PEER REVIEWED
Primary Open Angle Glaucoma
Jasmine Wong Yumori OD, FAAO, Dipl AAO, FNAP
Abstract
This teaching case report involves a newly diagnosed patient with primary open angle glaucoma (POAG) and subsequent challenges to adherence. While this case may be appropriate as a teaching guide for optometry students at all levels because it provides a review of anatomical structures related to the aqueous humor and optic nerve and a discussion regarding key findings, differential diagnoses, epidemiology, pathophysiology, risk factors, assessment strategies, and treatment and management options associated with POAG, it would best serve third- and fourth-year students given the more complex nature of non-optimal patient reliability and adherence challenges.
Keywords
Background
This teaching case report features a newly diagnosed 71-year-old White male with primary open angle glaucoma and challenges to adherence. Key learning goals of this case, including listing differential diagnoses associated with bilateral elevated intraocular pressures in a patient without acute ocular symptoms, explaining common assessment and treatment and management options associated with primary open angle glaucoma (POAG), discussing methods to address challenges with adherence to ophthalmic medications, and identifying key components of a referral to a specialist, are covered. While this case may be appropriate as a teaching guide for optometry students at all levels because it provides a review of anatomical structures related to the aqueous humor and optic nerve and a discussion regarding key findings, differential diagnoses, epidemiology, pathophysiology, risk factors, assessment strategies, and treatment and management options associated with POAG, it would best serve third and fourth year students given the more complex nature of non-optimal patient reliability and adherence challenges. It is important for eye care providers caring for patients with POAG to recognize the clinical findings, understand assessment strategies and treatment and management options, and modify the treatment plan as necessary.
Case Description
Patient AB, a 71-year-old White male, presented to our clinic reporting long-standing eye watering and noted relief with the use of artificial tears as needed. The patient’s ocular history was significant for dry eye disease in both eyes, a developing cataract in the right eye, pseudophakia in the left eye (unknown surgical date), and anisometropia.
His medical history was significant for hyperlipidemia, hypertension and rheumatoid arthritis for which he took Lipitor, Diovan, Tenormin and Zyloprim. He had no known drug allergies. His blood pressure was 140/80 mm Hg right arm sitting with a pulse of 65 beats per minute. The rest of the ocular and medical history was negative and non-contributory. The family ocular history was significant for glaucoma in his mother. The family medical history was significant for hypertension in his father. The patient denied smoking, drinking alcohol and using narcotics. He reported neither history of ocular trauma nor use of oral or ophthalmic steroids.
His best-corrected visual acuity with a manifest refraction of +0.75-0.25×128 in the right eye and -1.25-0.75×162 in the left eye and was 20/30 in each eye. Pupils were round and reactive to light, and no afferent pupil defect was noted. Extraocular muscles were unrestricted in all gazes. Confrontation visual fields were full to finger counting in each eye.
Anterior segment evaluation by slit lamp examination revealed clear lashes and a quiet bulbar and palpebral conjunctiva. Lagophthalmos was not noted in either eye. A reduced tear meniscus was noted and a tear break-up time of 4 seconds in both eyes. Grade 2+ inferior punctate epithelial erosions were present at the inferior one-third of his corneas. No corneal infiltrates or endothelial pigmentation were noted in either eye. His irises were flat without signs of transillumination defects in both eyes. The anterior chambers appeared clear without cells or flare. Intraocular pressures were 27 mmHg in the right eye and 28 mmHg in the left eye with Goldmann applanation tonometry at 8:43AM following the instillation of one drop of Fluress.
Based on the presence of bilateral elevated intraocular pressure reading in a patient without acute ocular symptoms such as pain, the following differential diagnoses were considered as listed in Table 1, ranked starting with more likely.

Table 1: Differential diagnoses for bilateral elevated intraocular pressure readings in a patient without acute ocular symptoms Click to enlarge
The patient was dilated using 1 drop of 0.5% tetracaine, 1 drop 1% tropicamide and 1 drop 2.5% phenylephrine. Grade 1+ nuclear sclerosis in the right eye and a well-centered posterior chamber lens implant with mild posterior capsular opacification in the left eye were noted. These findings may explain why the best-corrected visual acuity was 20/30 for each eye.
A clear vitreous was noted in both eyes. An evaluation of the posterior segment was completed by slit lamp with a 90D lens and by Binocular Indirect Ophthalmoscopy with a 20D lens. Fundus assessment revealed asymmetric optic disc sizes with the right eye greater than the left eye and moderate optic nerve cupping with a cup-to-disc ratio of .70H x .85V in the right eye and .65H x .75V in the left eye. Focal rim thinning superiorly>inferiorly was noted in the right eye greater than in the left eye. The neuroretinal rims appeared pink without any splinter (Drance) hemorrhages and disc margins were distinct in both eyes. Stereo-disc photography was not available. No apparent nerve fiber layer dropout was observed. Retinal vessels appeared normal with an arteriole-venule ratio of 2/3 in both eyes. His maculae appeared flat and homogenous, and the peripheral retina was flat and intact with no pathology noted in either eye.
Primary open angle glaucoma was the highest on the list of differential diagnoses given the characteristic appearance of his optic nerve head with elevated intraocular pressures, open anterior chamber angle and normal anterior segment, including clear cornea without endothelial pigment, quiet anterior chamber, normal iris appearance without transillumination defects.
Given the patient’s eagerness to further evaluate for primary open angle glaucoma, a Humphrey visual field 24-2 SITA-FAST was completed, demonstrating borderline reliability with a possible moderate incomplete superior and inferior arcuate (versus a possible cloverleaf defect due to challenges with patient attention) in the right eye (MD -11.02dB) and non-optimal reliability but no pattern defects in the left eye (MD -6.56dB). Results are available below in Figure 1.
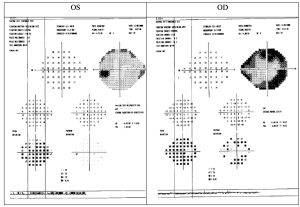
Figure 1: First Humphrey visual field 24-2 SITA-FAST Click to enlarge
The patient was educated thoroughly about the asymptomatic nature of the disease, importance of routine follow-up and future possibility of medical and/or surgical intervention. An appointment was scheduled for follow-up in 2 weeks to repeat threshold visual field testing, obtain optical coherence tomography scans of the retinal nerve fiber layer and optic nerve, re-check intraocular pressure and complete pachymetry and gonioscopy.
Dry eye disease likely secondary to the patient’s rheumatoid arthritis and related to his long-standing eye watering and anterior segment findings was also discussed. Patient AB was encouraged to continue use of artificial tears at least 4 times per day in both eyes, with no mention regarding non-preserved versus preserved. The possibility of further intervention was discussed if his dry eye symptoms and/or signs persist or worsen.
Follow-up #1
Patient AB returned 2 weeks later for a repeat threshold visual field examination, optical coherence tomography of the retinal nerve fiber layer and optic nerve, intraocular pressure check, pachymetry and gonioscopy. He reported no changes in vision or ocular discomfort. His corrected visual acuity remained stable at 20/30 in the right eye, with no improvement with pinhole acuities in the left eye. Pupils were round and reactive to light and no afferent pupil defect was noted in both eyes. Extraocular muscles were unrestricted in all gazes.
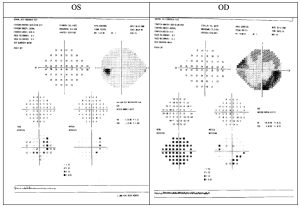
Figure 2: Repeat Humphrey visual field 24-2 SITA-FAST Click to enlarge
The patient’s second Humphrey visual field 24-2 SITA FAST results demonstrated improved reliability with a remaining incomplete inferior arcuate and superior nasal step in the right eye (MD -10.22dB) and improved reliability but again no pattern defects consistent with his optic nerve head appearance in the left eye (MD -3.75dB). Visual field results are in Figure 2.
Optical coherence tomography (OCT) was performed at this visit. As viewed in Figure 3 from the RTVue Fourier-Domain OCT System, imaging of Patient AB’s retinal nerve fiber layer was completed.
Relative retinal nerve fiber layer rim thinning was noted inferiorly and superiorly in the right eye and possibly inferiorly in the left eye with average retinal nerve fiber layer (RNFL) thicknesses of 68.59 µm in the right eye and 78.81 µm in the left eye. Asymmetric optic disc sizes in the right eye are greater than in the left eye were quantified, and the ganglion cell complex maps demonstrated thinning inferiorly and superiorly in the right eye and inferiorly in the left eye with average thicknesses of 65.61 µm in the right eye and 74.48 µm in the left eye. These findings were consistent with his optic nerve head appearance and visual field defects. Figure 3 provides the retinal nerve fiber layer, optic disc scan and both the thickness of the ganglion cell complex maps and significance map.
