PEER REVIEWED
Risk-Benefit Analysis of Monotherapy with Prednisolone Acetate 1% for Irvine-Gass Syndrome Following Cataract Extraction in a Glaucoma Patient
Alfredo Mazzuca, OD, FAAO, Richard Madonna, OD, MA, FAAO
Abstract
Irvine-Gass syndrome (IGS) is widely recognized as a form of cystoid macular edema that can cause visual decline following cataract extraction. Although a variety of treatment options exist, the role of corticosteroid monotherapy remains clinically relevant. We present the case of a patient with primary angle-closure glaucoma (PACG) who developed pseudophakic cystoid macular edema consistent with IGS and was treated successfully with prednisolone acetate 1% monotherapy. Intraocular pressure (IOP) was monitored closely throughout therapy. This case underscores the importance of individualized management, highlights the therapeutic potential of corticosteroid monotherapy, and emphasizes the necessity of vigilant IOP surveillance when corticosteroids are used in glaucoma patients.
Keywords
Background
The purpose of this case study is to report the effectiveness of topical prednisolone acetate 1% for treating IGS, with a focus on its ability to improve visual acuity and central macular thickness. This case underscores the importance of distinguishing the causes of reduced vision following cataract extraction. It highlights a patient in whom IGS developed after uncomplicated cataract extraction. Optometric management plays an essential role in evaluating and addressing postoperative complications, thereby ensuring optimal visual outcomes and ocular health for patients. Specifically, third- and fourth-year optometry students and residents should demonstrate proficiency in identifying common causes of postoperative reduced vision following cataract extraction.
Case History
A 73-year-old female with moderate-stage primary angle closure glaucoma in both eyes presented to the State University of New York (SUNY) Optometry University Eye Center for a scheduled follow-up. Despite excellent uncorrected vision immediately following cataract extraction, she reported progressively declining vision in her left eye over several months after the surgery.
Her pertinent ocular history included laser peripheral iridotomy (LPI) of the right eye in 2011 prior to cataract extraction of the right eye in 2012. LPI was performed in the right eye due to the appearance of peripheral anterior synechiae (PAS) observed inferiorly and nasally on 4-mirror gonioscopy. The left eye did not undergo LPI, presumably because there was no evidence of PAS and IOP had been adequately controlled with topical IOP-lowering therapy. Cataract extraction was performed on the left eye in late 2021. She was subsequently inherited for care following cataract extraction of the left eye.
Prior to cataract extraction, maximum recorded intraocular pressures (IOP) were 56 mmHg and 40 mmHg in the right and left eyes, respectively. At presentation, she was using latanoprost 0.005% once daily in both eyes, dorzolamide–timolol ophthalmic solution (22.2 mg/mL – 6.8 mg/mL; Cosopt ®) twice daily in both eyes, and brimonidine 0.2% twice daily in both eyes, with excellent reported adherence.
Her medical history included medical management of rheumatoid arthritis, diagnosed in 2002, overseen by an outside rheumatologist who prescribed abatacept (Orencia ®). She also had a reported positive family history of glaucoma on her father’s side, which was relevant to her own diagnosis and management considerations.
Case Description
Visit One
Visual acuities at presentation were 20/20 -2 in the right eye and 20/40 -2 in the left without correction. The left eye pinhole acuity was 20/25 +1. Pupils were round, reactive and responsive to light. There was no relative afferent pupillary defect (RAPD) in either eye. Confrontation visual fields were grossly full to finger counting in each eye. Extraocular motilities were full in each eye. Best-corrected visual acuities following manifest refraction were 20/20 in the right eye and 20/25 in the left.
Slit lamp biomicroscopy of the anterior segments revealed no pathology of the adnexa, eyelids, sclera and conjunctiva. Both corneas had temporal cataract excision scars, with a well-demarcated stromal opacification. Angles were determined to be “open” on Van Herrick technique using slit lamp biomicroscopy. A patent peripheral iridotomy at 11:00 was present in the iris of the right eye, but otherwise, it was unremarkable. The iris of the left eye was unremarkable. In both eyes, the anterior chamber was deep and quiet, with no cells noted. Posterior chamber intraocular lenses (PCIOLs) were present in both eyes, with acceptable positioning and posterior capsules were clear.
IOP in each eye was 16 mmHg on Goldmann applanation tonometry. Tropicamide 1% and phenylephrine 2.5% were used to dilate the patient’s pupils. Indirect ophthalmoscopy with a 90D fundus lens revealed posterior vitreous detachments in both eyes. The right eye had a cup-to-disc ratio of 0.70, with inferior rim thinning, but otherwise a distinct border and a well-perfused appearance. The left eye had a cup-to-disc ratio of 0.60 with distinct borders and a well-perfused appearance. The macula of the right eye was flat with an evident foveal reflex. The macula of the left eye was elevated with an absent foveal reflex. Indirect ophthalmoscopy with a 20D lens was performed for peripheral evaluation. No evidence of accompanying retinopathy, such as hemorrhages or exudates, was noted on examination. Both eyes had normal vessel contour and caliber, and the retina was flat and intact 360 degrees.
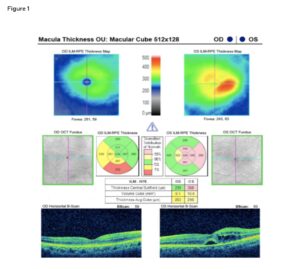
Figure 1: Baseline SD-OCT imaging at Visit One. Scan quality of the right eye was 8; scan quality of the left eye was 9; these were considered acceptable. Baseline imaging of the left eye macula revealed central subfield thickness of 358 microns. The left eye demonstrated intraretinal cystic changes and subretinal fluid on the B-scan, consistent with active central macular edema. The right eye macular scan showed a central thickness of 239 microns with an apparent mildly thin inferior quadrant. The right eye showed normal retinal architecture and no signs of fluid or structural abnormality. Click to enlarge
A macular cube 512 x 218 scan was acquired using Zeiss Cirrus 5000 spectral-domain optical coherence tomography (SD-OCT) to determine central macular thickness. The right eye demonstrated normal foveal contour without subretinal or intraretinal fluid on the B-scan. However, the inferior quadrant demonstrated relative thinning on the Early Treatment of Diabetic Retinopathy Study (ETDRS) grid, falling just below the average range for age-matched controls. There was no evidence of edema, subretinal fluid or structural disruption. The central subfield thickness of the right eye was 239 microns – stable and within normal age-adjusted limits. The left eye demonstrated a central subfield thickness of 358 microns which was “Above Normal” relative to normative data for age, consistent with macular thickening. There was evident loss of foveal contour with cystoid intraretinal spaces and subretinal fluid on the B-scan, shown to be predominantly in the central and temporal macula on the ETDRS grid. (Figure 1).
