al. Polypoidal choroidal vasculopathy masquerading as central serous chorioretinopathy. Ophthalmology. 2000 Apr;107(4):767-77. DOI: 10.1016/s0161-6420(99)00173-6
- Ijuin N, Tsujinaka H, Hirai H, et al. Clinical implications of pachyvessels in polypoidal choroidal vasculopathy. BMC Ophthalmol. 2020 Apr 29;20(1):170. DOI: 10.1186/s12886-020-01443-8
- Tong JP, Chan WM, Liu DT, et al. Aqueous humor levels of vascular endothelial growth factor and pigment epithelium-derived factor in polypoidal choroidal vasculopathy and choroidal neovascularization. Am J Ophthalmol. 2006 Mar;141(3):456-62. DOI: 10.1016/j.ajo.2005.10.012
- Zhou H, Zhao X, Chen Y. Plasma Cytokine Profiles in Patients With Polypoidal Choroidal Vasculopathy and Neovascular Age-Related Macular Degeneration. Asia Pac J Ophthalmol (Phila). 2022 Nov 1;11(6):536-542. DOI: 10.1097/APO.0000000000000577
- Borgersen NJ, Møller-Lorentzen T, Sørensen TL, Subhi Y. Association between C-reactive protein and polypoidal choroidal vasculopathy: a systematic review and meta-analysis. Acta Ophthalmol. 2021 Aug;99(5):470-477. DOI: 10.1111/aos.14655
- Zhang C, Zhao Y, Zhang S, Xiang Z, Shen L, Mao J.Analysis of cytokine profiles in aqueous humor based on optical coherence tomography in pachychoroid spectrum diseases. Sci Rep. 2025 Jul 1;15(1):20639. DOI: 10.1038/s41598-025-04130-0
- Liu C, Zhang S, Deng X, et al. Comparison of Intraocular Cytokine Levels of Choroidal Neovascularization Secondary to Different Retinopathies. Front Med (Lausanne). 2021 Dec 21;8:783178. DOI: 10.3389/fmed.2021.783178
- Kato Y, Oguchi Y, Omori T, et al. Complement Activation Products and Cytokines in Pachychoroid Neovasculopathy and Neovascular Age-Related Macular Degeneration. Invest Ophthalmol Vis Sci. 2020 Nov 2;61(13):39. DOI: 10.1167/iovs.61.13.39
- Hu J, Leng X, Hu Y, et al. The Features of Inflammation Factors Concentrations in Aqueous Humor of Polypoidal Choroidal Vasculopathy. PLoS One. 2016 Jan 22;11(1):e0147346. DOI: 10.1371/journal.pone.0147346
- Sasaki S, Miyazaki D, Miyake K, et al. Associations of IL-23 with polypoidal choroidal vasculopathy. Invest Ophthalmol Vis Sci. 2012 Jun 5;53(7):3424-30. DOI: 10.1167/iovs.11-7913
- Zeng R, Wen F, Zhang X, Su Y. Serum levels of matrix metalloproteinase 2 and matrix metalloproteinase 9 elevated in polypoidal choroidal vasculopathy but not in age-related macular degeneration. Mol Vis. 2013;19:729-36.
- Ardeljan D, Wang Y, Park S, et al. Interleukin-17 retinotoxicity is prevented by gene transfer of a soluble interleukin-17 receptor acting as a cytokine blocker: implications for age-related macular degeneration. PLoS One. 2014 Apr 29;9(4):e95900. DOI: 10.1371/journal.pone.0095900
- Chen Y, Zhong M, Liang L, Gu F, Peng H. Interleukin-17 induces angiogenesis in human choroidal endothelial cells in vitro. Invest Ophthalmol Vis Sci. 2014 Sep 16;55(10):6968-75. DOI: 10.1167/iovs.14-15029
- Edwards AO, Ritter R 3rd, Abel KJ, Manning A, Panhuysen C, Farrer LA. Complement factor H polymorphism and age-related macular degeneration. Science. 2005 Apr 15;308(5720):421-4. DOI: 10.1126/science.1110189
- Lee KY, Vithana EN, Mathur R, et al. Association analysis of CFH, C2, BF, and HTRA1 gene polymorphisms in Chinese patients with polypoidal choroidal vasculopathy. Invest Ophthalmol Vis Sci. 2008 Jun;49(6):2613-9. DOI: 10.1167/iovs.07-0860
- Kondo N, Honda S, Kuno S, Negi A. Coding Variant I62V in the Complement Factor H Gene Is Strongly Associated with Polypoidal Choroidal Vasculopathy. Ophthalmology. 2009 Feb;116:2. DOI: 10.1016/j.ophtha.2008.11.011
- Tanaka K, Nakayama T, Mori R, et al. Associations of complement factor H (CFH) and age-related maculopathy susceptibility 2 (ARMS2) genotypes with subtypes of polypoidal choroidal vasculopathy. Invest Ophthalmol Vis Sci. 2011 Sep 27;52(10):7441-4. DOI: 10.1167/iovs.11-7546
- Kameda T, Tsujikawa A, Otani A, et al. Polypoidal choroidal vasculopathy examined with en face optical coherence tomography. Clin Exp Ophthalmol. 2007 Sep-Oct;35(7):596-601. DOI: 10.1111/j.1442-9071.2007.01554.x
- Sato T, Kishi S, Watanabe G, Matsumoto H, Mukai R. Tomographic features of branching vascular networks in polypoidal choroidal vasculopathy. Retina. 2007 Jun;27(5):589-594. DOI: 10.1097/01.iae.0000249386.63482.05