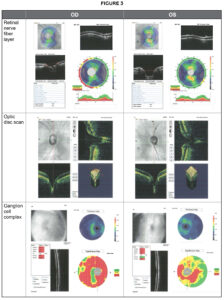
Figure 3: Optical coherence tomography Click to enlarge
Slit lamp findings were unchanged and no Drance hemorrhages were seen in both eyes at this undilated visit. Intraocular pressures were 30 mmHg in each eye with Goldmann Applanation Tonometry (GAT) following the instillation of 1 drop of Fluress. Pachymetry revealed a central corneal thickness of 500 microns in the right eye and 510 microns in the left eye. Gonioscopy was also performed, and his anterior chamber angles demonstrated a flat approach and were open to the ciliary body in all views of both eyes. Moderate pigment and no peripheral anterior synechiae or angle recession were visualized during gonioscopy.
As intraocular pressure readings remained elevated with repeatable visual field defects that corresponded to his optic nerve head appearance and optic coherence tomography results, Patient AB was diagnosed with primary open angle glaucoma which was greater in the right eye than in the left eye.
Primary open angle glaucoma, the importance of treatment, adherence and regular follow-up was re-discussed with the patient. The benefits and side effects of medical treatment were also discussed, and Travatan-Z was initiated before bedtime in both eyes. Instillation procedures including punctual occlusion were reviewed. A target intraocular pressure of a 30% reduction was set at 20 mmHg for each eye. Patient AB was instructed to return in 6 to 8 weeks for an intraocular pressure check. Patient AB was encouraged to continue use of artificial tears 4 times per day in both eyes and the patient was given personal contact information if he ran into any questions or concerns.
Follow-up #2
Patient AB returned 8 weeks later for an intraocular pressure check and presented with a chief concern of continued long-standing ocular irritation and tearing relieved by artificial tears 4 times per day in both eyes. The patient suspected that the irritation and tearing may have been exacerbated using Travatan-Z. He reported good adherence with the Travatan-Z before bedtime in both eyes and reported that his last dose was at 10:00PM. He was able to demonstrate competency in proper drop instillation technique with a bottle of artificial tears but noted difficulties with punctual occlusion. Notably, he was unable to describe both the color and size of the medication bottle and brought up concerns over the cost of Travatan-Z.
His corrected visual acuity remained stable at 20/30 in each eye with no improvement with pinhole acuities. Pupils were round and reactive to light and no afferent pupil defect was noted in both eyes. Extraocular muscles were unrestricted in all gazes and confrontation visual fields were full to finger counting in both eyes. Slit lamp findings were unchanged. His intraocular pressures were 26 mmHg in the right and left eyes with GAT. Optic nerve findings remained stable with no Drance hemorrhages seen in both eyes at this undilated visit.
Based on the patient’s dry eye symptoms and signs despite his reported continued use of artificial tears, collagen punctal plugs were inserted in both his right and left lower lid punctum. The potential for punctal plugs to also maximize the amount of Travatan-Z that stayed in his eye and thus increasing the effectiveness of his glaucoma medication was also considered.
Because Patient AB was unable to recall either the color or size of the medication bottle and he expressed concerns regarding the cost of medications, there were concerns regarding adherence. Because generic intraocular pressure-lowering medications with a once a day dosing were not yet available at the patient’s visit and given concerns relating to exacerbating dry eye symptoms and signs with the introduction of preservatives, Patient AB was kept on the Travatan-Z before bedtime in both eyes, counseled extensively on the importance of adherence, consequences of untreated glaucoma, and the possibility of additional intervention if intraocular pressure readings continued to remain above target levels. An appointment was scheduled in 2 to 4 weeks for an intraocular pressure check and dry eye disease follow-up. If the patient reported relief from symptoms with collagen punctal plugs, silicone punctual plugs would be considered. Patient AB was encouraged to continue use of artificial tears at least 4 times per day in both eyes and was requested to bring the bottle of Travatan-Z he was using at the next follow-up visit.
Follow-up #3
Patient AB returned a month later for an intraocular pressure check and dry eye follow-up. He Patient AB returned a month later for an intraocular pressure check and dry eye follow-up. He reported no relief of ocular irritation or tearing following the insertion of the punctal plugs. He reported good adherence with Travatan-Z at bedtime in both eyes (last dose 10:00PM) and artificial tears at least 4 times per day in both eyes. However, he failed to bring his bottle of Travatan-Z to the visit. His corrected visual acuity remained stable at 20/30 in each eye with no improvement with pinhole acuities.
Pupils were round and reactive to light and no afferent pupil defect was noted in both eyes. Extraocular muscles were unrestricted in all gazes and confrontation visual fields were full to finger counting in both eyes. Intraocular pressures were 26 mmHg and 25 mmHg with GAT following the instillation of 1 drop of Fluress. Slit lamp findings were unchanged and no Drance hemorrhages were seen in both eyes at this undilated visit.
Because Patient AB’s intraocular pressure remained above the target intraocular pressure despite his reported adherence with Travatan-Z, the addition of a topical beta-blocker, such as Timolol 2 times per day in both eyes, to the patient’s medication regimen was also considered. However, the patient was on an oral beta-blocker so Alphagan-P 2 times per day in both eyes was added to the patient’s regimen. The importance of treatment, adherence and regular follow-up were emphasized. The benefits and side effects of both medications were reviewed, and follow-up was scheduled in 1 to 2 months for an intraocular pressure check. The patient was encouraged to use his artificial tears at least 4 times per day in both eyes for relief of his symptoms from dry eye disease. Patient AB was also given the dosing schedule for each of his three eye drops on the back of a business card.
Follow-up #4
Two months later, Patient AB returned for an intraocular pressure check. He reported no change in vision but presented with a chief concern about the inconvenience in using multiple eye drops throughout the day. He continued to report use of Refresh Tears®, Alphagan-P and Travatan-Z but was unable to recall the dosing schedule for each medication. Intraocular pressures remained above target at 25 mmHg in each eye with GAT following the instillation of 1 drop of Fluress. His corrected visual acuity remained stable at 20/30 for each eye with no improvement with pinhole acuities.
Pupils were round and reactive to light and no afferent pupil defect was noted in both eyes. Extraocular muscles were unrestricted in all gazes and confrontation visual fields were full to finger counting in both eyes. Slit lamp findings were unchanged including at the optic nerve and no Drance hemorrhages were seen with 90D in both eyes at this undilated visit.
Because the 30% target reduction in intraocular pressure was not achieved with two topical medications (and use of his oral beta blocker) and questionable patient adherence was noted, surgical and laser intervention were considered. The benefits and risks of surgical and laser interventions were reviewed with Patient AB. Specifically, selective laser trabeculoplasty was briefly reviewed with mention of argon laser trabeculoplasty and trabeculectomy.
While Patient AB was cautious about the prospect of surgical and/or laser intervention, he was agreeable to a referral to a local glaucoma specialist. A referral letter was written and mailed to the glaucoma specialist along with the patient’s contact information and a request for their office to contact the patient for scheduling.
Educators’ Guide
Learning Objectives
At the end of this case discussion, readers should be able to:
- Identify the anatomical structures related to:
- Aqueous humor production and outflow
- The optic nerve
- List appropriate differential diagnoses for bilateral elevated intraocular pressures in a patient without acute ocular symptoms
- Describe the definition, epidemiology, pathophysiology and risk factors associated with POAG
- Describe common assessment strategies for diagnosing POAG
- Explain common treatment and management options for POAG
- Discuss methods to address challenges with adherence to ophthalmic medications
- Identify key components of a referral to a specialist
Key Concepts
- Describe an abnormal optic nerve in a systematic manner.
- Recognize clinical findings seen in POAG.
- List common assessment strategies for diagnosing POAG.
- Identify treatment and management options for POAG.
Discussion Questions
- Knowledge and concepts required for critical review of the case
- What anatomical structures are within the anterior chamber angle and optic nerve?
- Describe the epidemiology, pathophysiology and risk factors associated with POAG.
- Explain key clinical findings that are found in patients with POAG.
- Differential diagnoses
- List appropriate differential diagnoses for elevated intraocular pressure and/or abnormal optic nerve appearance.
- What assessment strategies are helpful in formulating a definitive diagnosis?
- Disease treatment and management
- What are common initial treatment options for POAG?
- List alternative/additional treatment options and discuss the rationale behind each one of them.
- What are potential complications associated with anti-glaucoma therapies?
- When should laser and surgical procedures be considered in POAG?
