PEER REVIEWED
Elevated Intraocular Pressure After Cataract Surgery
Mallory McLaughlin, OD, FAAO
Abstract
Elevated intraocular pressure (IOP) is a potential complication of cataract surgery. This report reviews a patient with post-operative elevated IOP and highlights the importance of understanding its pathophysiology, risk factors and applying evidence-based management strategies. This report aims to enhance the clinical approach to managing elevated IOP in the postoperative setting.
Keywords
Background
Elevated Intraocular Pressure (IOP) typically peaks 3-7 hours after surgery.1,2 When patients are evaluated within 3-7 hours postoperatively, elevated IOP occurs in approximately 18% of those without glaucoma and 47% of those with glaucoma.2 When evaluated 1 day postoperatively, elevated IOP is seen in 2-9% of patients.1 While elevated postoperative IOP is usually transient, chronic or exceptionally high IOP can result in vision loss. This case is intended for third- and fourth-year optometry students and new practitioners with a basic understanding of how cataract surgery is performed and postoperative care. Doctors involved in co-managing cataract surgery must be equipped to evaluate and manage this complication with confidence.
Case Description
Initial Encounter
A 77-year-old Asian male presented for a 1-day postoperative (POD1) examination after uncomplicated phacoemulsification in the left eye. He had a past ocular history of degenerative myopia with posterior staphyloma in both eyes. His axial lengths were 27.52 mm and 28.05 mm in the right and left eyes, respectively. He previously had cataract surgery in his right eye, complicated by postoperative ocular hypertension at POD1, managed successfully with in-office anterior chamber decompression, sometimes called “wound burping”. Phacoemulsification under intravenous anesthesia was performed in the left eye 1 day prior. The patient received an intracameral injection of moxifloxacin, and the corneal wound was closed with stromal hydration. The patient reported his vision had significantly improved in the left eye since the surgery, and he denied any pain, redness or light sensitivity. He reported starting prednisolone acetate 1% 4 times daily and ketorolac 0.5% 4 times daily in the left eye immediately following the surgery and he reported adherence to the prescribed dosing schedule.
During the present POD1 examination, his entering uncorrected distance visual acuity was 20/20-1 in the left eye. Both pupils were round and reactive to light without afferent pupillary defect. Extraocular motility and confrontation fields were full in both eyes. Slit lamp biomicroscopy was performed on the left eye. The conjunctiva was white and quiet. The cornea had two wounds, superior temporal and superior nasal, with 1+ microcystic corneal edema and 1+ superficial punctate keratitis adjacent to each wound. There was no corneal edema or corneal staining centrally. The Seidel test was negative for wound leak. There was trace viscoelastic material present on the corneal endothelium. The anterior chamber had 1+ white blood cells, trace pigment and trace flare. The intraocular lens (IOL) implant was clear and centered. IOP measured with Goldmann applanation tonometry was 42 mmHg in the left eye. The right eye was not tested. IOP in each eye historically ranged from 14-15 mmHg.
Anterior chamber decompression was performed in-office to lower the intraocular pressure in the left eye. One drop each of topical fluorescein sodium 0.25%/benoxinate sodium 0.4% ophthalmic solution (Altaire Pharmaceuticals, Aquebogue, NY, USA) and ofloxacin 0.3% ophthalmic solution (Bausch + Lomb, Bridgewater, NJ, USA) were instilled in the left eye. Using a cotton-tip applicator, pressure was applied to the bulbar conjunctiva immediately superior to the corneal wound (Figure 1). The cobalt blue slit lamp filter was used in conjunction with the sodium fluorescein to observe the flow of aqueous out of the corneal wound. Another drop of ofloxacin 0.3% was instilled in the left eye immediately following the procedure. Goldmann applanation tonometry was measured at 12 mmHg in the left eye. Twenty minutes later, IOP was measured again with Goldmann applanation at 32 mmHg.
Figure 1: A blunt instrument is used to put pressure on the bulbar conjunctiva adjacent to the paracentesis to release aqueous. This video was shared with permission of the author, Dr. Eric Harris.