The presence of cystoid macular edema in the context of recent cataract extraction suggested IGS. To treat the condition, ketorolac 0.5% was initiated 4 times daily in the left. The patient was instructed to discontinue the use of latanoprost 0.005% in both eyes and continue with dorzolamide–timolol and brimonidine 0.2% twice daily in both eyes. A follow-up was scheduled in 4 weeks to assess the progress, and the patient was released with return precautions, such as acute onset pain, blurred vision or distorted vision. She was instructed to return immediately to the clinic in the event any changes in vision were noted.
Visit Two
The patient returned for a doctor-directed follow-up 4 weeks later. She had self-discontinued ketorolac 0.5% 2 weeks prior due to ocular surface irritation. She had continued use of latanoprost 0.005% since the last clinic visit. She reported adherence to brimonidine tartrate 0.2% and dorzolamide–timolol twice daily in both eyes. No new visual symptoms were reported during case history. Entering visual acuities were 20/25 -1 in the right eye and 20/80 -1 in the left eye without correction. Assessment of pupillary testing, extraocular motilities and confrontation visual fields were within normal limits and stable compared to previous examinations.
Anterior segment evaluation revealed no cells or flare of either eye. IOP were 16 mmHg in the right eye and 14 mmHg in the left on Goldmann applanation tonometry. Tropicamide 1% and phenylephrine 2.5% were used to dilate the patient’s pupils. Posterior pole findings observed with indirect ophthalmoscopy using a 20D and 90D lens on biomicroscopy were stable compared to the previous visit. No vitreous cells were noted on direct ophthalmoscopy using a slit lamp.
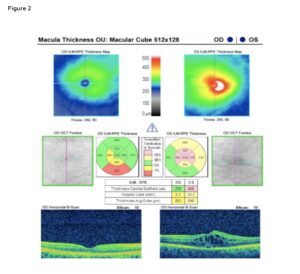
Figure 2: SD-OCT imaging at Visit Two. Scan quality of the right eye was 9; scan quality of the left eye was 7; these were considered acceptable. At the follow-up visit, SD-OCT of the LE showed progression of the macular edema, with central subfield thickness increasing to 458 microns. There was worsening of cystoid spaces and expansion of subretinal fluid, leading to increased foveal disruption in the left eye. These findings prompted initiation of corticosteroid therapy. The right eye central macular thickness remained stable at 239 microns. The retinal contour of the right eye was unchanged with no structural changes. Click to enlarge
A macular cube 512 x 218 scan was acquired using Carl Zeiss Cirrus 5000 SD-OCT to assess the response to ketorolac 0.5%. The scan demonstrated stable central subfield thickness of the right eye with no structural abnormalities or fluid present on the B-scan. The central subfield thickness remained stable at 239 microns – stable and within normal age-adjusted limits. The mild inferior parafoveal thinning remained unchanged on the ETDRS grid. The central subfield of the left eye increased from 358 to 458 microns, representing increasing central macular edema. On the ETDRS grid, more inner and outer subfields shifted into the “Above Normal” zone, particularly in the temporal and superior parafoveal regions, deviating further from age-based norms. The B-scan showed progression of the cystoid changes and increased subretinal fluid, resulting in greater foveal disruption (Figure 2).
Given the presence of increased intraretinal thickness in the left eye, the patient was started on prednisolone acetate 1% every 2 hours while awake. The patient was again advised to discontinue latanoprost 0.005%. A follow-up was scheduled in 1 week to assess progress and the patient was released with clear instructions for follow-up if symptoms worsened.
Visit Three
The patient returned for a doctor-directed follow-up 1 week later. She reported excellent adherence to prednisolone acetate 1% every 2 hours while awake in the left eye. She had discontinued using latanoprost 0.005% as instructed and reported adherence to brimonidine tartrate 0.2% and dorzolamide–timolol twice daily in both eyes. No new visual symptoms were reported. Entering visual acuities were 20/25 -1 in the right eye and 20/60 +2 in the left, without correction. Pinhole acuity of the left eye demonstrated no improvement. Assessment of pupillary testing, extraocular motilities and confrontation visual fields were within normal limits and stable compared to previous examinations.
Anterior segment evaluation revealed no cells or flare of either eye. IOP were 16 mmHg in the right eye and 13 mmHg in the left on Goldmann applanation tonometry. Due to the lack of visual improvement in the left eye, a dilated fundus exam was recommended to evaluate for the progression of macular pathology. However, the patient deferred dilation due to a time constraint. In lieu of dilation, indirect ophthalmoscopy using a 90D lens was performed, allowing for a limited yet sufficient evaluation of the posterior pole. Findings were stable compared to the previous visit. SD-OCT imaging of the macula was not repeated at this visit given the deferred dilation and the absence of new visual symptoms.
Given the clinical stability and absence of acute signs of inflammation, a steroid taper was initiated to minimize the risk of steroid-induced side effects while maintaining control of any residual inflammatory activity. The patient was instructed to taper prednisolone acetate 1% to 4 times daily for 1 week, then twice daily for 1 week, and once daily for 1 week in the left eye. A follow-up was scheduled in 1 week to assess progress and the patient was reeducated on return precautions.
Visit Four
The patient returned for a doctor-directed follow-up 1 week later. She reported using the prednisolone acetate 1% 4 times daily in the left, which was twice the daily frequency that had been prescribed. She had discontinued using latanoprost 0.005% as instructed and reported adherence to brimonidine tartrate 0.2% and dorzolamide–timolol twice daily, in both eyes. Improved vision of the left eye was reported. Entering visual acuities were 20/25 -1 in the right eye and 20/80 in the left, without correction. Pinhole acuity of the left eye was 20/40. Assessment of pupillary testing, extraocular motilities and confrontation visual fields were within normal limits and stable compared to previous examinations.