- Critical thinking
- How would you have managed this case? Justify your answer based on the findings.
- What are indications of poor adherence to a treatment regimen? How can these be addressed?
Learning Activity
- Assign the case with selected discussion questions as a homework assignment during a didactic course featuring glaucoma or as part of a clinical assignment.
- Require students to provide at least 2-4 recent, peer-reviewed articles to support their answers.
- Block off 1-2 hours during class or clinic time to allow students to present their answers and learn from each other; encourage questions from other students by providing participation credit incentives.
Learning Assessment
- Assess knowledge regarding POAG and differential diagnoses by use of multiple choice and/or short answer questions.
- Facilitate demonstration of clinical skills associated with a glaucoma evaluation including slit lamp examination with optic nerve assessment, gonioscopy and pachymetry, and systematically describing abnormal clinical findings and relationships to ancillary testing such as threshold visual fields and optical coherence tomography.
- Incorporate the case into clinical or didactic discussions.
Discussion
Identify anatomical structures
Related to aqueous humor
A transparent fluid located in the anterior and posterior chambers of the eye, the aqueous humor facilitates circulation of nutrition, removes excretory products and maintains pressure within the eye. Aqueous humor is produced at the ciliary body, which is located at the anterior portion of the scleral spur. The aqueous humor travels through the ciliary body, around the lens, through the pupils, into the anterior chamber, and passively leaves the eye through either the trabecular meshwork (conventional) or the uveoscleral pathway (non-conventional).2 When aqueous humor travels through the trabecular meshwork, it crosses the inner wall of Schlemm’s canal and drains through the aqueous and episcleral veins.3 When aqueous humor travels through the uveoscleral pathway, it enters the connective tissue between the ciliary muscle bundles, goes through the suprachoroidal space, and leaves through the sclera.4 The uveoscleral pathway accounts for around 10% of aqueous outflow.5
The average intraocular pressure is approximately 15mmHg in a healthy eye2; a dis-equilibrium between the production and drainage of aqueous humor, which results in an increase in intraocular pressure, is a major risk factor for glaucoma progression.6 As such, current pharmacological and surgical therapies to lower intraocular pressure and thus reduce the risk of vision loss from glaucoma are mainly focused on reducing aqueous production or increasing aqueous humor outflow.
Related to the optic nerve
The optic nerve is the only tract in the central nervous that can be clinically visualized and is where the axons of retinal ganglion cells convergence.7 These axons form the retinal nerve fiber layer and intrapapillary neuroretinal rim.8 In normal eyes, the neuroretinal rim is broadest inferiorly, followed by superiorly, nasally and lastly temporally9, which forms the basis of the ISNT rule.10
Changes to the nerve fibers lead to visual field defects. Specifically, damage to the macular fibers that enter the disc temporally can lead to central, centrocecal, and paracentral scotomas. Alterations to the fibers from the temporal retina that enter the disc superiorly and inferiorly cause arcuate and nasal step scotomas, while changes to the fibers that enter the disc nasally can lead to wedge-shaped temporal scotomas. Defects in the thickness of nerve fibers can also be appreciated through optical coherence tomography and scanning laser polarimetry.7 Because there are no photoreceptors at the optic nerve, it represents a blind spot in vision.
List differential diagnoses for bilateral elevated intraocular pressures in a patient without acute ocular symptoms
Normal intraocular pressure ranges from 12-21 mmHg; a reading of 22 mmHg or higher is considered elevated.11 The top three differential diagnoses for bilateral elevated intraocular pressures in a patient without acute ocular symptoms are: ocular hypertension, primary open angle glaucoma and secondary open-angle glaucoma. Chronic narrow angle could also be a possibility for increased IOP without symptoms.
A diagnosis of ocular hypertension is applied in a patient that presents with elevated intraocular pressures without acute ocular symptoms and no optic nerve head damage and no visual field defects.1
Clinical findings that support the diagnosis of primary open angle glaucoma include elevated intraocular pressures with characteristic optic nerve head changes, nerve fiber layer defects, and visual field loss despite a normal-appearing open angle as viewed by gonioscopy. Optic nerve head changes can include cup: disc enlargement and/or asymmetry, the presence of thinning and/or notching of the neurosensory rim of the optic nerve, splinter (Drance) hemorrhages and nerve fiber layer defects.1
Bilateral elevated intraocular pressures in a patient without acute ocular symptoms can also be due to secondary open-angle glaucoma, such as pseudoexfoliation (or exfoliation) and pigmentary glaucoma. Signs of pseudoexfoliation glaucoma include: iris transillumination defects, exfoliative material on the lens capsule and/or on the iris, anterior hyaloid, or zonules, and increased, oftentimes patchy, pigment deposit on the trabecular meshwork and at Schwalbe line with gonioscopy.12 Signs of pigmentary dispersion glaucoma include: pigment on the corneal endothelium (Krukenberg spindle), mid-peripheral iris transillumination defects, and significant gonioscopy findings including increased homogenous pigment in the trabecular meshwork and/or irregular pigment deposit on the trabecular meshwork, particularly inferiorly, anterior to Schwalbe line (Sampaolesi line).13 It is important to mention that patients with dark irides may occasionally have pigment on or slightly above Schwalbe’s line and not have glaucoma. Furthermore, patients with either pigmentary dispersion or pseudoexfoliation glaucoma must demonstrate a characteristic optic nerve head appearance and corresponding visual field defects consistent with glaucoma. If such findings are not found, the patient is considered not to have glaucoma but may remain a glaucoma suspect.
Describe the definition, epidemiology, pathophysiology and risk factors associated with POAG
POAG is a “chronic, progressive disease that most often presents with characteristic optic nerve (ON) damage, retinal nerve fiber layer (NFL) defects and subsequent visual field (VF) loss” that primarily occurs bilaterally but asymmetrically and mostly in adults that have elevated intraocular pressure (IOP).14
Glaucoma has been reported to affect 2.2 million Americans and is the third most common cause of blindness in the United States.15 Primary open angle glaucoma (POAG) is the most common type of glaucoma. Primary open angle glaucoma is an adult-onset, typically bilateral disorder that produces characteristic optic nerve head and visual field changes.
While the etiology of primary open angle glaucoma is constantly undergoing further investigation, there is strong evidence that suggests glaucoma is caused by damage to the retinal ganglion cell axons within the lamina cribrosa of the optic nerve head16,17 leading to gradual vision loss. Two main theories have emerged to explain the mechanism of ganglion cell dysfunction: mechanical and vasogenic. According to the mechanical theory, pressure-induced deformation of the lamina cribrosa structure leads to blockage of axon transport and subsequent axon damage.18 According to the vasogenic theory, vascular insufficiency at the optic nerve head results in hypoxia and decreased nutrition to optic nerve axons ultimately with ganglion cell death.19
According to the Ocular Hypertension Treatment Study, risk factors for primary open angle glaucoma include: increased age, African-American heritage, increased intraocular pressure, decreased corneal thickness, larger vertical cup-to-disc ratio, larger horizontal cup-to-disc ratio, heart disease and greater Humphrey visual field pattern standard deviation.20 The Los Angeles Latino Eye Study suggests a similar prevalence of open angle glaucoma among Latinos as for those with African American heritage.21 Table 2 briefly summarizes these risk factors for primary open angle glaucoma. The primary modifiable risk factor is reducing intraocular pressure22,23, which is most addressed with prescription ophthalmic medications. Because patients with primary open angle glaucoma are typically asymptomatic until later stages of the disease24, adherence is oftentimes a challenge.
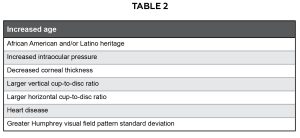
Table 2: Risk factors for primary open angle glaucoma Click to enlarge
Describe common assessment strategies for diagnosing POAG
Intraocular pressure
Intraocular pressure is determined by the balance between the rate of aqueous secretion and the rate of outflow. A common method to measure intraocular pressure is via Goldmann applanation tonometry, which has been shown to have excellent test-retest repeatability.25 Short-term variations are associated with factors such as diurnal variation and posture.