- McCleary CD, Guier CP, Dunbar MT. Polypoidal choroidal vasculopathy. Optometry. 2004 Dec;75(12):756-70. DOI: 10.1016/s1529-1839(04)70235-3
- Cheung CMG, Lai TYY, Teo K, et al. Polypoidal Choroidal Vasculopathy: Consensus Nomenclature and Non-Indocyanine Green Angiograph Diagnostic Criteria from the Asia-Pacific Ocular Imaging Society PCV Workgroup. Ophthalmology. 2021 Mar;128(3):443-452. DOI: 10.1016/j.ophtha.2020.08.006
- Kim K, Yang J, Feuer W, et al. A Comparison Study of Polypoidal Choroidal Vasculopathy Imaged with Indocyanine Green Angiography and Swept-Source Optical Coherence Tomography Angiography. Am J Ophthalmol. 2020 Sep;217:240-251. DOI: 10.1016/j.ajo.2020.05.017
- Gomi F, Sawa M, Mitarai K, Tsujikawa M, Tano Y. Angiographic lesion of polypoidal choroidal vasculopathy on indocyanine green and fluorescein angiography. Graefes Arch Clin Exp Ophthalmol. 2007 Oct;245(10):1421-7. DOI: 10.1007/s00417-007-0564-y
- Scassellati-Sforzolini B, Mariotti C, Bryan R, Yannuzzi LA, Giuliani M, Giovannini A. Polypoidal choroidal vasculopathy in Italy. Retina. 2001;21(2):121-5. DOI: 10.1097/00006982-200104000-00004
- Manayath GJ, Shah VS, Saravanan VR, Narendran V. Polypoidal Choroidal Vasculopathy Associated With Central Serous Chorioretinopathy: Pachychoroid Spectrum of Diseases. Retina. 2018 Jun;38(6):1195-1204. DOI: 10.1097/IAE.0000000000001665
- Ciardella AP, Donsoff IM, Huang SJ, Costa DL, Yannuzzi LA. Polypoidal choroidal vasculopathy. Surv Ophthalmol. 2004 Jan-Feb;49(1):25-37. DOI: 10.1016/j.survophthal.2003.10.007
- Koh A, Lee WK, Chen LJ, et al. EVEREST study: efficacy and safety of verteporfin photodynamic therapy in combination with ranibizumab or alone versus ranibizumab monotherapy in patients with symptomatic macular polypoidal choroidal vasculopathy. Retina. 2012 Sep;32(8):1453-64. DOI: 10.1097/IAE.0b013e31824f91e8
- Lee JH, Lee WK. Choroidal neovascularization associated with focal choroidal excavation. Am J Ophthalmol. 2014 Mar;157(3):710-8. DOI: 10.1016/j.ajo.2013.12.011
- Akaza E, Yuzawa M, Matsumoto Y, Kashiwakura S, Fujita K, Mori R. Role of photodynamic therapy in polypoidal choroidal vasculopathy. Jpn J Ophthalmol. 2007 Jul-Aug;51(4):270-7. DOI: 10.1007/s10384-007-0452-3
- Nishijima K, Takahashi M, Akita J, et al. Laser photocoagulation of indocyanine green angiographically identified feeder vessels to idiopathic polypoidal choroidal vasculopathy. Am J Ophthalmol. 2004 Apr;137(4):770-3. DOI: 10.1016/j.ajo.2003.09.059
- Lim TH, Lai TYY, Takahashi K, et al. Comparison of Ranibizumab With or Without Verteporfin Photodynamic Therapy for Polypoidal Choroidal Vasculopathy: The EVEREST II Randomized Clinical Trial. JAMA Ophthalmol. 2020 Sep 1;138(9):935-942. DOI: 10.1001/jamaophthalmol.2020.2443
- Lee WK, Iida T, Ogura Y, et al. Efficacy and Safety of Intravitreal Aflibercept for Polypoidal Choroidal Vasculopathy in the PLANET Study: A Randomized Clinical Trial. JAMA Ophthalmol. 2018 Jul 1;136(7):786-793. DOI: 10.1001/jamaophthalmol.2018.1804
- Kang HM, Koh HJ. Long-term visual outcome and prognostic factors after intravitreal ranibizumab injections for polypoidal choroidal vasculopathy. Am J Ophthalmol. 2013 Oct;156(4):652-60. DOI: 10.1016/j.ajo.2013.05.038
- Kim JY, Son WY, Kim RY, Kim M, Park YG, Park YH. Recurrence and visual prognostic factors of polypoidal choroidal vasculopathy: 5-year results. Sci Rep. 2021 Nov 3;11(1):21572. DOI: 10.1038/s41598-021-00904-4
- Gomi F, Tano Y. Polypoidal choroidal vasculopathy and treatments. Curr Opin Ophthalmol. 2008 May;19(3):208-12. DOI: 10.1097/ICU.0b013e3282fb7c33
- Uyama M, Wada M, Nagai Y, et al. Polypoidal choroidal vasculopathy: natural history. Am J Ophthalmol. 2002 May;133(5):639-48. DOI: 10.1016/s0002-9394(02)01404-6
- Wong CW, Cheung CM, Mathur R, et al. Three-Year Results of Polypoidal Choroidal Vasculopathy Treated with Photodynamic Therapy: Retrospective Study and Systematic Review. Retina. 2015 Aug;35(8):1577-93. DOI: 10.1097/IAE.0000000000000499
- Sho K, Takahashi K, Yamada H, et al. Polypoidal choroidal vasculopathy: incidence, demographic features, and clinical characteristics. Arch Ophthalmol. 2003 Oct;121(10):1392-6. DOI: 10.1001/archopht.121.10.1392