Anterior segment evaluation revealed no cells or flare of the left eye. IOP were 18 mmHg in the right eye and 15 mmHg in the left on Goldmann applanation tonometry. Tropicamide 1% and phenylephrine 2.5% were used to dilate the patient’s pupils. Posterior pole findings observed with indirect ophthalmoscopy using a 20D and 90D lens on biomicroscopy were stable compared to the previous visit. No vitreous cells were noted on direct ophthalmoscopy using a slit lamp.
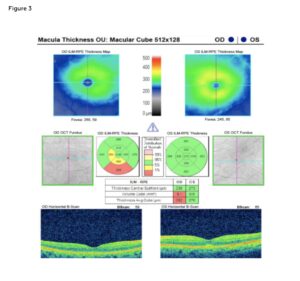
Figure 3: SD-OCT imaging at Visit Four. Scan quality of the right eye was 9; scan quality of the left eye was 10; these were considered acceptable. Follow-up SD-OCT imaging demonstrated stable central subfield thickness in the right eye (238 microns) with no structural abnormalities. The left eye showed marked improvement in central subfield thickness from 458 to 273 microns, with near complete resolution of subretinal fluid and intraretinal cystic changes. The right eye macular thickness and contour remained stable with a central subfield thickness of 238 microns. The retinal contour of the right eye was unchanged with no structural changes. Click to enlarge
A macular cube 512 x 218 scan was acquired using Carl Zeiss Cirrus 5000 SD-OCT to determine central macular thickness. SD-OCT of the right eye demonstrated stable central subfield thickness at 238 microns. No structural changes were noted. SD-OCT of the left eye showed improvement in central subfield thickness from 458 to 273 microns, with near complete resolution of subretinal fluid and intraretinal cystic changes. These findings support a positive anatomical response to corticosteroid therapy, consistent with improved control of macular edema (Figure 3).
Given substantial resolution of subretinal fluid and intraretinal cystic spaces, the patient was instructed to taper the prednisolone acetate 1% to 3 times daily for 1 week, then twice daily for 1 week in the left eye. She was instructed to not restart the latanoprost 0.005% at this time. The patient was reminded to continue with brimonidine tartrate 0.2% and dorzolamide–timolol twice daily, in both eyes. A follow-up was scheduled in 1 week to assess progress and the patient was released with clear instructions to return sooner if symptoms worsened.
Visit Five
The patient returned for a doctor-directed follow-up 1 week later. She reported using the prednisolone acetate 1% 2 times daily in the left eye, as instructed. She reported adherence to brimonidine tartrate 0.2% and dorzolamide–timolol twice daily, in both eyes. No new symptoms were reported. Entering visual acuities were 20/25 -2 in the right eye and 20/25 in the left, without correction. Assessment of pupillary testing, extraocular motilities and confrontation visual fields were within normal limits and stable compared to previous examinations. Anterior segment evaluation revealed no cells or flare of the left eye.
IOP were 16 mmHg and 14 mmHg in the right and left eyes, respectively, on Goldmann applanation tonometry. Tropicamide 1% and phenylephrine 2.5% were used to dilate the patient’s pupils. Posterior pole findings observed with indirect ophthalmoscopy using a 20D and 90D lens on biomicroscopy were stable compared to the previous visit. No vitreous cells were noted on direct ophthalmoscopy using a slit lamp.
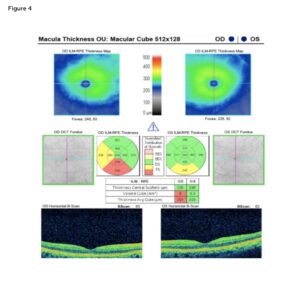
Figure 4: SD-OCT imaging at Visit Five. Scan quality of the right eye was 9; scan quality of the left eye was 8; these were considered acceptable. At the follow-up visit SD-OCT of the left eye demonstrated marked improvement, with central subfield thickness reduced to 245 microns and complete resolution of prior intraretinal and subretinal fluid. The right eye demonstrated central subfield thickness of 235 microns. The retinal contour of the right eye was unchanged with no structural changes. Click to enlarge
A macular cube 512 x 218 scan was acquired using Carl Zeiss Cirrus 5000 SD-OCT to determine central macular thickness (Figure 4). SD-OCT macula for the right eye demonstrated structural stability with a central subfield thickness of 235 microns, consistent with prior measurements. The left eye showed substantial improvement, with central subfield thickness decreasing from a peak of 458 microns (Figure 2) to 245 microns, nearing the normal range. B-scan imaging confirming resolution of both subretinal fluid and intraretinal cystic spaces, indicating effective response to the prednisolone acetate 1% therapy. The retinal architecture in both eyes was preserved, with no ongoing edema or distortion of the macular layers.
Given the complete resolution of the intraretinal and subretinal fluid, the patient was instructed to taper the prednisolone acetate 1% once daily for 1 week in the left eye. She was instructed to not restart the latanoprost 0.005% at this time. The patient was reminded to continue with brimonidine tartrate 0.2% and dorzolamide–timolol twice daily, in both eyes. A follow-up was scheduled in 1 week to assess progress and the patient was released with clear instructions for follow-up if symptoms worsened.
Visit Six
The patient returned for a doctor-directed follow-up 1 week later. No new symptoms were reported. She reported using the prednisolone acetate 1% once daily in the left eye, as instructed. She reported adherence to brimonidine tartrate 0.2% and dorzolamide–timolol twice daily, in both eyes. Assessment of pupillary testing, extraocular motilities and confrontation visual fields were within normal limits and stable compared to previous examinations. Entering visual acuities were 20/25 -1 in the right eye and 20/20 -1 in the left, without correction. Anterior segment evaluation revealed no cells or flare of the left eyeon direct ophthalmoscopy with slit lamp.
IOP were 18 mmHg in the right eye and 17 mmHg in the left on Goldmann applanation tonometry. Indirect ophthalmoscopy using a 90D lens was performed, allowing for a limited yet sufficient evaluation of the posterior pole. Findings were stable compared to the previous visit. Given the excellent visual outcome, along with absence of patient symptoms, the patient was instructed to discontinue prednisolone acetate 1%. The patient was placed on a maintenance dose of ketorolac tromethamin 0.5% (Acular ®) 1 time daily in the left eye. No changes were noted at the final visit scheduled for the management of this condition. A follow-up was scheduled in 3 weeks to assess progress and the patient was released with clear instructions for follow-up if symptoms worsened.