The patient was prescribed timolol 0.5% twice daily in the left eye and was told to continue prednisolone acetate ophthalmic suspension 1% 4 times daily and ketorolac ophthalmic solution 0.5% 4 times daily in the left eye. He was advised to return in 1 week and to call the clinic immediately if redness, pain or a decrease in his vision occurred.
Follow-up #1
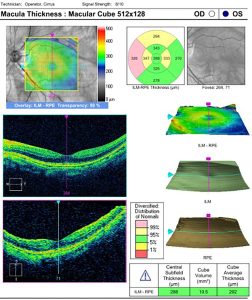
Figure 2: Cirrus OCT Macular Cube of the left eye, demonstrating no cystoid macular edema was present. Click to enlarge
At the 1-week postoperative (POW1) examination, the patient reported adherence to the prescribed eye drop regimen described above. He reported a constant “haze” in the vision of his left eye starting after the POD1 examination. He denied any pain or redness. His entering uncorrected distance visual acuity was 20/40 with pinhole testing to 20/25 in the left eye. The left pupil was round and reactive to light without afferent pupillary defect. Extraocular motility and confrontation fields were full in both eyes. Slit lamp biomicroscopy was performed in the left eye. The conjunctiva was white and quiet. The cornea had two wounds, superior temporal and superior nasal, with 1+ superficial punctate keratitis adjacent to each wound. There was 1+ diffuse corneal edema centrally. The Seidel test was negative for wound leak. There was trace viscoelastic material on the corneal endothelium. The anterior chamber had 1+ white blood cells, trace pigment and trace flare. The intraocular lens was clear and centered. IOP measured with Goldmann applanation tonometry was 15 mmHg in the left eye. Dilated fundus examination was unremarkable in the left eye except for posterior staphyloma surrounding the optic nerve but sparing the macula. Optical coherence tomography (Cirrus HD-OCT, Carl Zeiss Meditec, Dublin, CA, USA) scans of the macula confirmed there was no cystoid macular edema present in the left eye (Figure 2).
The patient was instructed to decrease the timolol 0.5% to once daily in the left eye and continue prednisolone acetate 1% 4 times daily and ketorolac 0.5% four times daily in the left eye. He was advised to return for the 1-month postoperative (POM1) examination and to call the clinic immediately if redness, pain or a decrease in his vision occurred.
Follow-up #2
At the POM1 examination, the patient reported confusion with dosage instructions, so was incorrectly dosing the prescribed eye drops. He was taking timolol 0.5%, prednisolone acetate 1% and ketorolac 0.5% all twice a day in the left eye. He reported the vision in his left eye had improved, but he was now experiencing light sensitivity. He denied pain and redness. His uncorrected visual acuity was now 20/25 but refracted to 20/20 with a small hyperopic prescription. His slit lamp examination was only notable for corneal scars superior temporal and inferior temporal. The corneal edema was resolved. The anterior chamber was quiet, and the intraocular lens was clear and centered. IOP was measured with Goldmann applanation tonometry at 13 mmHg in the left eye. Dilated fundus examination was again unremarkable in the left eye except for the posterior staphyloma.
The patient was instructed to stop the timolol 0.5% and ketorolac 0.5% and to reduce the prednisolone acetate 1% to once daily for 1 week in the left eye and then stop. He was advised to return in 6 months for a follow-up examination but did not return for that visit.
Educational Guidelines:
Key Concepts
- Pathophysiology of elevated IOP after cataract surgery.
- Identifying patient and procedural risk factors for elevated IOP after cataract surgery.
- Effective approaches for managing elevated IOP after cataract surgery.
Learning Objectives
- Recognize the causes and predisposing factors for elevated IOP after cataract surgery.
- Assess when elevated postoperative IOP requires treatment, and when it can be safely monitored without intervention.
- Prescribe appropriate treatments and establish follow-up procedures based on patient symptoms and clinical evaluation.
Discussion Questions
Review of prior knowledge of cataract surgery procedure and postoperative care:
- How is cataract surgery performed? Outline the key steps involved in the procedure.
- What findings are expected during the first postoperative day examination?
Differential diagnosis and risk factors:
- What is the pathophysiology of elevated postoperative IOP?
- What are the common risk factors associated with increased IOP after cataract surgery?
- For a patient with elevated postoperative IOP, which diagnostic tests are recommended to identify the underlying cause?
