PEER REVIEWED
Superior Segmental Optic Nerve Hypoplasia
Ryan Terry, OD, ABCMO, Tyler Wyatt, OD, ABCMO
Abstract
Superior segmental optic nerve hypoplasia (SSONH) is a congenital disorder characterized by a reduction in retinal ganglion cells in the superior aspect of the optic disc which results in retinal nerve fiber layer thinning with an associated visual field defect. These findings are often misdiagnosed as glaucoma as ancillary findings are similar. This teaching case report discusses the features and clinical presentation of SSONH in patients that were being followed for glaucoma and the associated biases that influenced management.
Key Words: hypoplasia, topless disc syndrome, segmental
Background
Superior Segmental Optic Nerve Hypoplasia (SSONH), also known as topless disc syndrome, is a nonprogressive congenital anomaly of the optic nerve head.1 The superior nerve fibers are underdeveloped which leads to superior nerve pallor and possible superior displacement of the central retinal artery, which distinguishes SSONH from other optic nerve hypoplasias. These changes lead to a thin superior retinal nerve fiber layer with a corresponding inferior visual field defect which is often misdiagnosed as glaucoma.2
These cases highlight two patients being managed for glaucoma, who were born with undiagnosed SSONH. The purpose of presenting these cases is to emphasize the need for careful, critical evaluation of the optic nerve in ophthalmic examinations.
These cases are intended for students and clinicians alike to increase awareness of SSONH and the biases that can influence our medical decision-making. They emphasize the importance of critically thinking through diagnosed cases and looking for inconsistencies in the clinical picture through careful examination.
Case 1 Description
A 63-year-old male presented to the clinic for a dilated eye exam. He was new to the clinic and had no visual complaints. He self-reported temporal visual field loss in his left eye following head trauma years ago. Recent A1C was 9.2% and family history was positive for maternal gestational diabetes.
On examination, pupils, extraocular muscle movement and anterior segment were all normal except for mild cataracts. His best corrected visual acuity was 20/20 in both eyes. Confrontation visual field testing revealed a temporal restriction in the left eye, and intraocular pressure was asymmetric between the right and left eye at 16 and 18 mm Hg, respectively.
The peripheral retina was normal in the right eye, but two blot hemorrhages were discovered superior nasal in the left eye. See Image 1 for fundus photos and Image 2 for optical coherence tomography (OCT) of the retinal nerve fiber layer (RNFL).
He had a history of IOP lowering treatment but hadn’t used any drops for 3 years. See Image 3 for a 24-2 SITA standard Humphrey visual field (HVF) testing.
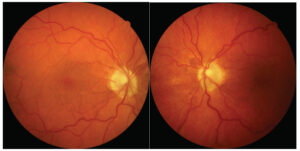
Image 1. Fundus photos of the right (a) and left (b) eye of Patient 1. Click to enlarge
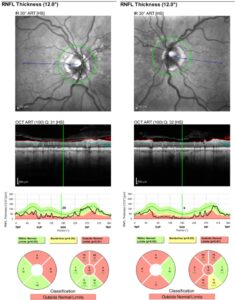
Image 2. RNFL OCT of the right (a) and left (b) eye of Patient 1. Click to enlarge

Imaage 3. HVF testing 24-2 SITA Standard of the right and left eye for Patient 1. Click to enlarge
Case 2 Description
A 50-year-old white male presented to the clinic for a primary open angle glaucoma follow-up. He had been followed for glaucoma for 4 years at this clinic, but he reported being managed since he was 32. He has been taking latanoprost 0.005% 1 drop in both eyes every evening and dorzolamide 2% 1 drop in both eyes 2 times a day with good compliance. His medical history includes type 2 diabetes mellitus, hyperlipidemia, obstructive sleep apnea, and a history of prematurity. He was taking simvastatin and glipizide in addition to his glaucoma medications. His family history is positive for diabetes (mother).
On examination, pupils, extraocular muscle movement and anterior segment were all normal except for mild cataracts. He had a best corrected visual acuity of 20/20 in each eye. Confrontation visual field testing revealed a mild inferior right defect in the right eye and an inferior right defect in the left eye. IOP was measured at 13 in both eyes (treated with latanoprost and dorzolamide). His IOP history shows that his untreated IOP was 26 in the right eye and 22 in the left. Average IOP with the treatment of latanoprost is 17 in the right eye and 15 in the left. Average IOP with the use of latanoprost and the addition of dorzolamide is 14 in the right eye and 14 in the left.