Education Guidelines
In a didactic setting, third- and fourth-year optometry students and residents can be presented with this case and associated SD-OCT images in slideshow format or imaging forums to develop a primary diagnosis, differential diagnosis and management plan specific to IGS. Subsequently, structured discussion questions can then guide exploration of the case history, characteristic SD-OCT features and treatment considerations in the context of postoperative inflammation. In a clinical setting, students and residents should be encouraged to review postoperative SD-OCT scans and identify early signs of IGS, such as parafoveal cystic changes, retinal thickening and changes to subfoveal contour. During follow-up visits, students and residents can be tasked with explaining the inflammatory pathophysiology of IGS including surgical trauma, cytokine-mediated endothelial changes and blood-retinal barrier disruption and discussing treatment strategies in patients with comorbidities such as glaucoma. Learners should also participate in decision-making strategies regarding when to escalate therapy or discontinue specific drops (e.g., prostaglandin analogs). Finally, these concepts can be reinforced by tracking the resolution of IGS across visits, emphasizing the importance of serial SD-OCT imaging in monitoring treatment response and preventing chronic visual loss.
Learning Objectives
-
- Identify common causes of reduced vision following cataract extraction.
- Outline the pathophysiology, related mediators and timeline of intraocular inflammation following cataract extraction.
- Recognize the presentation of macular edema on SD-OCT and describe how serial imaging can be implemented to track treatment response.
- Compare appropriate treatment modalities for IGS, including topical therapy, intraocular injections and laser therapy.
- Determine indications for escalating treatment for refractory cases of IGS.
Key Concepts
-
- Complications that can arise from iatrogenic postoperative inflammation that can contribute to the development of IGS.
- The timeline, risks factors and key differential diagnoses associated with IGS following cataract extraction.
- The value of SD-OCT in characterizing the disease course and in guiding the evaluation, treatment and management of IGS.
- Appropriate therapeutic and management strategies to improve visual outcomes in IGS.
Discussion Questions
-
- What disease processes result in reduced vision following cataract surgery?
- What are the components of intraocular inflammation?
- What complications can arise from chronic intraretinal fluid that lead to reduced vision?
- What are common vasculopathic disorders that can also present with macular edema?
- How do you classify presentations of macular edema?
- What proposed role does intraocular inflammation play in Irvine-Gass Syndrome?
- What treatment modalities exist to reduce intraocular inflammation?
- What is the mechanism of action of steroidal and non-steroidal agents in reducing intraocular inflammation?
- When would you refer a patient with Irvine-Gass syndrome to an external provider for further treatment?
Learning Assessment
In a case-based discussion, learners should be able to explain the underlying pathophysiology of IGS, including the role of postoperative inflammation and disruption of the blood-retina barrier. Participants will review clinic snapshots involving reduced vision following cataract extraction to identify likely causes and differentiate them from unrelated conditions. Group discussion and guided questioning will be used to assess comprehension of etiology and mechanisms of intraocular inflammation, as well as the underlying pathophysiology of IGS. Sequences of SD-OCT imaging can be presented to emphasize the role of serial analysis at follow-up visits to guide the treatment timeline. Furthermore, SD-OCT imaging can play a role in emphasizing intra- and sub-retinal anatomical landmarks. Learners should be able to identify mediators involved in the inflammatory process and how treatment targets specific mediators. Finally, awareness of referral criteria will be gauged by having participants outline the decision-making process for escalating care to a specialist when complications arise. This multifaceted approach ensures that learners do not rely on factual information recall but rather apply it to realistic clinical scenarios.
In evaluating the etiology of macular edema, learners should be encouraged to begin with the more common causes and then systemically work toward the less frequent ones, guided by the accompanying clinical context. This process should consider both the patient’s medical and ocular histories, as well as adjunctive clinical signs. For example, diabetic macular edema typically develops without associated anterior segment inflammation, postoperative inflammatory changes following cataract surgery may point more directly to IGS. Emphasizing this structured, context-driven approach helps students prioritize their differential diagnosis logically and tailor management more effectively.
Discussion
Irvine-Gass syndrome (IGS) is the most common complication of cataract surgery.1 The mechanism of pathogenesis was first thought to be incarceration of the vitreous in the anterior segment with resulting vitromacular traction; however, it is now widely recognized as an inflammatory-mediated process.2–4 The occurrence of IGS in modern cataract extraction has an estimated incidence of 2–12%.5 Peak incidence of IGS following cataract extraction is around 4 to 6 weeks postoperatively.6 Risk factors for IGS include diabetes, capsule rupture, pre-existing epiretinal membrane, uveitis, retinal vein occlusion and retinal detachment repair.7 Furthermore, higher incidences are noted with rupture of the posterior capsule, intracapsular cataract extraction and iris fixed intraocular lenses.8 Diabetes mellitus, regardless of the presence of diabetic retinopathy, is an independent risk factor for the development of postoperative IGS in cataract surgery patients.9 Although most cases are self-limiting, persistent or visually significant IGS requires timely treatment to prevent chronic visual impairment.10
Pathophysiology and Complications of IGS
IGS is characterized by fluid accumulation in the macula following uncomplicated cataract extraction, which leads to reduced visual acuity, distorted vision and visual disturbances.4 Postoperative inflammation is considered the primary catalyst for IGS following cataract surgery.4 During cataract extraction, manipulation of the anterior chamber releases arachidonic acid from uveal tissue, initiating the release of inflammatory mediators such as leukotrienes and prostaglandins.11–12 These mediators diffuse throughout the vitreous cavity and toward the retina, where they disrupt the blood–retinal barrier by increasing perifoveal capillary permeability.13 Despite their diffuse distribution, fluid accumulates preferentially at the fovea due to its high metabolic activity and lack of vascular supply within the foveal avascular zone.8 As a result, leakage of intravascular contents from dilated perifoveal capillaries accumulates in the outer plexiform and inner nuclear layers, forming cystic spaces that can coalesce into larger pockets of intraretinal fluid.8 When vascular permeability exceeds the drainage capacity of the retinal pigment epithelium, microcysts develop in the outer plexiform and inner nuclear layers, eventually merging into larger intraretinal cysts.8 If left unresolved, prolonged edema can progress to structural complications. Chronic fluid accumulation may lead to lamellar macular holes, persistent subretinal fluid, foveal atrophy, epiretinal membrane, macular ischemia and macular fibrosis.8,14 Because of these risks, rapid recognition and treatment of IGS is essential to prevent photoreceptor dropout and irreversible central retinal damage.15
Prognosis is guided by SD-OCT biomarkers. These include disorganization of the inner retinal layers, presence of intraretinal cystoid spaces, photoreceptor outer segment length and integrity of the external limiting membrane and ellipsoid zone.14 Disorganization of the inner retinal layers involving more than 50% of the central 1 mm foveal zone is associated with poor visual prognosis.16 Larger intraretinal cysts are often linked to macular ischemia, with giant cysts disrupting the outer nuclear layer and ellipsoid zone and resulting in worse visual prognosis.14
Differential Diagnosis of IGS
Differential diagnoses for macular edema should include several key conditions that must be distinguished from IGS. Differential diagnoses for the patient’s IGS included diabetic macular edema (DME), retinal vein occlusion (RVO), uveitic macular edema and central serous chorioretinopathy (CSCR). Each condition has distinct clinical presentations, patient demographics and SD-OCT characteristics that aid in accurate diagnosis and appropriate treatment selection.