Specifically, a maximum intraocular pressure value is usually detected in the early morning and the lowest value in the early afternoon.26 Head position is also a concern since an erect sleeping position lowers perfusion pressure, which could jeopardize perfusion to the nerve. Another factor that has been evaluated closely is the association between intraocular pressure and corneal thickness. Corneal thickness measurement is important to obtain a more accurate understanding of the intraocular pressure and risk for the development of glaucoma.27 While there are various factors that may alter our assessment of intraocular pressure, decreasing intraocular pressure early and consistently is the only reported means of reducing progressive visual field loss. In particular, the Collaborative Normal Tension Glaucoma Study found a slower rate of visual field loss with a 30% or more reduction of intraocular pressure.22
Anterior chamber angle
The main four structures that are seen in normal angles include, from anterior to posterior, are:28
- Schwalbe’s line, which appears as a thin, translucent line and represents collagen condensation of Descemet’s membrane. The presence of pigmentation at Schwalbe’s line can be suggestive of pigmentary dispersion or pseudo-exfoliative syndrome and is known as Sampaolesi’s line; pigmentation can also be found in angle-closure glaucoma, due to the lens moving forward and zonule involvement, and in previous trauma, which is typically accompanied by other signs such as angle recession, dis-inserted or ruptured iris processes and/or pupillary sphincter ruptures. Pigmentation is not expected in patients with primary open angle glaucoma
- Trabecular meshwork, which has a dull grey appearance at the top half and a pigmented lower half
- Scleral spur, which is appreciated as a thin white line and represents where Schlemm’s canal rests and the longitudinal ciliary muscle inserts
- Ciliary body, which is a pale grey to brown appearance
Angle assessment not only includes visualization of structures and thus determination of the level of iris insertion, but also an evaluation of the width of the angle, approach of the peripheral iris, amount of pigment, and any signs not consistent with primary open angle glaucoma, such as pseudo-exfoliative material, peripheral anterior synechiae, neovascularization, or signs of angle recession.
The oldest and most common method to evaluate characteristics of an anterior chamber angle is gonioscopy, which is either direct, because it involves the use of a contact gonio lens and provides a direct, magnified view, or indirect, because it involves the use of a contact gonio lens and provides an inverted view of the anterior chamber angle.29
While the Van Herrick grading system, which involves placing a slit lamp beam of light obliquely and comparing the width of the light beam and space between the corneal endothelium and anterior iris surface28, can be used as an initial screening tool to detect for possible narrow angles and devices such as ultra-biomicroscopy, anterior segment optical coherence tomography and Pentacam can be used, gonioscopy is essential. Gonioscopy allows for the differentiation between both open angle and closed angle glaucoma and primary from secondary glaucomas.15
Visual field
Various test strategies exist for evaluating visual field changes in patients with primary open angle glaucoma. A commonly used instrument is the Humphrey Field Analyzer, an automated computer-driven perimeter that offers various threshold strategies; the 24-2 testing strategy is more commonly used for patients with glaucoma.30 Visual field defects typically associated with primary open angle glaucoma are those that correspond to optic nerve fiber bundle defects: arcuate defects, nasal steps and paracentral defects.31 Visual field severity classification systems for glaucoma have been established and are often used in clinical practice; the most common criteria to stage glaucoma is that of Hodapp, Parish and Anderson (H-P-A).32 The H-P-A classification system considers the overall extend of the damage using the MD value, the number of defective points, and the proximity of the defect(s) to fixation.33
Other non-specific visual field defects seen in patients with glaucoma may include a generalized depression in sensitivity or increased diffuse variability. Cataracts and other age-related changes in the retina can cause a generalized depression in visual field sensitivity. Increased diffuse variability as seen in early glaucomatous visual fields makes determining the diagnosis and judging progression particularly difficult. Variability necessitates keeping a consistent test strategy for a patient and, most importantly, evaluating visual field defects and their correspondence to the optic nerve head appearance.
Optic nerve head appearance
Abnormality in the appearance of the optic nerve is oftentimes the earliest sign of glaucoma.34 It is important to look for cup: disc enlargement and/or asymmetry and the presence of thinning and/or notching of the neurosensory rim of the optic nerve. While deviations from the ISNT rule, which refers to the inferior neuroretinal rim typically being the broadest, followed by the superior, nasal and then temporal, has increased clinicians’ suspicions of glaucoma, a more recent variant, the IST and IS rule, has been championed as more valid.35 Other signs of glaucomatous damage include Drance hemorrhages,36 nerve fiber layer defects, changes to the circumlinear vessels and peripapillary atrophy.37 A stereoscopic evaluation of the optic nerve head appearance in a fully dilated patient, typically using an indirect technique with a 60D, 78D, or 90D lens with the slit lamp, continues to be the gold standard to evaluate for signs of glaucomatous progression.
Another technique to assess for changes to the structure of the optic nerve head is the use of optical coherence tomography. OCT provides cross-sectional images of the optic nerve and retinal nerve fiber layer (RNFL).38 Studies have demonstrated a correlation between retinal nerve fiber layer thickness and visual field defects with Time Domain OCT. Recent research has indicated that Fourier Domain OCT measurements of cross-sectional peri-papillary RNFL thicknesses have similar diagnostic performance as Time Domain OCT measurements with respect to glaucoma detection.39 Practitioners commonly focus on the RNFL thickness average analysis report, which displays the results of the RNFL circumpapillary scans (NFL thickness versus position around the optic nerve head) taken at a diameter of 3.45 mm. This diameter is used because it intercepts all of the nerve fibers from the optic disc while avoiding inaccuracies from peripapillary atrophy.40 Scans are displayed on a graph broken up into the superior, inferior, temporal and nasal quadrants. The thickest RNFL corresponds to the superotemporal and inferotemporal nerve fiber bundles. Each region is color-coded based on a comparison to normative data. Green indicates the 5th to 95th percentile, or statistically “normal” thickness within the population. Yellow specifies 1st to 5th percentile while red indicates the 0 to 1st percentile, indicating borderline and abnormal RNFL thickness, respectively. Values are calculated in the four quadrants around the disc as well as an average NFL thickness around the entire disc. Optic disc scans including cross-sectional images of the optic disc and 3D view assist in demonstrating disc boundaries and the optic disc contour.
Thickness of the ganglion cell complex (GCC) thickness maps and significance maps that illustrate the percent loss value at each location from an age-matched normative database may also be useful to detecting early changes from glaucoma. More research is needed to compare the ganglion cell complex (GCC) values with visual field defects in glaucoma and the normative database is limited but expanding. Research to date has indicated that the retinal ganglion cells, encompassing the retinal nerve fiber layer, ganglion cell layer and inner plexiform layer, become thinner in glaucoma.41
Explain common treatment and management options for POAG
Lowering intraocular pressure, either by decreasing aqueous humor production and/ or increasing aqueous humor outflow, is associated with a reduced progression of visual field defects42,43,44 and is the standard treatment for POAG. Currently intraocular pressure is the only risk factor for glaucoma that is currently amenable to therapy. The main categories of medications used to treat primary open angle glaucoma are: prostaglandin analogs, beta-blockers, adrenergic agonists, carbonic anhydrase inhibitors, rho kinase inhibitors, miotic agents, epinephrine compounds and osmotic diuretics. Laser and surgical intervention are also available. Table 3 features common drug classes with select examples and dosages for open angle glaucoma. While pharmacologic intervention has traditionally been considered the first line of treatment,45 laser therapy and surgical intervention may also be considered.15
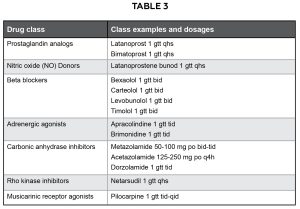
Table 3: Common drug classes with select examples and dosages for open angle glaucoma Click to enlarge
Prostaglandin analogs
Because this category is typically effective, well-tolerated and requires only a once daily dosing for 24-hour efficacy, prostaglandin analogs, such as latanoprost, are currently the most common first line medications prescribed to reduce intraocular pressure for treatment of primary open angle glaucoma.46,47 This group of drugs lowers intraocular pressure by enhancing uveoscleral outflow48 and is also suspected to increase the ocular perfusion pressure.49 Studies indicate the prostaglandins lower intraocular pressure by around 30 percent when dosed once a day.50 Additionally, the once-a-day dosing regimen is better-tolerated by patients and encourages adherence. Ocular side effects can include hypertrichosis51, which is seen in 50% of patients within 12 months46, increased peri-ocular skin pigmentation5, deepening of the upper eyelid sulcus52, cystoid macular edema53, and hyperemia15; contraindications include a history of uveitis, cystoid macular edema and herpes simplex keratitis.