Management of the complication:
- What are the available treatment options for managing elevated postoperative IOP, and how should they be selected?
- When is it appropriate to monitor a patient with elevated postoperative IOP without initiating treatment?
- When would anterior chamber decompression be indicated, and what are the associated risks?
- What factors determine the timing of follow-up examinations for patients with elevated postoperative IOP?
Teaching Methodology
This learning assignment is intended for third- and fourth-year optometry students, optometry residents and new practitioners with a basic understanding of the cataract surgery procedure and postoperative care. The case discussion may be completed in a small group setting with an optometrist or ophthalmologist acting as the facilitator. The students should read the case and consider the discussion questions before the meeting. To start the discussion, the facilitator should read the abstract to set the tone for the discussion and then utilize the Socratic Method of teaching to review the discussion questions. In the Socratic Method, each student should be encouraged to share their ideas, and the facilitator should ask questions that facilitate logical thinking, challenge students’ thought processes and allow the students to come to their own conclusions.
Learning Assessment
- Change small details of the case and ask learners how the management should change. For example, if the postoperative IOP was slightly higher or lower, if the patient had other comorbidities like glaucoma or corneal endothelial disease, or if IOP did not respond to initial treatment.
- Have learners participate in a paired role-playing activity simulating a patient education encounter. The learner designated as the physician should explain the complications identified during the examination. They should provide clear, jargon-free education on anterior chamber decompression to obtain informed consent from the “patient”.
Discussion
This case report details the management of a patient with myopic degeneration who presented with asymptomatic increased IOP 1 day after cataract surgery. The complication was managed in-office with anterior chamber decompression, which temporarily reduced the IOP. However, within 20 minutes, the IOP returned to near-baseline levels. The patient was subsequently prescribed topical ocular hypotensive drops and advised to return in 1 week. At the POW1 visit, the IOP had normalized, but the patient developed mild corneal edema, which resolved without intervention by the POM1 visit. He experienced no lasting complications.
It is important to standardize the definition of postoperative ocular hypertension. Historically, studies have employed varying definitions, complicating comparisons. It has been suggested that postoperative IOP elevation should be defined as an IOP above 30 mmHg or an increase of 10 mmHg relative to preoperative levels.1 This standardization is essential for improving patient outcomes and guiding treatment decisions.
In the early postoperative period, elevated IOP is often caused by obstructions of aqueous outflow from the trabecular meshwork by retained ophthalmic viscosurgical device (OVD), hyphema or pigment dispersion.1 The anterior chamber should be evaluated with gonioscopy to rule out retained lens material, which can cause increased IOP due to obstruction of aqueous outflow by lens proteins and inflammatory cells.3
Steroid-induced glaucoma should be considered if elevated IOP occurs several weeks after initiation of the ophthalmic steroid.4 The pathophysiology of steroid-induced glaucoma is not well understood, but is thought to be due to glycosaminoglycan accumulation, alteration of trabecular meshwork structure and dysregulation of aqueous outflow.4,5 Patients with a history of glaucoma, longer axial length and more pre-operative glaucoma medications have an increased risk of steroid-induced glaucoma following cataract surgery.6
Rarely, elevated IOP can be caused by pupillary block, which occurs when the iris margin forms synechiae to the IOL due to excessive intraocular inflammation, causing iris bombé and secondary angle closure.7 This complication can occur at any point postoperatively, even years after surgery. Gonioscopy should be performed if pupillary block is suspected.
Several conditions can predispose patients to elevated IOP after cataract surgery. If a patient is deemed to be at risk of this complication, prophylactic treatment may be considered. However, there is no consensus on the optimal prophylactic treatment regimen for this complication.8
History of glaucoma, ocular hypertension, pseudoexfoliation syndrome or pigment dispersion syndrome increases risk due to pre-existing compromised trabecular meshwork function.1,9,10 Patients with a history of steroid-induced glaucoma are at increased risk, so less potent steroids or topical non-steroidal anti-inflammatory drugs may be substituted.5
Research has demonstrated that postoperative IOP tends to be higher in eyes with longer axial lengths. The patient in this case had an axial length of 28.05 mm in the surgical eye, which is longer than the average axial length of an emmetropic adult of 22 to 25 mm.11 The increased risk of elevated IOP may be due to excessive movement of the lens-iris diaphragm, causing the iris to contact the IOL or lens capsule and resulting in uveitis and subsequent aqueous outflow obstruction.9,12 Eyes with longer axial lengths may be less rigid, and therefore more susceptible to mechanical trauma.12 A study of highly myopic eyes undergoing cataract surgery found morphologic differences in Schlemm’s canal and the trabecular meshwork in patients who had elevated IOP after surgery compared to those who did not.13 The study suggested preoperative evaluation of these structures with anterior segment optical coherence tomography to identify patients at risk.