Macular edema is the primary cause of vision loss in patients with diabetic retinopathy.17 In diabetic retinopathy, cell-cell junctions between endothelial cells are damaged, resulting in the increased leakage of plasma, lipid and red blood cells; clinically, this is noted as edema, hard exudates and intraretinal hemorrhages.18-19 Retinal hypoxia causes capillary hyperpermeability through vascular autoregulation dysfunction, primarily due to hypoxia-induced vascular endothelial growth factor (VEGF) upregulation.20 Expression of VEGF leads to increased permeability, obstruction and damage of retinal capillaries, resulting in serous blood components leaking.21 Since the patient did not report having diabetes mellitus and no other signs of diabetic retinopathy were observed, DME was ruled out as a potential differential. Given the unilateral presentation of macular edema in this case, along with the patient’s history of recent cataract extraction, IGS became a more likely diagnosis.
Macular edema can occur as a complication of RVO. The pathogenesis of RVO is believed to involve vascular endothelial damage and compression of the retinal vein, ultimately leading to thrombus formation.22 Hypoxic retinal cells release VEGF, which promotes vascular permeability and vascular proliferation by binding to endothelial cell receptors.22 Vascular endothelial injury increases retinal capillary pressure, promoting fluid translocation into the extracellular space and development of macular edema.23 The patient presented with no history of vasculopathy. Fundoscopic examination revealed no accompanying retinopathy, including hemorrhages or exudates. The normal fundus appearance and absence of vasculopathic disease ruled out RVO as the cause of macular edema.
Uveitic macular edema involves a breakdown of the blood-retinal barrier due to release of inflammatory mediators such as VEGF, which results in leakage of fluid into retinal tissue and formation of cystic spaces in the outer plexiform layer.24 Defective RPE can also lead to accumulation of fluid underneath the neurosensory retina.24 Changes to the choroid, which influences fluid homeostasis across the outer blood-retinal barrier, can affect the process of retinal fluid reabsorption across RPE cells.24 Corticosteroids are the mainstay of controlling inflammation in noninfectious uveitis complicated by macular edema; topical corticosteroids may be effective in milder cases of uveitic macular edema.25 Topical non-steroid anti-inflammatory drugs (NSAIDs) can be used in milder cases or as adjunctive treatment with steroids.25 Periocular corticosteroids are usually effective in controlling uveitic macular edema, though repeat injections may be required during the course of the disease.25 Uveitic macular edema was excluded given the lack of anterior chamber inflammation on slit-lamp examination and the rarity of uveitis in rheumatoid arthritis patients.26
Central serous chorioretinopathy (CSCR) should be considered as a differential due to the presence of subretinal fluid noted on SD-OCT. CSCR is a retinal disorder characterized by localized serous detachment of the macula.27 During the disease process, choroidal vessels become hyperpermeable, leading to increased tissue pressure and disruption of the anatomic integrity of the retinal pigment epithelium (RPE); this results in choroidal fluid detaching the neurosensory retina.27 Treatment of CSCR consists of restoring choroidal vasculature and restoring RPE and photoreceptor function.28 The temporal association with recent ocular surgery made the diagnosis of CSCR less likely, along with preceding intraretinal fluid accumulation.
Treatment Options for IGS
Universal first-line treatment for IGS appears to be topical NSAIDs, either as monotherapy or in combination with corticosteroids.4 NSAIDs reduce inflammation by inhibiting cyclo-oxygenase (COX) enzymes, thereby decreasing prostaglandin production.29-30 Topical NSAIDs have demonstrated efficacy in reducing acute or chronic postoperative IGS after cataract extraction.31 They offer advantages such as stable IOP during application, a lower risk of infection and analgesic effects.31 Transient stinging and irritation are common adverse effects from topical NSAIDs.32 The patient was initially managed with NSAID monotherapy as first-line treatment, but developed ocular surface irritation that limited tolerability. The decision was made to discontinue the NSAID and proceed with corticosteroid monotherapy to maintain anti-inflammatory coverage while avoiding further surface discomfort.
Corticosteroids act higher in the inflammatory cascade by inhibiting phospholipase-A2 and preventing the formation of arachidonic acid, while also inhibiting COX enzymes.33 However, they are associated with risks such as elevated IOP, delayed wound healing and increased susceptibility to secondary bacterial infection.34 Furthermore, cataract formation is a well-known side effect of steroid treatment that leads to reversible vision impairment.35 Despite their well-established anti-inflammatory and VEGF-inhibiting properties, and the potential risk associated with their use, the relative benefits of corticosteroids compared with NSAIDs for postoperative inflammation remain unclear.36 Given this uncertainty and the patient’s intolerance to NSAIDs, initiating corticosteroid monotherapy was deemed the most appropriate strategy to ensure adequate anti-inflammatory control while minimizing discomfort, with appropriate monitoring for IOP and ocular surface health.