Historically, the retina has been unremarkable with no signs of diabetic retinopathy in either eye. No disc hemes were noted at this exam, and see Image 4 for past fundus photos. See Image 5 for recent OCT testing of the RNFL with baseline data from three years prior. See Image 6 for a summary of three years of HVF testing.
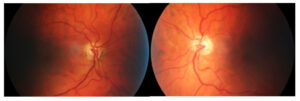
Image 4. Fundus photos of the right (a) and left (b) eye of Patient 2. Click to enlarge
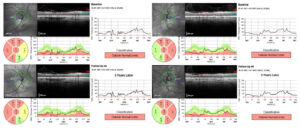
Image 5. Baseline RNFL OCT and a 3-year follow-up of the right (a) and left (b) eye of Patient 2. Click to enlarge
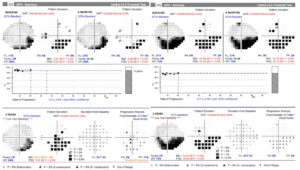
Image 6. HVF Testing 24-2 SITA Standard with baseline data and 3-year follow-up, including a GPA Summary for the right and left eye of Patient 2. Click to enlarge
Education Guidelines
Learning objectives
At the conclusion of the case, participants should be able to:
- Identify the typical characteristics associated with SSONH
- Describe the differences in optic nerve changes associated with SSONH vs glaucoma
- Identify how to utilize ancillary testing to aid in the diagnosis of SSONH
- Describe the similarities and differences in ancillary test findings between SSONH and glaucoma
- Understand bias in medical decision making
Key concepts
- Recognize the clinical presentation of SSONH
- Recognize the importance of thorough optic nerve evaluation in distinguishing various optic neuropathies
- Recognize the typical ancillary test findings of SSONH
- Recognize the differences and similarities between SSONH and glaucoma
- Understand optic nerve appearance and the need to investigate further if the current clinical picture does not match examination findings
- Identify and describe the bias of diagnosis momentum, premature closure, and search satisfying
Discussion questions
- What are the characteristics of a typical visual field in SSONH?
- Explain the differences and commonalities between visual field defects in glaucoma and SSONH.
- What features of the optic nerve lead to the diagnosis of SSONH vs glaucoma?
- What other clinical data (i.e., case history, ancillary tests, clinical findings, etc.) can aid in the diagnosis of SSONH?
- What is the long-term prognosis for SSONH, and how does that differ from glaucoma?
- Does SSONH effect visual acuity?
- What differential diagnosis should be considered in suspected cases of SSONH?
- How did diagnosis momentum, premature closure, and search satisfying play a role in the management of Cases 1 and 2?
- In Cases 1 and 2, what one key finding or observation best supports the diagnosis of SSONH vs glaucoma?
Teaching instructions
Reviewing cases in a classroom setting changes the attentiveness to detail and broadens the differentials of the participants. When a particular case is highlighted in lecture format, more rare findings are considered that may not have been when just regularly seen in clinic. To help avoid biasing the participant with buzz words or indicating a significant finding, descriptions of the figures and photographs will be omitted in the labels of the images, but they will be included in the discussion section. These patients presented to the clinic as simply glaucoma management cases, portraying them as such will help reduce any bias and offer the best learning opportunity for the participants.
If setting permits, separate the students into two groups with each reviewing a case. The first case should be presented as a glaucoma suspect and the group is to determine whether the patient has glaucoma. Encourage the participants to develop a treatment or monitoring plan.
The second case should be presented as a primary open-angle glaucoma patient, and the group is to determine the best management plan such as monitor with time frame, change medication, establish a new diagnosis, etc.
Once the groups have finalized a diagnosis and plan for their respective cases, highlight how the optic nerve appearance has features not typically found in glaucoma (superior displaced optic nerve cup and pallor). Then using the discussion questions above as a guide, lead a discussion on the defining features of SSONH and how it compares to glaucoma.
Alternatively, Case 1 could be presented and discussed as a group. The second case could then be reserved for an assessment where the students could apply what they have learned in the first case in discussing the clinical features of the second.