Patients taking alpha-1 adrenergic antagonists like tamsulosin are also at increased risk of postoperative elevated IOP, but the pathophysiology is unclear.14 These medications are well known to cause intraoperative floppy iris syndrome, which can complicate surgery and potentially lead to incomplete removal of OVD. Alpha-1 adrenergic antagonists may also relax the iris sphincter muscle, releasing tension on the scleral spur and narrowing the trabecular meshwork.
Additionally, surgeries performed by resident surgeons have been associated with an increased likelihood of postoperative IOP elevation.9 This risk may be due to longer surgical times and greater trauma, which exacerbate the release of pigment and inflammatory cells that obstruct aqueous outflow through the trabecular meshwork.15 Resident surgeons may be less likely to aggressively clear OVD, which also obstructs aqueous outflow.
The long-term effect of increased IOP after cataract surgery has not been well studied.8 If the patient experiences eye pain or nausea, treatment is indicated. When making treatment decisions, clinicians should consider the extent of the IOP elevation, the patient’s comorbidities and the risk of permanent vision loss. Elevated IOP after cataract surgery has been found to cause progression of visual field defects in patients with glaucoma, so treatment should be considered in those patients, particularly if their visual field defects are already severe.16 Elevated IOP can cause microcystic corneal edema due to alteration of the pressure gradient, causing excessive fluid uptake into the corneal stroma, which necessitates treatment to restore corneal health.1 Increased IOP may contribute to a greater risk of non-arteritic ischemic optic neuropathy in the first few days after surgery, so treatment may be considered in patients who have a small cup-to-disc ratio or systemic vascular risk factors.18,19
The first-line treatments for elevated postoperative IOP are oral or topical aqueous suppressant medications.8 Anterior chamber decompression and surgical paracentesis are more invasive procedures that may be reserved for severe, symptomatic cases or when topical and oral medications have been unsuccessful. The choice of treatment depends on the cause of the IOP elevation, the presence of symptoms and the patient’s risk of permanent vision loss.
When retained lens fragments lead to increased IOP, initial treatment involves anti-inflammatory and anti-glaucoma medications. This continues until the fragments and associated inflammation clear, and IOP returns to normal. If medical treatment isn’t effective and there’s a risk of glaucomatous vision loss or other complications like corneal edema, retinal detachment or endophthalmitis, pars plana vitrectomy may be necessary.3
For steroid-induced glaucoma, the initial step is to taper or discontinue the steroid if medically appropriate. IOP control must be balanced with management of intraocular inflammation. Often, topical or oral aqueous suppressants are used to control IOP until the steroid can be safely discontinued. If medical management fails, glaucoma filtering surgery may be considered.4
In cases of increased IOP due to pupillary block, the first line of treatment is neodymium-YAG peripheral iridotomy.7 If this isn’t successful, surgical iridectomy or anterior vitrectomy may be indicated.
Anterior chamber decompression may be considered when the IOP is exceptionally high, when the patient experiences symptoms such as eye pain, nausea or vomiting, or when less invasive methods fail to reduce the IOP adequately. The procedure is performed by obtaining informed consent and then instilling a topical ocular anesthetic, antibiotic and sodium fluorescein.20,21 A blunt, sterile device is used to apply firm but brief pressure to the posterior aspect of the paracentesis to release aqueous humor.22-25 The flow of aqueous out of the corneal wound should be observed using a cobalt blue filter. IOP should be rechecked, and the procedure may be repeated until the IOP reaches an acceptable level. After the procedure, an additional antibiotic drop should be applied and the IOP should be reassessed 1 hour later. Follow-up is recommended within 1 week.25
Potential complications of anterior chamber decompression include pain, subconjunctival hemorrhage, hyphema, decompression retinopathy and endophthalmitis.25 The IOP-lowering effect is transient, returning to 80% of its original level within 1 hour of the procedure, likely because the underlying causes of the elevated IOP persist.24 It could be argued that the potential risks of the procedure outweigh the transient benefit. As a result, it is typically reserved for patients who are highly symptomatic or not responsive to conventional treatment. It is most effective when used in conjunction with longer lasting IOP-lowering treatments, such as oral or topical aqueous suppressants.