Nitric Oxide (NO) donors
Nitric oxide donors such as latanoprostene bunod are suspected to increase outflow of aqueous humor through the uveoscleral pathway (by up-regulating matrix metalloproteinase expression and remodeling the extracellular matrix of the ciliary muscle) and via the trabecular meshwork (by inducing cytoskeletal relaxation via the soluble guanylyl cyclase-cyclic guanosine monophosphate signaling pathway).54
Beta-blockers
Second choice is typically the topical beta-blockers, such as timolol, which bind to beta-receptors in the ciliary body to decrease aqueous formation and thus lower intraocular pressure.46 While twice a day dosing has been shown to be most effective55, once a daily dosing in the morning has been proposed either as solo or adjunctive therapy46 because of systemic concerns from absorption that may occur via the nasolacrimal mucosa.56 In particular, studies have suggested that beta-adrenergic antagonists may induce bronchoconstriction, brady-arrhythmias and nocturnal systemic hypotension57,58, the latter of which is concerning because systemic hypotension can lead to optic nerve hypo-perfusion and increased glaucoma progression.59 Selective beta-blockers are available but may be less effective at reducing intraocular pressure.60 Reduced efficacy and potential for more side-effects in prescribing topical beta blockers in patients who are also being treated with systemic beta blockers are also concerns61 that may require either modification of treatment regimens or consideration for change in intervention.
Adrenergic agonists
Adrenergic agonists, such as brimonidine, cause both a reduction of aqueous humor production and increase in uveoscleral outflow, thereby lowering intraocular pressure.62 While effective as a short-term therapeutic means to reduce intraocular pressure, adrenergic agonists are less tolerated long-term due to high rates of conjunctival hyperemia, high frequency of allergic reactions and tachyphylaxis, and frequent dosing schedule (typically 3 times a day).63,64 Use of brominidine purite 0.15%, which contains a more gentle preservative and decreased concentration, has shown to reduce rates of allergic reaction.65
Contraindications include patients that are on monoamine oxidase inhibitors and central nervous system depressants. Studies on brimonidine, an alpha-2 adrenergic agonist, have shown that brimonidine is one thousand times more selective for alpha-2 than alpha-1 adrenergic receptors66 and thus better tolerated.67 Studies have furthermore demonstrated possible neuroprotective properties;68 however, it may not be as effective in monotherapy at lowering intraocular pressure when compared to latanoprost, a prostaglandin analog.69 Brimonidine demonstrates an additive effect with prostaglandin analogs such as latanoprost69 and is often used in combination therapy.
Carbonic anhydrase inhibitors
Carbonic anhydrase inhibitors (CAIs), which are available in both oral and topical forms, reduce intraocular pressure by reducing bicarbonate and thereby water within the aqueous humor. This decreases aqueous volume and lowers intraocular pressure.70 Although this category of medications is effective at lowering intraocular pressure, CAIs are typically only used in for short-term control of acutely elevated intraocular pressure or as third- or fourth-line agents.46 Concerns with CAIs include their multiple dosing schedules and poor safety profiles, particularly with oral preparations. For example, acetazolamide (Diamox) is usually given orally in doses of 125-250 mg every 4 hours and can cause rare but life-threatening issues such as electrolyte imbalances and Stevens-Johnson syndrome.71 Side effects of the oral regimen include a metallic taste, malaise and kidney stones; patients using the topical formulations can also report a bitter taste, burning and present with secondary corneal edema.72 Sulfonamide allergies are a relative contraindication for both the oral and topical formulations.
Rho kinase inhibitors
Rho kinase inhibitors, such as netarsudil, cause a relaxation of the trabecular meshwork, enhancing aqueous humor outflow and thus lowering intraocular pressure.73 Netarsudil is also suspected to act as a norepinephrine transport inhibitor, which causes constriction of ocular vascular structures, including blood flow to the ciliary processes, thus inhibiting production of aqueous humor.74 More than half of patients in clinical trial settings report conjunctival hyperemia and around 20% present with other side effects such as corneal verticillata.75
Muscarinic receptor agonists
While muscarinic receptor agonists, such as topical pilocarpine, can increase parasympathetic tone of the ciliary muscle, thereby increasing trabecular outflow76 and reducing intraocular pressure, they are most commonly used as short-term preparatory agents before laser treatment for glaucoma or in cases of acute angle closure.46 Significant ocular side effects (stinging, irritation and blurred vision), systemic side effects (headache, sweating, salivation and bradycardia), and difficult dosing schedule (up to 4 times per day) limit its use.
Methods to address challenges with adherence to anti-glaucoma medications
As glaucoma is typically an asymptomatic disease, adherence is an important issue in the prevention of visual consequences secondary to glaucoma.77 In some patients there may be little motivation for a lifelong therapeutic regimen.78 In others, adherence can be related to difficulties with obtaining the medication, problems successfully instilling it into the eye and/or challenges in remember to use the medication at the appropriate time each and every day.79 Rates of non-adherence have been cited ranging from 30-80%.80,81 Barriers particularly associated with poor adherence include low self-efficacy, forgetfulness and difficulty with drop administration and the medication schedule.82
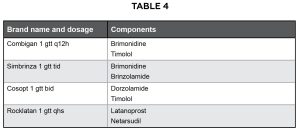
Table 4: Examples of fixed combination medications for open angle glaucoma Click to enlarge
Providers can work to address these barriers, such as simplifying the dosing schedule. Patients who used eye drops once a day were more likely to comply than those using eye drops twice a day or more.83 Other examples of methods that practitioners can use to increase adherence to glaucoma medications include using eye drops with milder side effects, adding medication reminders and recommending drop assistance devices.84 For patients that require the use multiple glaucoma medications to adequately reduce intraocular pressure, fixed combination medications can simply dosing schedules and improve adherence. Table 4 shows examples of fixed combination medications for open angle glaucoma.
With respect to Patient AB, he was started on Travatan-Z, a prostaglandin analog, because of its simple dosing regimen and known efficacy. The target pressure was set to 20 mmHg in the right eye and in the left eye, a 30% reduction from Patient AB’s maximum measured intraocular pressure. A 30% reduction was established based on lessons from the Ocular Hypertension Treatment,43 Early Manifest Glaucoma Trial,85 Collaboratively Initial Glaucoma Treatment Study,86 and Collaborative Normal Tension Glaucoma Treatment Study, with the latter showing that 30% lowering reduced the risk of glaucoma progression to 12% in the study group compared to 35% in the control arm.87
Because his intraocular pressure was not at target levels after treatment initiation, the risks and benefits of additional medications were weighed and due to its additive effects with prostaglandin analogs, less frequent dosing regimen and lack of interaction with his other medications Alphagan-P, an adrenergic agonist, was added. The addition of a beta-blocker was avoided due to Patient AB’s long-standing and successful use of an oral beta-blocker to control his hypertension. When Patient AB returned, he reported difficulty in remembering the dosing regimen for both drops despite extensive education and written dosing schedules. Furthermore, because an appropriate reduction in intraocular pressure was still not demonstrated a consultation for consideration for surgical or laser intervention was placed to reach an appropriate intraocular pressure level and thereby reducing the risk of glaucomatous progression.
Laser and surgical procedures
While medical intervention by means of prescription eye drops is typically considered first line of treatment45 for patients with open angle glaucoma, if adherence to medication(s) becomes a problem or intraocular pressures are not adequately reduced and/or there is a rapid rate of destruction and advanced damage despite good patient adherence, laser and/or surgical intervention may be recommended to reduce intraocular pressure and thus decrease glaucomatous progression.88,89
Trabeculoplasty, which involves the application of laser spots to the trabecular meshwork leads to an increase in outflow facility of the aqueous humor and subsequent decrease in intraocular pressure.90 There are two main forms that are used to treat open angle glaucoma: argon laser trabeculoplasty and selective laser trabeculoplasty. Argon laser trabeculoplasty (ALT) involves laser-induced thermal burns of the trabecular meshwork, causing contraction and thus increase in aqueous outflow.91 Selective laser trabeculoplasty (SLT) involves selective photothermolysis of pigmented trabecular meshwork cells. Because SLT causes no histologic scarring or damage to the trabecular meshwork and because repeat treatments are possible, SLT is preferred over ALT.92 According to the Laser in Glaucoma and Ocular Hypertension (LiGHT) Trial, more than 70% of SLT-treated eyes achieved adequate IOP control without the need for medical or surgical treatment after 6 years of treatment and monitoring and is the recommended first-line treatment in the United Kingdom and is considered first-line treatment alongside IOP-lowering eye drops in the European Union and the United States.93
Trabeculectomy is currently considered the gold standard surgical intervention94 and involves the creation of an alternative drainage pathway for aqueous humor outflow through a drainage bleb in the conjunctiva. Blebs are commonly positioned superiorly because the upper eyelid can provide protection, thus reducing the risk of infection. While use of anti-metabolites such as mitomycin C can improve long-term bleb success, risks associated with trabeculectomy include hypotony, bleb leak and endophthalmitis.95
Tube shunt surgery is also a method to reduce intraocular pressure, however it is typically reserved for patients with refractory glaucoma. With tube shunt surgery, a tube is placed in the anterior chamber and is connected to a plate, which acts as a drainage reservoir and is anchored onto the sclera behind the superior and lateral recti. Concerns with tube shunt surgery include hypotony, particularly initially after surgery, and possible risk of bullous keratopathy.96
Minimally-invasive glaucoma surgeries (MIGS) have been increasing in popularity, particularly in patients with mild to moderate glaucoma and/or along with cataract surgery, because they are safer and less traumatic than other forms of glaucoma surgery.97 There are three main types of MIGS devices:98
- Trabecular, which improve trabecular outflow through Schlemm’s canal
- Suprachoroidal, which connect the anterior chamber and suprachoroidal space to improve uveoscleral outflow
- Subconjunctival, which offer an alternative pathway of aqueous outflow to the subconjunctival space
Additional but less commonly used treatment options for open angle glaucoma include laser cyclophotocoagulation of the ciliary body, cyclocryotherapy and cyclodialysis.