Periocular steroids, including intravitreal and sub-tenon administration, have been shown to improve vision and promote resolution of IGS, particularly in cases refractory to topical therapy.4 Sub-tenon injections are considered a cost-effective alternative and avoid the risk of intraocular inflammation associated with intravitreal administration.10 Ozurdex, a biodegradable intravitreal drug delivery system that maintains continuous delivery of preservative free dexamethasone, effectively treats refractory cases and may decrease the need for retreatment.37 In this patient, these periocular and intravitreal options were not considered necessary, as topical prednisolone acetate 1% provided adequate anti-inflammatory effect while minimizing procedural risk. This highlights the principle of therapeutic escalation: invasive approaches should be reserved for cases where simpler, lower-risk strategies have failed to provide sufficient response.
Corticosteroid Response in Glaucoma Patients
Steroid-induced glaucoma is a form of secondary glaucoma that occurs when elevated IOP leads to optic neuropathy.38 The underlying mechanism involves increased resistance to aqueous outflow at the level of the trabecular meshwork.38 Trabecular meshwork cells, which contain glucocorticoid receptors, are directly affected by corticosteroids, leading to alterations in cell migration and phagocystosis.39 In addition, corticosteroids promote extracellular matrix deposition, particularly collagen and fibronectin, in the juxtacanalicular region, further contributing to increased outflow resistance.39
Individuals who demonstrate elevated IOP after steroid use are termed “steroid responders,” defined as IOP of 21–24 mmHg or an increase of 5–10 mmHg from baseline.40 Among commonly used agents, prednisolone is generally associated with a lower risk of significant IOP elevation than dexamethasone, though both can cause clinically meaningful IOP spikes.41 Approximately one third of the population are steroid responders after 2 or more weeks or topical glucocorticoid use, with the prevalence exceeding 90% in patients with pre-existing POAG.42–44 Steroid-induced IOP increase occurs 3-6 weeks following topical corticosteroid use and often normalizes within 2 weeks of cessation.38 Sustained IOP rises secondary to steroid use can cause vision loss, usually through glaucomatous damage to the optic nerve.45 From a management standpoint, this means that timing and duration of therapy matter as clinicians cannot assume that short-term use is entirely risk-free. In practice, even modest IOP spikes can matter in a glaucomatous eye already near its threshold of optic nerve tolerance.
Notably, our patient with a history of PACG was treated with cataract extraction, which has been shown to significantly reduce IOP and glaucoma drug requirements.46 Although pseudophakia deepens the anterior chamber, patients with a history of PACG remain susceptible to steroid-induced elevation, as trabecular outflow compromise persists and these eyes continue to show IOP fluctuations and glaucomatous progression after lens extraction.47–48 In our patient with known PACG, these risks were carefully considered. While corticosteroid therapy was necessary for postoperative inflammation control, we closely monitored IOP for abnormal spikes, recognizing both the heightened risk of steroid response and potential for glaucomatous progression.
SD-OCT imaging was critical for tracking the temporal response to prednisolone acetate 1%. Its non-invasive nature and superior morphologic resolution compared with color photography and angiography make it well-suited for detecting subtle macular changes, particularly in IGS, where defective RPE function and choroidal alterations can allow intraretinal fluid to accumulate subretinally where the RPE is unable to efficiently clear it.24,49 SD-OCT also provides a normative database with a color-based scale for measurement comparison, though thinner-than-average macular thickness in the RE maye reflect physiologic interocular variation.50 Importantly, SD-OCT normative databases may misrepresent certain “normal” demographic profiles, exaggerating apparent thinning of central retinal thickness, and age-related thinning of retinal layers, particularly photoreceptors, is a well-documented trend.50 Subacute autoimmune retinopathy may be considered in the differential diagnosis, as retinal atrophy and reduced central macular thickness on SD-OCT are characteristic findings, although discussion of this possibility is beyond the scope of this report.51 In this case, the absence of fluid or structural abnormalities on imaging, coupled with a lack of visual symptoms, supported a benign explanation for asymmetry.
For glaucoma patients with IGS, a tailored management strategy is required. Corticosteroid therapy carries the risk of steroid-induced IOP elevation, making close surveillance of IOP mandatory. Adjustments to glaucoma therapy or discontinuation of steroids may be necessary if acute spikes occur. In our patient with PACG, SD-OCT imaging in combination with vigilant IOP monitoring helped guide the use of corticosteroid monotherapy. While this case did not incorporate an Amsler grid, such tools can provide additional value by allowing patients to self-monitor central vision changes at home. Ultimately, coordination of diagnostic imaging, IOP monitoring and clinical examination allows for effective use of corticosteroids in this high-risk population.
Conclusion
Prednisolone acetate 1% monotherapy can be effective for IGS, even in patients with established glaucoma. In such cases, however, the risk of corticosteroid-induced IOP elevation must be carefully balanced against the anti-inflammatory benefit. Frequent monitoring is essential to ensure treatment does not inadvertently accelerate glaucomatous damage. Prompt initiation of therapy increases the likelihood of full visual recovery while reducing the risk of chronic macular changes.
Management should remain flexible and individualized. Patients with glaucoma may require closer follow-up or adjustments in their IOP-lowering medications. A stepwise approach can be useful, for example beginning with topical monotherapy but remaining prepared to escalate therapy if clinic response is incomplete or if side effects arise.
Equally important is patient education. Setting expectations regarding the need for regular visits, the possibility of treatment adjustments and the importance of adherence helps optimize outcomes. With timely diagnosis, thoughtful therapeutic selection and close monitoring, favorable long-term visual outcomes can be achieved in IGS.
References
- Grzybowski A, Sikorski BL, Ascaso FJ, Huerva V. Pseudophakic cystoid macular edema: update 2016. Clin Interv Aging. 2016 Sep 9;11:1221-9. DOI: 10.2147/CIA.S111761
- Gass JD, Norton EW. Cystoid macular edema and papilledema following cataract extraction: a fluorescein fundoscopic and angiographic study. Arch Ophthalmol. 1966 Nov;76(5):646-61. DOI: 10.1001/archopht.1966.03850010648005
- Gass JD, Norton EW. Fluorescein studies of patients with macular edema and papilledema following cataract extraction. Trans Am Ophthalmol Soc. 1966;64:232-49.