In reviewing the management of this case, several considerations arise. Given the patient’s history of myopic degeneration and elevated IOP following cataract surgery in the first eye, prophylactic treatment might have been considered when planning surgery for the fellow eye. Gonioscopy should have been performed to rule out retained lens material. Additionally, as the patient was asymptomatic and lacked a history of glaucoma or corneal disease, pharmacological methods to lower IOP could have been attempted before performing anterior chamber decompression. The rapid IOP rebound within 20 minutes suggested further attempts to lower IOP were warranted. While the patient was scheduled for follow-up in 1 week, a 24-hour follow-up may have been more appropriate, particularly given the IOP rebound. At the POW1 examination, timolol was reduced from twice daily to once daily, even though central corneal edema persisted. Continuing the twice daily dosage should have been considered until the corneal edema resolved.
Conclusion
This case highlights the management of increased intraocular pressure following cataract surgery, specifically the use of anterior chamber decompression. It emphasizes the importance of recognizing when postoperative IOP elevation requires intervention and the management options available. This case serves as a valuable learning experience for practitioners involved in comanaging cataract surgery, emphasizing the need for confidence and competence in handling such complications to ensure optimal patient outcomes.
References
- Grzybowski A, Kanclerz P. Early postoperative intraocular pressure elevation following cataract surgery. Curr Opin Ophthalmol. 2019 Jan;30(1):56–62. DOI: 10.1097/ICU.0000000000000545
- Ahmed II, Kranemann C, Chipman M, Malam F. Revisiting early postoperative follow-up after phacoemulsification. J Cataract Refract Surg. 2002 Jan;28(1):100-8. DOI: 10.1016/s0886-3350(01)00994-4
- Salabati M, Mahmoudzadeh R, Wakabayashi T, Hinkle JW, Ho AC. Indications for surgical management of retained lens fragments. Curr Opin Ophthalmol. 2022 Jan 1;33(1):15-20. DOI: 10.1097/ICU.0000000000000823
- Kersey JP, Broadway DC. Corticosteroid-induced glaucoma: a review of the literature. Eye (Lond). 2006 Apr;20(4):407-16. DOI: 10.1038/sj.eye.6701895
- Phulke S, Kaushik S, Kaur S, Pandav SS. Steroid-induced Glaucoma: An Avoidable Irreversible Blindness. J Curr Glaucoma Pract. 2017 May-Aug;11(2):67-72. DOI: 10.5005/jp-journals-l0028-1226
- Bojikian KD, Nobrega P, Roldan A, Forrest SL, Tsukikawa M, Chen PP. Incidence of and Risk Factors for Steroid Response After Cataract Surgery in Patients With and Without Glaucoma. J Glaucoma. 2021 Apr 1;30(4):159-163. DOI: 10.1097/IJG.0000000000001785
- Gaton DD, Mimouni K, Lusky M, Ehrlich R, Weinberger D. Pupillary block following posterior chamber intraocular lens implantation in adults. Br J Ophthalmol. 2003 Sep;87(9):1109-11. DOI: 10.1136/bjo.87.9.1109
- Miller KM, Oetting TA, Tweeten JP, et al. Cataract in the Adult Eye Preferred Practice Pattern. Ophthalmology. 2022 Jan;129(1):1-126. DOI: 10.1016/j.ophtha.2021.10.006
- Kim JY, Jo MW, Brauner SC, et al. Increased intraocular pressure on the first postoperative day following resident-performed cataract surgery. Eye (Lond). 2011 Jul;25(7):929-36. DOI: 10.1038/eye.2011.93