When the students feel comfortable identifying SSONH, highlight the possible medical decision-making biases found in the cases and observe whether the groups fell into the same trap. Encourage the participants to evaluate bias in their own medical decisions and ways that they can self-guard against them.
Learning Assessment
- Meaningful case discussion that emphasizes learning objectives
- OPTIONAL: Case 2 could be reserved as an assessment after the discussion of Case 1. (See teaching instructions)
- Internalization of information and critical thinking skills can be assessed by having students present differential diagnosis and identifying what key clinical features or ancillary tests rule out those diagnosis.
- Meaningful discussion of the applicable bias and how they relate to clinical care.
Discussion
Superior Segmental Optic Nerve Hypoplasia
SSONH is a congenital disorder characterized by a reduction in retinal ganglion cells in the superior aspect of the optic disc. Due to the anatomical loss, a non-progressive inferior visual field defect is often present, however variable. The etiology of optic nerve hypoplasia is thought to be related to maternal factors, prenatal exposures, and genetic factors.3 In some studies, maternal diabetes has been identified as a risk factor for developing SSONH, however, a causal relationship has not been established.4 (Anecdotally, both Cases 1 and 2 have a history of maternal diabetes.)
SSONH was first described by Peterson and Walton in 1977 as a form optic nerve hypoplasia occurring in children of diabetic mothers with normal visual acuity with visual field defects.5 In 1989, Kim et al. furthered described SSONH and identified four distinct clinical features of the optic discs: entrance of the central artery was relatively displaced superiorly, superior segmental pallor of the disc, adjacent peripapillary scleral “halo”, and thinning of the peripapillary retinal nerve fiber layer.6 As studies have progressed, there is a variable nature to the presentation of these characteristics which will be discussed later.1,2,4,7,8 The prevalence of SSONH has been studied by various groups and has been estimated to be between 0.08% and 0.24% of people.9 This prevalence may be underestimated due to the misidentification of this disease with other optic atrophies such as glaucoma.
SSONH is usually found incidentally on ophthalmic evaluation as it tends to have little impact on daily living.2 Visual acuity is often unaffected and visual field defects are often in the periphery.2 The ophthalmic examination is overall normal including best corrected visual acuities, extraocular motilities, and intraocular pressures. A relative afferent pupillary defect may be present in asymmetric presentations of SSONH as laterality has been found to be variable in recent studies indicating that both bilateral and unilateral cases are possible.4
Visual Field Defects
The prevalence of visual field defects in SSONH is variable. Most studies have found that greater than one half of patients or more with SSONH had a corresponding inferior visual field defect in at least one eye.2,4 This means that many patients may not have detectable visual field loss. The inferior visual field defects can be sectoral or altitudinal and, in many instances, connect to the blind spot, which correlates to the missing superior retinal nerve fibers.10 In contrast, visual field defects from glaucoma often occur in the nasal step region, and sometimes in the paracentral region.9
The presented cases both have significant inferior visual field defects consistent with SSONH.
Case 1: Both the right and left eyes have varying degrees of inferior visual field defects that extend from the blind spot and do not include the nasal step region which is more consistent with SSONH than glaucoma. (Image 3)
Case 2: The right eye shows an inferior defect that extends from the optic nerve sparing the nasal region, which can help differentiate this defect from glaucoma. However, the left eye has a more severe defect that extends from the optic nerve and affects the nasal step region, which shows the variable pattern of SSONH and the difficultly of using a field alone to distinguish between glaucoma and SSONH. (Image 6)
Disc Appearance
The “topless disc” appearance in SSONH is a hallmark clinical feature that distinguishes it from other optic atrophies including a superior displaced central artery, pallor of the superior disc, and superior peripapillary halo.9 In reviewing the published literature, Wu et al. noted that recent studies have shown a variability in the presentation of these clinical features.9 One particular study noted that only 65% had the superior central artery insertion, 60% had the superior peripapillary scleral halo, and 30% had the superior disc pallor.11 Given the variable presentation of disc findings, careful observation of the disc, in addition to other clinical features, can be helpful in differentiating SSONH from normal tension glaucoma. In contrast to SSONH, glaucoma is typically associated with thinning of the inferior temporal and superior temporal rim.12
Case 1: Both optic nerves have a symmetrical appearance with the central artery being displaced superiorly, a partial peripapillary halo with no distinct pallor. (Image 1)
Case 2: The right optic nerve has a superiorly displaced central artery, no rim pallor superiorly, and a peripapillary halo superior. The left eye does not have a superior displaced central artery, but it does have superior pallor with minimal peripapillary halo. This patient shows the variability of SSONH even within fellow eyes of the same patient. (Image 4)
Nerve Fiber Layer Thinning
Given the characteristics of the superior disc in SSONH, it is understandable that multiple studies have confirmed thinning of the superior retinal nerve fiber layer (RNFL) as measured by optical coherence tomography (OCT).9 Interestingly, further studies have found that the inferotemporal sector is spared in patients with SSONH while the remaining RNFL is thinned when compared to the normal population.9,13 Recognizing this characteristic is another helpful tool in differentiating SSONH from glaucoma as glaucoma frequently impacts the inferotemporal sector.