- Ferro Desideri L, Arun K, Bernardi E, Sagurski N, Anguita R. Incidence, pathogenesis, risk factors, and treatment of cystoid macular oedema following cataract surgery: a systematic review. Diagnostics (Basel). 2025 Mar 10;15(6):667. DOI: 10.3390/diagnostics15060667
- Guo S, Patel S, Baumrind B, et al. Management of pseudophakic cystoid macular edema. Surv Ophthalmol. 2015 Mar-Apr;60(2):123-37. DOI: 10.1016/j.survophthal.2014.08.005
- Bahrami B, Zhu M, Hong T, Chang A. Diabetic macular oedema: pathophysiology, management challenges and treatment resistance. Diabetologia. 2016 Aug;59(8):1594-608. DOI: 10.1007/s00125-016-3974-8
- Chu CJ, Johnston RL, Buscombe C, et al. Risk factors and incidence of macular edema after cataract surgery: a database study of 81,984 eyes. Ophthalmology. 2016 Feb;123(2):316-23. DOI: 10.1016/j.ophtha.2015.10.001
- Flach AJ. The incidence, pathogenesis, and treatment of cystoid macular edema following cataract surgery. Trans Am Ophthalmol Soc. 1998;96:557-634.
- Oyewole K, Tsogkas F, Westcott M, Patra S. Benchmarking cataract surgery outcomes in an ethnically diverse and diabetic population: final post-operative visual acuity and rates of post-operative cystoid macular oedema. Eye (Lond). 2017 Dec;31(12):1672-7. DOI: https://doi.org/10.1038/eye.2017.96
- Orski M, Gawęcki M. Current management options in Irvine-Gass syndrome: a systemized review. J Clin Med. 2021 Sep 25;10(19):4375. DOI: 10.3390/jcm10194375
- Benitah NR, Arroyo JG. Pseudophakic cystoid macular edema. Int Ophthalmol Clin. 2010 Winter;50(1):139-53. DOI: 10.1097/IIO.0b013e3181c551da
- Hudes GR, Li WY, Rockey JH, White P. Prostacyclin is the major prostaglandin synthesized by bovine retinal capillary pericytes in culture. Invest Ophthalmol Vis Sci. 1988 Oct;29(10):1511-6.
- Panteleontidis V, Detorakis ET, Pallikaris IG, Tsilimbaris MK. Latanoprost-dependent cystoid macular edema following uncomplicated cataract surgery in pseudoexfoliative eyes. Ophthalmic Surg Lasers Imaging. 2010 Mar 9;41(3):1-5. DOI: 10.3928/15428877-20100215-91
- Kohli P, Tripathy T, Patel CP. Macular edema [Internet]. Bethesda, MD: US National Library of Medicine; c2024 [cited 2025 January]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK576396/
- Reis A, Birnbaum F, Hansen LL, Reinhard T. Successful treatment of cystoid macular edema with valdecoxib. J Cataract Refract Surg. 2007 Apr;33(4):682-5. DOI: 10.1016/j.jcrs.2007.01.011
- Ota M, Nishijima K, Sakamoto A, et al. Optical coherence tomographic evaluation of foveal hard exudates in patients with diabetic maculopathy accompanying macular detachment. Ophthalmology. 2010 Oct;117(10):1996-2002. DOI: 10.1016/j.ophtha.2010.06.019
- Shukla UV, Tripathy K. Diabetic retinopathy [Internet]. Bethesda, MD: U.S. National Library of Medicine; c2023 [cited 2025 January]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560805/
- Antonetti DA, Klein R, Gardner TW. Diabetic retinopathy. N Engl J Med. 2012 Mar 29;366(13):1227-39. DOI: 10.1056/NEJMra1005073
- Frank RN. Diabetic retinopathy. N Engl J Med. 2004 Jan 1;350(1):48-58. DOI: 10.1056/NEJMra021678
- Zhang J, Zhang J, Zhang C, et al. Diabetic macular edema: current understanding, molecular mechanisms and therapeutic implications. Cells. 2022 Oct 25;11(21):3362. DOI: 10.3390/cells11213362
- Sakini ASA, Hamid AK, Alkhuzaie ZA, et al. Diabetic macular edema (DME): dissecting pathogenesis, prognostication, diagnostic modalities along with current and futuristic therapeutic insights. Int J Retina Vitreous. 2024 Oct 28;10(1):83. DOI: 10.1186/s40942-024-00603-y
- Rhoades W, Dickson D, Nguyen QD, Do DV. Management of macular edema due to central retinal vein occlusion – The role of aflibercept. Taiwan J Ophthalmol. 2017 Apr-Jun;7(2):70-6. DOI: 10.4103/tjo.tjo_9_17
- Braithwaite T, Nanji AA, Lindsley K, Greenberg PB. Anti-vascular endothelial growth factor for macular oedema secondary to central retinal vein occlusion. Cochrane Database Syst Rev. 2014 May 1;2014(5):CD007325. DOI: 10.1002/14651858.CD007325.pub3
- Haydinger CD, Ferreira LB, Williams KA, Smith JR. Mechanisms of macular edema. Front Med (Lausanne). 2023 Mar 7;10:1128811. DOI: 10.3389/fmed.2023.1128811
- Koronis S, Stavrakas P, Balidis M, Kozeis N, Tranos PG. Update in treatment of uveitic macular edema. Drug Des Devel Ther. 2019 Feb 19;13:667-80. DOI: 10.2147/DDDT.S166092
- Artifoni M, Rothschild PR, Brézin A, Guillevin L, Puéchal X. Ocular inflammatory diseases associated with rheumatoid arthritis. Nat Rev Rheumatol. 2014 Feb;10(2):108-16. DOI: 10.1038/nrrheum.2013.185
- Gupta A, Tripathy K. Central serous chorioretinopathy [Internet]. Bethesda, MD: U.S. National Library of Medicine; c2023 [cited 2025 January]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK558973/
- Iacono P, Battaglia Parodi M, Falcomatà B, Bandello F. Central serous chorioretinopathy treatments: a mini review. Ophthalmic Res. 2015;55(2):76-83. DOI: 10.1159/000441502