- Levkovitch-Verbin H, Habot-Wilner Z, Burla N, et al. Intraocular pressure elevation within the first 24 hours after cataract surgery in patients with glaucoma or exfoliation syndrome. Ophthalmology. 2008 Jan;115(1):104-8. DOI: 10.1016/j.ophtha.2007.03.058
- Bhardwaj V, Rajeshbhai GP. Axial length, anterior chamber depth-a study in different age groups and refractive errors. J Clin Diagn Res. 2013 Oct;7(10):2211-2. DOI: 10.7860/JCDR/2013/7015.3473
- Cho YK. Early intraocular pressure and anterior chamber depth changes after phacoemulsification and intraocular lens implantation in nonglaucomatous eyes: Comparison of groups stratified by axial length. J Cataract Refract Surg. 2008 Jul;34(7):1104–9. DOI: 10.1016/j.jcrs.2008.03.023
- Qi J, He W, Lu Q, Zhang K, Lu Y, Zhu X. Schlemm Canal and Trabecular Meshwork Features in Highly Myopic Eyes With Early Intraocular Pressure Elevation After Cataract Surgery. Am J Ophthalmol. 2020 Aug;216:193-200. DOI: 10.1016/j.ajo.2020.02.005
- Bonnell LN, SooHoo JR, Seibold LK, et al. One-day postoperative intraocular pressure spikes after phacoemulsification cataract surgery in patients taking tamsulosin. J Cataract Refract Surg. 2016 Dec;42(12):1753-1758. DOI: 10.1016/j.jcrs.2016.10.009
- Elfersy AJ, Prinzi RA, Peracha ZH, et al. IOP elevation after cataract surgery: results for residents and senior staff at Henry Ford Health System. J Glaucoma. 2016 Oct;25(10):802-806. DOI: 10.1097/IJG.0000000000000421
- Savage JA, Thomas JV, Belcher CD 3rd, Simmons RJ. Extracapsular cataract extraction and posterior chamber intraocular lens implantation in glaucomatous eyes. Ophthalmology. 1985 Nov;92(11):1506–16. DOI: 10.1016/s0161-6420(85)33828-9
- Briceno-Lopez C, Burguera-Giménez N, García-Domene MC, Díez-Ajenjo MA, Peris-Martínez C, Luque MJ. Corneal Edema after Cataract Surgery. J Clin Med. 2023 Oct 25;12(21):6751. DOI: 10.3390/jcm12216751
- Hayreh SS. Anterior ischemic optic neuropathy. IV. Occurrence after cataract extraction. Arch Ophthalmol. 1980 Aug;98(8):1410-6. DOI: 10.1001/archopht.1980.01020040262010
- Shew W, Wang MTM, Danesh-Meyer HV. Nonarteritic Anterior Ischemic Optic Neuropathy After Cataract Surgery: A Systematic Review and Meta-Analysis. J Neuroophthalmol. 2023 Mar 1;43(1):17-28. DOI: 10.1097/WNO.0000000000001625
- Tsai L, ed. 2021-2022 Basic and clinical science course, section 11: lens and cataract. American Academy of Ophthalmology; 2021. p. 184.
- Johnson S. Cataract surgery in the glaucoma patient. New York, NY: Springer; 2009. p. 54
- Bacsal KE, Chee SP. Modified technique for anterior chamber decompression for high intraocular pressure following phacoemulsification. J Cataract Refract Surg. 2006 Dec;32(12):1983; author reply 1983-4. DOI: 10.1016/j.jcrs.2006.07.034
- Chawdhary S, Anand A. Early post-phacoemulsification hypotony as a risk factor for intraocular contamination: in vivo model. J Cataract Refract Surg. 2006 Apr;32(4):609-13. DOI: 10.1016/j.jcrs.2006.01.020
- Hildebrand GD, Wickremasinghe SS, Tranos PG, Harris ML, Little BC. Efficacy of anterior chamber decompression in controlling early intraocular pressure spikes after uneventful phacoemulsification. J Cataract Refract Surg. 2003 Jun;29(6):1087-92. DOI: 10.1016/s0886-3350(02)01891-6
- Skorin L, Lighthizer N, McGee S, Castillo R, Stonecipher K. The ophthalmic office procedures handbook. Philadelphia, PA: Wolters Kluwer; 2024. p. 660-662