Key components of referral to a specialist
A referral is “a request from one physician to another to assume responsibility for management of one or more of a patient’s specified problems”99 and can be completed in person, electronically, or, most commonly, through written communication.100 Regardless of the referral method, clear and well-organized communication is essential101,102 and can be achieved by using structured referral sheets102 or standardized communication methods such as the SBAR (Situation, Background, Assessment and Recommendation) technique.103 Using the SBAR technique as a structure, key referral information includes:
- Situation: Patient demographics and specific reason for the referral
- Background: Patient history, medications and allergies, appropriate diagnostic studies
- Assessment: Referring provider’s analysis of the patient
- Recommendation: Specific action that is requested or recommended by the referring provider
Educating the patient regarding their referral and scheduling a follow-up examination help to ensure that the patient’s needs are being met.
Conclusion
This case demonstrates the importance of individualized care for patients with primary open angle glaucoma. This case emphasizes the importance of recognizing clinical findings and ruling out other possible causes, understanding the course of the disease and constantly evaluating the need for modification of treatment plans. Key learning goals of this case, including listing differential diagnoses associated with bilateral elevated intraocular pressures in a patient without acute ocular symptoms, explaining common assessment and treatment and management options associated with POAG, discussing methods to address challenges with adherence to ophthalmic medications, and identifying key components of a referral to a specialist, are covered.
In most cases of primary open angle glaucoma, intraocular pressure can be reduced with topical therapy alone to a target pressure, which studies demonstrate reduces glaucomatous progression. However, in some instances, particularly with patients who have difficulty with adherence or demonstrate progressive damage on pharmacological interventions, a referral to a glaucoma specialist for consideration of laser or surgical intervention may be necessary. It is important for clinicians to recognize clinical findings, understand the course of the disease, reinforce the need for adherence and modify the treatment plan as necessary.
Acknowledgements
Dr Yumori is grateful for Dr. Phillip Kwok for his feedback, as well as Drs. Aurora Denial, Elizabeth Hoppe and Marlee Spafford, who provided feedback on an earlier version of the manuscript.
References
- Gervasion KA, Peck RJ, Fathy CA, Sivalingam, MD. The Willis Eye Manual: Office and Emergency Room Diagnosis and Treatment of Eye Disease. 8th Riverwoods, IL: Lippincott Williams & Wilkins, a Wolters Kluwer business; 2021.
- Goel M, Picciani RG, Lee RK, Bhattacharya SK. Aqueous humor dynamics: a review. Open Opthalmol J. 2010 Sep 3;4:52-9. DOI: 10.2174/1874364101004010052
- Ascher KW. [Veins of the aqueous humor in glaucoma]. Boll Ocul. 1954 Mar;33(3):129-44.
- Goel M, Picciani RG, Lee RK, Bhattacharya SK. Aqueous Humor Dynamics: Open Ophthalmol J. 2010 Sep 3;52-59. DOI: 2174/1874364101004010052
- Winkler NS, Fautsch MP. Effects of prostaglandin analogues on aqueous humor outflow pathways. J Ocul Pharmacol Ther. 2014 Mar-Apr;30(2-3):102-0. DOI: 10.1089/jop.2013.0179
- Kass MA, Hart Jr WM, Gordon M, Miller JP. Risk factors favoring the development of glaucomatous visual field loss in ocular hypertension. Surv Opthalmol. 1980 Nov-Dec;25(3):155-62. DOI: 10.1016/0039-6257(80)90092-2
- Salazar J, Ramirez A, De Hoz R, et al. Anatomy of the Human Optic Nerve: Structure and Function. In: Ferreri FM, editor. Optic Nerve. London: IntechOpen Limited; 2019. DOI: 10.5772/intechopen.79827
- Jonas JP, Budde WM, Lang P. Neuroretinal rim width ratios in morphological glaucoma diagnosis. Br J Ophthalmol. 1998 Dec;82(12):1366-71. DOI: 10.1136/bjo.82.12.1366
- Sihota R, Angmo D, Sharma S, et al. Is the ISNT rule violated in early primary open-angle glaucoma–a scanning laser tomography study. Eye (Lond). 2008 Jun;22(6):819-24.
- Jonas JB, Gusek GC, Naumann GO. Optic disc, cup and neuroretinal rim size, configuration and correlations in normal eyes. Invest Ophthalmol Vis Sci. 1988 Jul;29(7):1151-8.
- Jonas JB, Aung T, Bourne RR, et al. Glaucoma. Lancet. 2017 Nov 11;390(10108):2183-93.
- Sternfeld A, Rosner M, Weinberger D, et al. Diagnosis of Pseudoexfoliation Syndrome in Pseudophakic Patients. Ophthalmic Res. 2021;64(1):28-33.
- Scuderi G, contestabile MT, Carnovale C, et al. Pigment dispersion syndrome and pigmentary glaucoma: a review and update. Int Ophthalmol. 2019 Jul;39(7):1651-62.
- Fingeret M, et al. Optometric Clinical Practice Guideline: Care of the Patient with Open Angle Glaucoma [Internet]. St. Louis, MO: American Optometric Association; c2010 [cited 2021 Feb 4]. Available from: https://www.aoa.org/AOA/Documents/Practice%20Management/Clinical%20Guidelines/Consensus-based%20guidelines/Care%20of%20the%20Patient%20with%20Open%20Angle%20Glaucoma.pdf.
- Fingeret M, et al. Care of the Patient with Open Angle Glaucoma. American Optometric Association Optometric Clinical Practice Guideline; 2011.
- Gaasterland D, Tanishima T, Kuwabara T. Axoplasmic flow during chronic experimental glaucoma. 1. Light and electron microscopic studies of the monkey optic nervehead during development of glaucomatous cupping. Invest Ophthalmol Vis Sci. 1978 Sep;17(9):838-46.
- Burgoyne CF, Downs JC, Bellezza AJ, et al. Three-dimensional reconstruction of normal and early glaucoma monkey optic nerve head connective tissues. Invest Ophthalmol Vis Sci. 2004 Dec;45(12):4388-99. DOI: 10.1167/iovs.04-0690
- Quigley HA. Neuronal death in glaucoma. Prog Retin Eye Res. 1999 Jan;18(1):39-57.
- Osborne NN, Wood JP, Chidlow G, et al. A hypothesis to explain ganglion cell death caused by vascular insults at the optic nerve head: possible implication for the treatment of glaucoma. Br J Ophthalmol. 2001 Oct;85(10):1252-9.
- Gordon MO, Kass MA, Heuer Jr DK, et al. The Ocular Hypertension Treatment Study: baseline factors that predict the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002 Jun;120(6):714-20; discussion 829-30.
- Varma R, Choudhury F, Torres M, et al. Four-year incidence of open-angle glaucoma and ocular hypertension: the Los Angeles Latino Eye Study. Am J Ophthalmol. 2012 Aug;154(2):315-25.e1.
- Anderson DR, Normal Tension Glaucoma Study Group. Collaborative normal tension glaucoma study. Curr Opin Ophthalmol. 2003 Apr;14(2):86-90.
- Leske MC. The epidemiology of open-angle glaucoma: a review. Am J Epidemiol. 1983 Aug;118(2):166-91.
- Lee BL, Bloom W, Harizman N, et al. The Glaucoma Symptom Scale. A brief index of glaucoma-specific symptoms. Arch Ophthalmol. 1998 Jul;116(7):861-6.
- Thorburn W. The accuracy of clinical applanation tonometry. Acta Ophthalmol (Copenh). 1978 Feb;56(1):1-5.
- Sacca SC, Tognon MS, Marletta A, et al. Fluctuations of intraocular pressure during the day in open-angle glaucoma, normal-tension glaucoma and normal subjects. Ophthalmologica. 1998;212(2):115-9.