Case 1: The RNFL is significantly reduced superiorly in both eyes. The thinning also effects other sectors sparing the inferotemporal and temporal region, which is more characteristic of SSONH as opposed to glaucoma. (Image 2)
Case 2: The RNFL is significantly reduced superiorly in both eyes. The thinning also affects all sectors except for the inferonasal sector. Contrary to some studies, thinning is also present in the inferotemporal region. This raises suspicion for concomitant glaucoma, especially given their ocular hypertension. (Image 4)
In an effort to help further differentiate SSONH from NTG, Lee et al. examined how these two differed on Ocular Coherence Tomography Angiography (OCTA). They reported that in patients with SSONH, the peripapillary Vessel density (VD) was lower in the superionasal/nasal segments and higher in the superiotemporal segment when compared to NTG patients. Although limited, this study indicates that OCTA may provide another tool in distinguishing between SSOH and NTG specifically.14
Bias In Clinical Care
Just as classroom presentations have bias, the cases presented illustrate several biases, but for the sake of discussion, we will highlight premature closure, diagnosis momentum, and search satisfying.
Premature closure is one of the most common errors in medical decision-making. It manifests when a clinician fails to consider reasonable alternatives after an initial diagnosis is made.15 In the first case, atypical nerves were noted leading to ancillary testing being performed. Once the data showed thinning on OCT with corresponding visual field defects, a premature conclusion of glaucoma was made rather than closely assessing the nerve and all differentials.
The second case illustrates the bias of diagnosis momentum, which is similar to anchoring bias. This refers to the tendency that after a diagnosis is made, the more “sticky” that diagnosis becomes over time.16 It is difficult to then analyze the data skeptically, especially when it has been carried over through multiple providers.17 In the case above, the diagnosis of glaucoma was carried over for years and all the data was interpreted through the lens of glaucoma. One can see how momentum likely influenced the providers to never question the initial diagnosis.
It is important, however, to not fall into another bias: search satisfying, which is when one calls off the search once a disease or problem is found. With this thinking, comorbidities may be missed.16 For example, we may overlook ocular ischemic syndrome in a diabetic patient, corneal herpes in a chronic dry eye patient, and glaucoma in a previous optic nerve ischemic event. In the cases discussed, it is possible for the patient to have SSONH and glaucoma, especially in Case 2, where ocular hypertension and inferotemporal RNFL thinning is present.
Conclusion
The clinical presentation of SSONH can lead to a misdiagnosis of glaucoma which can significantly alter a patient’s treatment and management. Careful observation of all clinical features is needed to correctly identify optic nerve abnormalities. SSONH is just one of many differentials when an anomalous optic nerve does not fit the typical glaucoma presentation.
These teaching cases offer a great learning opportunity to assess how biases can creep into our medical decision-making. Simply being aware of the bias can help safeguard against any negative effects, especially in complex conditions like glaucoma.