- Schalnus R. Topical nonsteroidal anti-inflammatory therapy in ophthalmology. Ophthalmologica. 2003 Mar-Apr;217(2):89-98. DOI: 10.1159/000068563
- Chastain JE, Sanders ME, Curtis MA, et al. Distribution of topical ocular nepafenac and its active metabolite amfenac to the posterior segment of the eye. Exp Eye Res. 2016 Apr;145:58-67. DOI: 10.1016/j.exer.2015.10.009
- Singhal D, Nanda A, Kanungo S, Sahoo K, Mohapatra S. A comparative analysis of topical corticosteroids and non-steroidal anti-inflammatory drugs to control inflammation and macular edema following uneventful phacoemulsification. Indian J Ophthalmol. 2022 Feb;70(2):425-33. DOI: 10.4103/ijo.IJO_1612_21
- Sandoval HP, De Castro LE, Vroman DT, Solomon KD. Evaluation of 0.4% ketorolac tromethamine ophthalmic solution versus 0.5% ketorolac tromethamine ophthalmic solution after phacoemulsification and intraocular lens implantation. J Ocul Pharmacol Ther. 2006 Aug;22(4):251-7. DOI: 10.1089/jop.2006.22.251
- Brooks HL, Caballero Jr S, Newell CK, et al. Vitreous levels of vascular endothelial growth factor and stromal-derived factor 1 in patients with diabetic retinopathy and cystoid macular edema before and after intraocular injection of triamcinolone. Arch Ophthalmol. 2004 Dec;122(12):1801-7. DOI: 10.1001/archopht.122.12.1801
- Simone JN, Whitacre MM. Effects of anti-inflammatory drugs following cataract extraction. Curr Opin Ophthalmol. 2001 Feb;12(1):63-7. DOI: 10.1097/00055735-200102000-00011
- Yang Y, Bailey C, Holz FG, et al. Long-term outcomes of phakic patients with diabetic macular oedema treated with intravitreal fluocinolone acetonide implants. Eye (Lond). 2015 Sep;29(9):1173-80. DOI: 10.1038/eye.2015.98
- Juthani VV, Clearfield E, Chuck RS. Non-steroidal anti-inflammatory drugs versus corticosteroids for controlling inflammation after uncomplicated cataract surgery. Cochrane Database Syst Rev. 2017 Jul 3;7(7):CD010516. DOI: 10.1002/14651858.CD010516.pub2
- Mayer WJ, Kurz S, Wolf A, et al. Dexamethasone implant as an effective treatment option for macular edema due to Irvine-Gass syndrome. J Cataract Refract Surg. 2015 Sep;41(9):1954-61. DOI: 10.1016/j.jcrs.2015.10.025
- Feroze KB, Zeppieri M, Khazaeni LSteroid induced glaucoma [Internet]. Bethesda, MD: U.S. National Library of Medicine; c2023 [cited 2025 January]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430903/
- Mohd Nasir NA, Agarwal R, Krasilnikova A, Sheikh Abdul Kadir SH, Iezhitsa I. Effect of dexamethasone on the expression of MMPs, adenosine A1 receptors and NFKB by human trabecular meshwork cells. J Basic Clin Physiol Pharmacol. 2020 Jul 22;31(6). DOI: 10.1515/jbcpp-2019-0373
- Sheppard JD, Comstock TL, Cavet ME. Impact of the topical ophthalmic corticosteroid loteprednol etabonate on intraocular pressure. Adv Ther. 2016 Apr;33(4):532-52. DOI: 10.1007/s12325-016-0315-8
- Nuyen B, Weinreb RN, Robbins SL. Steroid-induced glaucoma in the pediatric population. J AAPOS. 2017 Feb;21(1):1-6. DOI: 10.1016/j.jaapos.2016.09.026
- Armaly MF, Becker B. Intraocular pressure response to topical corticosteroids. Fed Proc. 1965 Nov-Dec;24(6):1274-8.
- Tripathi RC, Parapuram SK, Tripathi BJ, Zhong Y, Chalam KV. Corticosteroids and glaucoma risk. Drugs Aging. 1999 Dec;15(6):439-50. DOI: 10.2165/00002512-199915060-00004
- Moss EB, Buys YM, Low SA, et al. A randomized controlled trial to determine the effect of inhaled corticosteroid on intraocular pressure in open-angle glaucoma and ocular hypertension: the ICOUGH study. J Glaucoma. 2017 Feb;26(2):182-6. DOI: 10.1097/IJG.0000000000000429
- Razeghinejad MR, Katz LJ. Steroid-induced iatrogenic glaucoma. Ophthalmic Res. 2012;47(2):66-80. DOI: 10.1159/000328630
- Lai JS, Tham CC, Chan JC. The clinical outcomes of cataract extraction by phacoemulsification in eyes with primary angle-closure glaucoma and co-existing cataract: a prospective case series. J Glaucoma. 2006 Feb;15(1):47-52. DOI: 10.1097/01.ijg.0000196619.34368.0a
- Song MK, Sung KR, Shin JW, Jo YH, Won HJ. Glaucomatous progression after lens extraction in primary angle-closure disease spectrum. J Glaucoma. 2020 Aug;29(8):711-17. DOI: 10.1097/IJG.0000000000001537
- Song MK, Shin JW, Sung KR. Factors associated with deterioration of primary angle closure after lens extraction. J Clin Med. 2022 May 2;11(9):2557. DOI: 10.3390/jcm11092557
- Ouyang Y, Keane PA, Sadda SR, Walsh AC. Detection of cystoid macular edema with three-dimensional optical coherence tomography versus fluorescein angiography. Invest Ophthalmol Vis Sci. 2010 Oct;51(10):5213-8. DOI: 10.1167/iovs.09-4635
- Nakayama LF, Zago Ribeiro L, de Oliveira JAE, et al. Fairness and generalizability of OCT normative databases: a comparative analysis. Int J Retin Vitr. 2023 Aug 21;9(1):48. DOI: 10.1186/s40942-023-00459-8
- Abazari A, Allam SS, Adamus G, Ghazi NG. Optical coherence tomography findings in autoimmune retinopathy. Am J Ophthalmol. 2012 Apr;153(4):750-6, 756.e1. DOI: 10.1016/j.ajo.2011.09.012