- Doughty MJ, Zaman ML. Human corneal thickness and its impact on intraocular pressure measures: a review and meta-analysis approach. Surv Ophthalmol. 2000 May-Jun;44(5):367-408.
- Raluca M, Stefanescu D, Craciun AM, et al. Old and new in exploring the anterior chamber angle. Rom J Ophthalmol. 2015 Oct-Dec;59(4):208-16.
- Foster PJ, Baasanhu J, Alsbirk PH, et al. Detection of gonioscopically occludable angles and primary angle closure glaucoma by estimation of limbal chamber depth in Asians: modified grading scheme. Br J Ophthalmol. 2000 Feb;84(2):186-92.
- Khoury JM, Brown RH, Donahue SP, et al. Comparison of 24-2 and 30-2 perimetry in glaucomatous and nonglaucomatous optic neuropathies. J Neuroophthalmol. 1999 Jun;19(2):100-8.
- Broadway DC. Visual field testing for glaucoma – a practical guide. Community Eye Health. 2012;25(79-80):66-70.
- Hodapp E, Parrish RK, Anderson DR. Clinical decisions in glaucoma. St. Louis: The CV Mosby Co.; 1993.
- Susanna R Jr, Vessani RM. Staging glaucoma patient: why and how? Open Ophthalmol J. 2009 Nov 20;3:59-64.
- Weinreb RN, Khaw PT. Primary open-angle glaucoma. Lancet. 2004 May 22;363(9422):1711-20.
- Poon LY, Waisberg Y, Wong B, et al. The ISNT Rule: How Often Does It Apply to Disc Photographs and Retinal Nerve Fiber Layer Measurements in the Normal Population? Am J Ophthalmol. 2017 Dec;184:19-27. DOI: 10.1016/j.ajo.2017.09.020
- Bengtsson B, Heijl A, Agis Investigators. Disc hemorrhages and treatment in the early manifest glaucoma trial. Ophthalmology. 2008 Nov;115(11):2044-8. DOI: 10.1016/j.ophtha.2008.06.015
- Jonas JB. Clinical implications of peripapillary atrophy in glaucoma. Curr Opin Ophthalmol. 2005 Apr;16(2):84-8.
- Manassakorn A, Nouri-Mahdavi K, Caprioli J. Comparison of retinal nerve fiber layer thickness and optic disk algorithms with optical coherence tomography to detect glaucoma. Am J Ophthalmol. 2006 Jan;141(1):105-15.
- Sehi M, Golding MP, Singh K, et al. Diagnostic Ability of Fourier-Domain vs Time-Domain Optical Coherence Tomography for Glaucoma Detection. Am J Ophthalmol. 2009 Oct;148(4):595-603. DOI: 10.1016/j.ajo.2009.05.021
- Schuman JS, Pedut-Kloizman T, Hertzmark E, et al. Reproducibility of nerve fiber layer thickness measurements using optical coherence tomography. Ophthalmology. 1996 Nov;103(11):1889-98.
- Nickells RW. From ocular hypertension to ganglion cell death: a theoretical sequence of events leading to glaucoma. Can J Ophthalmol. 2007 Apr;42(2):278-87.
- The AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol. 2000 Oct;130(4):429-40.
- Kass MA, Heuer Jr DK, Higginbotham EG, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002 Jun;120(6):701-13; discussion 829-30.
- Leske MC, Heijl A, Hussein M, et al. Factors for glaucoma progression and the effect of treatment: the early manifest glaucoma trial. Arch Ophthalmol. 2003 Jan;121(1):48-56. DOI: 10.1001/archopht.121.1.48
- Vetrugno M, Trabucco T, Sisti E, et al. Primary open angle glaucoma: an overview on medical therapy. Prog Brain Res. 2008;173:181-93.
- Lusthaus J, Goldberg I. Current management of glaucoma. Med J Aust. 2019 Feb;210(4):180-7.
- Konstas AG, Nakos E, Karabatsas CH, et al. 24-hour intraocular pressure control with maximum medical therapy compared with surgery in patients with advanced open-angle glaucoma. Ophthalmology. 2006 May;113(5):761-5.e1.
- Alexander CL, Miller SJ, Abel SR. Prostaglandin analog treatment of glaucoma and ocular hypertension. Ann Pharmacother. 2002 Mar;36(3):504-11.
- Drance SM, Crichton A, Mills RP. Comparison of the effect of latanoprost 0.005% and timolol 0.5% on the calculated ocular perfusion pressure in patients with normal-tension glaucoma. Am J Ophthalmol. 1998 May;125(5):585-92.
- Camras CB, The United States Latanoprost Study Group. Comparison of latanoprost and timolol in patients with ocular hypertension and glaucoma: a six-month masked, multicenter trial in the United States. Ophthalmology. 1996 Jan;103(1):138-47.
- Johnstone MA. Hypertrichosis and increased pigmentation of eyelashes and adjacent hair in the region of the ipsilateral eyelids of patients treated with unilateral topical latanoprost. Am J Ophthalmol. 1997 Oct;124(4):544-7.
- Sakata R, Yagi S, Hirasawa K, et al. Incidence of deepening of the upper eyelid sulcus in prostaglandin-associated periorbitopathy with a latanoprost ophthalmic solution. Eye (Lond). 2014 Dec;28(12):1446-51. DOI: 10.1038/eye.2014.225
- Halpern DL, Pasquale LR. Cystoid macular edema in aphakia and pseudophakia after use of prostaglandin analogs. Semin Ophthalmol. 2002 Sep-Dec;17(3-4):181-6.
- Kaufman PL. Latanoprostene bunod ophthalmic solution 0.024% for IOP lowering in glaucoma and ocular hypertension. Expert Opin Pharmacother. 2017 Apr;18(4):433-44. DOI: 10.1080/14656566.2017.1292080
- Brooks AM, Gillies WE. Ocular beta-blockers in glaucoma management. Clinical pharmacological aspects. Drugs Aging. 1992 May-Jun;2(3):208-21.
- Everitt DE, Avorn J. Systemic effects of medications used to treat glaucoma. Ann Intern Med. 1990 Jan 15;112(2):120-5.
- Hayreh SS, Podhajsky P, Zimmerman MB. Beta-blocker eyedrops and nocturnal arterial hypotension. Am J Ophthalmol. 1999 Sep;128(3):301-9.
- Lin JC. The use of ocular hypotensive drugs for glaucoma treatment: changing trend in Taiwan from 1997 to 2007. J Glaucoma. 2015 May;24(5):364-71. DOI: 10.1097/IJG.0b013e31828f7584
- Kaiser HJ, Flammer J, Wiernsperger N, et al. Blood-flow velocities of the extraocular vessels in patients with high-tension and normal-tension primary open-angle glaucoma. Am J Ophthalmol. 1997 Mar;123(3):320-7.
- Van Buskirk EM, Dickerson JE, Morrisey SP. Betaxolol in patients with glaucoma and asthma. Am J Ophthalmol. 1986 May 15;101(5):531-4.
- Goldberg I, Adena MA. Co-prescribing of topical and systemic beta-blockers in patients with glaucoma: a quality use of medicine issue in Australian practice. Clin Experiment Ophthalmol. 2007 Nov;35(8):700-5. DOI: 10.1111/j.1442-9071.2007.01605.x
- Toris CB, Gleason WL, Camras CB, et al. Effects of brimonidine on aqueous humor dynamics in human eyes. Arch Ophthalmol. 1995 Dec;113(12):1514-7.
- Nagasubramanian S, Joseph JP, Vijaya L, et al. Comparison of apraclonidine and timolol in chronic open-angle glaucoma. A three-month study. Ophthalmology. 1993 Sep;100(9):1318-23.
- Arthur S, Cantor LB. Update on the role of alpha-agonists in glaucoma management. Exp Eye Res. 2011 Sep;93(3):271-83. DOI: 10.1016/j.exer.2011.02.016
- Mundorf T, Noecker R, Peace J, et al. A 3-month comparison of efficacy and safety of brimonidine-purite 0.15% and brimonidine 0.2% in patients with glaucoma or ocular hypertension. J Ocul Pharmacol Ther. 2003 Feb;19(1):37-44.
- Burke J, Schwartz M. Preclinical evaluation of brimonidine. Surv Ophthalmol. 1996 Aug;41 Suppl 1:S9-18.
- Lachkar Y, Migdal C, Dhanjil S. Effect of brimonidine tartrate on ocular hemodynamic measurements. Arch Ophthalmol. 1998 Dec;116(12):1591-4.