References
-
- Takagi M, Abe H, Hatase T, Yaoeda K, Miki A, Shirakashi M. Superior segmental optic nerve hypoplasia in youth. Jpn J Ophthalmol. 2008 Nov-Dec;52(6):468–474. doi: 10.1007/s10384-008-0588-9
- Yamamoto T. Superior segmental optic hypoplasia as a differential diagnosis of glaucoma. Taiwan J Ophthalmol. 2019 Apr-Jun;9(2):63-66. doi: 10.4103/tjo.tjo_28_19
- Garcia-Filion P, Borchert M. Optic nerve hypoplasia syndrome: a review of the epidemiology and clinical associations. Curr Treat Options Neurol. 2013 Feb;15(1):78–89. doi: 10.1007/s11940-012-0209-2
- Yagasaki A, Sawada A, Manabe Y, Yamamoto T. Clinical features of superior segmental optic hypoplasia: hospital-based study. Jpn J Ophthalmol. 2019 Jan;63(1):34–39. doi: 10.1007/s10384-018-0634-1
- Petersen RA, Walton DS. Optic nerve hypoplasia with good visual acuity and visual field defects: a study of children of diabetic mothers. Arch Ophthalmol. 1977 Feb;95(2):254–258. doi: 10.1001/archopht.1977.04450020055011
- Kim RY, Hoyt WF, Lessell S, Narahara MH. Superior segmental optic hypoplasia. A sign of maternal diabetes. Arch Ophthalmol. 1989 Sep;107(9):1312–1315. doi: 10.1001/archopht.1989.01070020382040
- Han SB, Park, KH, Kim DM, Kim TW. Prevalence of superior segmental optic nerve hypoplasia in Korea. Jpn J Ophthalmol. 2009;53:225–228. doi: 10.1007/s10384-009-0663-x
- Seo S, Lee CE, Kim DW, Kim YK, Jeoung JW, Kim CY, Kang SW, et al. Prevalence and risk factors of superior segmental optic hypoplasia in a Korean population: the Korea National Health and Nutrition Examination Survey. BMC Ophthalmol. 2014 Dec 15;14:157. doi: 10.1186/1471-2415-14-157
- Wu JH, Lin CW, Liu CH, Weinreb RN, Welsbie DS. Superior segmental optic nerve hypoplasia: A review. Surv Ophthalmol. 2022 Sep-Oct;67(5):1467-1475. doi: 10.1016/j.survophthal.2022.02.008
- Yamamoto T, Sato M, Iwase A. Superior segmental optic hypoplasia found in Tajimi Eye Health Care Project participants. Jpn J Ophthalmol. 2004 Nov-Dec;48(6):578–583. doi: 10.1007/s10384-004-0120-9
- Hayashi K, Tomidokoro A, Konno S, Mayama C, Aihara M, Araie M. Evaluation of optic nerve head configurations of superior segmental optic hypoplasia by spectral-domain optical coherence tomography. Br J Ophthalmol. 2010 Jun;94(6):768-772. doi: 10.1136/bjo.2009.168690
- Gandhi M, Dubey S. Evaluation of the Optic Nerve Head in Glaucoma. J Curr Glaucoma Pract. 2013 Sep-Dec;7(3):106-14. Epub 2013 Sep 6. doi: 10.5005/jp-journals-10008-1146
- Han JC, Choi DY, Kee C. The Different Characteristics of Cirrus Optical Coherence Tomography between Superior Segmental Optic Hypoplasia and Normal Tension Glaucoma with Superior Retinal Nerve Fiber Defect. J Ophthalmol. 2015;2015:641204 doi: 10.1155/2015/641204
- Lee SY, In JH, Kim CH, Hong YJ. Usefulness of Optical Coherence Tomography Angiography in the Differential Diagnosis Between Superior Segmental Optic Hypoplasia and Normal-tension Glaucoma. J Glaucoma. 2020 Aug;29(8):718-722. doi: 10.1097/IJG.0000000000001530
- Coen M, Sader J, Junod-Perron N, Audétat MC, Nendaz M. Clinical reasoning in dire times. Analysis of cognitive biases in clinical cases during the COVID-19 pandemic. Intern Emerg Med. 2022 Jun;17(4):979-988. Epub 2022 Jan 8. doi: 10.1007/s11739-021-02884-9
- Doherty TS, Carroll AE. Believing in Overcoming Cognitive Biases. AMA J Ethics. 2020 Sep 1;22(9):E773-778. doi: 10.1001/amajethics.2020.773
- Croskerry P. The importance of cognitive errors in diagnosis and strategies to minimize them. Acad Med. 2003 Aug;78(8):775-780. doi: 10.1097/00001888-200308000-00003