- Ahmed FA, Aglan NA, Fathy M. Neuroprotective effect of alpha(2) agonist (brimonidine) on adult rat retinal ganglion cells after increased intraocular pressure. Brain Res. 2001 Sep 14;913(2):133-9.
- Stewart WC, Garrison E, Usner N, et al. Therapeutic success of latanoprost 0.005% compared to brimonidine 0.2% in patients with open-angle glaucoma or ocular hypertension. J Ocul Pharmacol Ther. 2000 Dec;16(6):557-64.
- Kanski JJ. Carbonic anhydrase inhibitors and osmotic agents in glaucoma. Carbonic anhydrase inhibitors. Br J Ophthalmol. 1968 Aug;52(8):642-3.
- Hu CY, Huang YC, Liu CC, et al. Acetazolamide-related life-threatening hypophosphatemia in a glaucoma patient. J Glaucoma. 2015 Apr-May;24(4):e31-3. DOI: 10.1097/IJG.0b013e3182885994
- Mincione F, Scozzafava A, Supuran CT. The development of topically acting carbonic anhydrase inhibitors as anti-glaucoma agents. Curr Top Med Chem. 2007;7(9):849-54.
- Rao PV, Pattabiraman PP, Kopczynski C. Role of the Rho GTPase/Rho kinase signaling pathway in pathogenesis and treatment of glaucoma: Bench to bedside research. Exp Eye Res. 2017 May;158:23-32. DOI: 10.1016/j.exer.2016.09.018
- Lin CW, Liu J, Dinh T, et al. Discovery and Preclinical Development of Netarsudil, a Novel Ocular Hypotensive Agent for the Treatment of Glaucoma. J Ocul Pharmacol Ther. 2018 Jan-Feb;34(1-2):40-51. DOI: 10.1089/jop.2017.0090
- Moshirfar M, T. P. A., Ronquillo YC, et al. Use of Rho kinase Inhibitors in Ophthalmology: A Review of the Literature. Med Hypothesis Discov Innov Ophthalmol. 2018 Fall;7(3):101-11.
- Kaufman PL, Barany EH. Residual pilocarpine effects on outflow facility after ciliary muscle disinsertion in the synomolgus monkey. Invest Ophthalmol. 1976 Jul;15(7):558-61.
- Deokule S, Sadiq S, Shah S. Chronic open angle glaucoma: patient awareness of the nature of the disease, topical medication, compliance and the prevalence of systemic symptoms. Ophthalmic Physiol Opt. 2004 Jan;24(1):9-15.1
- Zimmerman TJ, Zalta AH. Facilitating patient compliance in glaucoma therapy. Surv Ophthalmol. 1983 Sep-Oct;28 Suppl:252-8.
- Muir KW, Lee PP. Glaucoma medication adherence: room for improvement in both performance and measurement. Arch Ophthalmol. 2011 Feb;129(2):243-5. DOI: 10.1001/archophthalmol.2011.1
- Olthoff CM, Schouten JS, van de Borne BW, et al. Noncompliance with ocular hypotensive treatment in patients with glaucoma or ocular hypertension an evidence-based review. Ophthalmology. 2005 Jun;112(6):953-61.
- Schwartz GF, Quigley HA. Adherence and persistence with glaucoma therapy. Surv Ophthalmol. 2008 Jul;53 Suppl1:S57-68. DOI: 10.1016/j.survophthal.2008.02.002
- Newman-Casey PA, Blachley TS, Pasquale LR, et al. The Most Common Barriers to Glaucoma Medication Adherence: A Cross-Sectional Survey. Ophthalmology. 2015 Jul;122(7):1308-16. DOI: 10.1016/j.ophtha.2015.03.023
- Davidson SI, Akingbehin T. Compliance in ophthalmology. Trans Ophthalmol Soc U K. 1980;100(Pt 2):286-90.
- Kinast RM, Mansberger SL. Glaucoma Adherence-From Theriac to the Future. Am J Ophthalmol. 2018 Jul;191:xiii-xv. DOI: 10.1016/j.ajo.2018.05.006
- Heijl A, Leske MC, Bengtsson B, et al. Reduction of intraocular pressure and glaucoma progression: results from the Early Manifest Glaucoma Trial. Arch Ophthalmol. 2002 Oct;120(10):1268-79. DOI: 10.1001/archopht.120.10.1268
- Musch DC, Gillespie BW, Niziol LM, et al. Visual field progression in the Collaborative Initial Glaucoma Treatment Study the impact of treatment and other baseline factors. Ophthalmology. 2009 Feb;116(2):200-7. DOI: 10.1016/j.ophtha.2008.09.025
- Collaborative Normal-Tension Glaucoma Study Group. The effectiveness of intraocular pressure reduction in the treatment of normal-tension glaucoma. Am J Ophthalmol. 1998 Oct;126(4):498-505.
- Parrish RK 2nd, Bradley D, Gordon MO, et al. Five-year follow-up optic disc findings of the Collaborative Initial Glaucoma Treatment Study. Am J Ophthalmol. 2009 Apr;147(4):717-24.e1. DOI: 10.1016/j.ajo.2008.11.006
- Lichter PR, Musch DC, Gillespie BW, et al. Interim clinical outcomes in the Collaborative Initial Glaucoma Treatment Study comparing initial treatment randomized to medications or surgery. Ophthalmology. 2001 Nov;108(11):1943-53.
- Rolim de Moura CR, Paranhos Jr A, Wormald R. Laser trabeculoplasty for open angle glaucoma. Cochrane Database Syst Rev. 2007 Oct 17; (4):CD003919. DOI: 10.1002/14651858.CD003919.pub2
- Garg A, Gazzard G. Selective laser trabeculoplasty: past, present, and future. Eye (Lond). 2018 May;32(5):863-76. DOI: 10.1038/eye.2017.294
- Song J. Complications of selective laser trabeculoplasty: a review. Clin Ophthalmol. 2016 Jan 11;10:137-43. DOI: 10.2147/OPTH.S95911
- Gazzard G, Xia T, Agis Investigators. Laser in Glaucoma and Ocular Hypertension (LiGHT) Trial: Six-Year Results of Primary Selective Laser Trabeculoplasty versus Eye Drops for the Treatment of Glaucoma and Ocular Hypertension. Ophthalmology. 2023 Feb;130(2):139-51.
- Gedde SJ, Herndon LW, Brandt JD, et al. The Tube Versus Trabeculectomy Study: interpretation of results and application to clinical practice. Curr Opin Ophthalmol. 2012 Mar;23(2):118-26. DOI: 10.1097/ICU.0b013e3283503d6d
- Kirwan JF, Lockwood G, Cousens S, et al. Trabeculectomy in the 21st century: a multicenter analysis. Ophthalmology. 2013 Dec;120(12):2532-9. DOI: 10.1016/j.ophtha.2013.04.026
- Gedde SJ, Herndon LW, Brandt JD, et al. Treatment outcomes in the Tube Versus Trabeculectomy (TVT) study after five years of follow-up. Am J Ophthalmol. 2012 May;153(5):789-803.e2. DOI: 10.1016/j.ajo.2011.10.038
- Francis BA, Singh K, Lin S, et al. Novel glaucoma procedures: a report by the American Academy of Ophthalmology. Ophthalmology. 2011 Jul;118(7):1466-80. DOI: 10.1016/j.ophtha.2011.02.043
- Lavia C, Dall’orto L, Maule M, et al. Minimally-invasive glaucoma surgeries (MIGS) for open angle glaucoma: A systematic review and meta-analysis. PLoS One. 2017 Aug 10;12(8):e0183142. DOI: 10.1371/journal.pone.0183142
- American Academy of Family Practice. Consultations, Referrals, and Transfers of Care [Internet]. Leawood, KS: American Academy of Family Practice; c2017 [cited 2019 Dec 26]. Available from: https://www.aafp.org/about/policies/all/consultations-transfers.html.
- Francois J. Tool to assess the quality of consultation and referral request letters in family medicine. Can Fam Physician. 2011 May;57(5):574-5.
- Thorsen O, Hartveit M, Baerheim A. The consultants’ role in the referring process with general practitioners: partners or adjudicators? a qualitative study. BMC Fam Pract. 2013 Oct 22;14:153.
- Grimshaw JM, Bayley L, Brebner HE, et al. Interventions to improve outpatient referrals from primary care to secondary care. Cochrane Database Syst Rev. 2005 Jul 20;(3):CD005471. DOI: 10.1002/14651858.CD005471
- Marshall S, Harrison J, Flanagan B. The teaching of a structured tool improves the clarity and content of interprofessional clinical communication. Qual Saf Health Care. 2009 Apr;18(2):137-40. DOI: 10.1136/qshc.2008.028984



